As it so happens, someone much smarter than me has already written on this topic, so check out Paul Sax’s post: https://blogs.jwatch.org/hiv-id-observations/index.php/the-curious-case-of-m184v-part-2-and-more/2019/09/08/
There is no doubt that anti-retroviral therapy has been one of the greatest accomplishments in HIV care. Nowadays, HIV is essentially a chronic disease that is easily managed with combined therapy, usually with a single pill (looking at Triumeq and Biktarvy). Having said that, mutations in HIV therapy are common, conferring resistance to several classes of anti-retrovirals. There are many resistance mutations that exist out there, but for today I will focus on perhaps my “favorite” mutation, the M184V mutation.
To go back a bit, the HIV-1 reverse transcriptase enzyme is actually prone to errors during viral replication. This specific mutation is the result of a change from methionine to valine at residue 184 in the RT gene (1), which in turn confers resistance to several drugs in the nucleoside reverse transcriptase inhibitor (NRTIs) family (i.e. emtricitabine, lamivudine, abacavir). In particular, this mutation occurs in setting of non-suppressive therapy with either 3TC or FTC-containing regimen (2) with one study citing the appearance of this resistance within 12 weeks of therapy with 3TC. The interesting tid bit of this mutation is the fact that it actually increases the susceptibility of certain NRTIs and decreases overall viral fitness. One review noted this mutation alters several mechanisms relating to RT function, including decreased RT processivity, increasing RT fidelity (i.e. reduces spontaneous mutation), and reducing nucleotide dependent primer unblocking (3). Further, the effects of M184V mutation was not the same across all NRTIs. For instance, one study of 27,000 samples (4) evaluated the NRTI phenotypic susceptibility and compared them to individual TAMs with or without M184V mutation and wild type viruses. Notably, those with the mutation remained sensitized to zidovudine, stavudine, and tenofovir. This was further confirmed in another study evaluating the patterns of cross resistance among NRTI drugs, which used a large database of clinical samples (5). Here, they found those with M184V mutation had hypersusceptibility to zidovudine, tenofovir, stavudine, and adevofir. This was present even with additional TAM mutations:
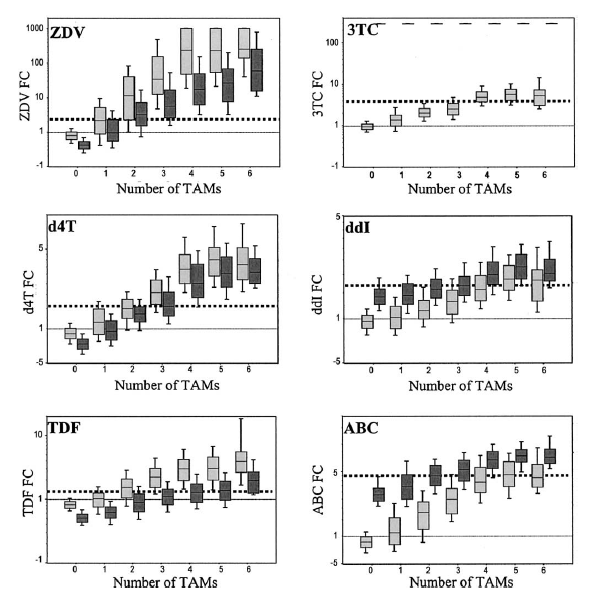
In another study of 180 patient samples (6) who were 3TC resistant, cross-resistance with other NRTIs was actually a rare occurrence:
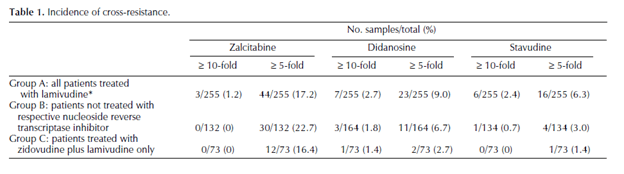

Cross-resistance to ddC, DDI, and D4T was likely associated with mutations other than M184V. In other words, the M184V mutation confers resistance to 3TC and FTC, however it does not apply to all NRTIs equally.
Clinical Data
One of the other interesting things about this particular mutation is that resistance to FTC or 3TC do not seem to translate into clinical practice. In other words, these drugs seem to retain some sort of anti-viral activity even in the face of M184V. One prospective, open label randomized (7) study evaluated lamivudine monotherapy vs no ART in patients with lamivudine resistance virus. Primary outcomes were occurrence of immunological or clinical failure. 29 patients in each group were evaluated, with treatment failure occurring in 20/29 patients (69%, 95% CI 51-83%) in the no ART group vs 12/29 patients (41%, 95% CI 26-59%) in the lamivudine monotherapy group at 48 weeks (p-value 0.061). The estimated time to clinical failure was also significantly shorter in the lamivudine group:
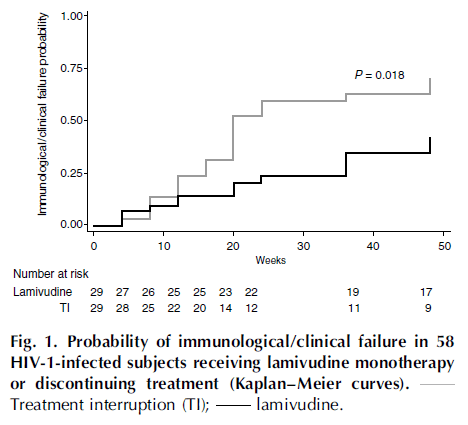
Further, patients with lamivudine therapy were less likely to have a drop in CD4 cells and were more likely to have lower viral loads:
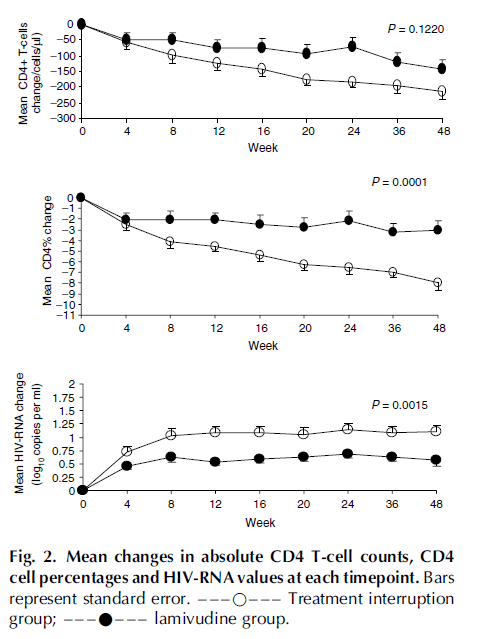
This suggest that, despite a mutation conferring resistance to lamivudine, it has some degree of antiviral activity in comparison to treatment interruption. In a prospective evaluation of 6 patients with M184V mutation (8) antiretroviral therapy with lamivudine was withdrawn, but continued with all other components. Notably, the median plasma HIV viral load increased 0.5 log10 copies/mL above baseline at 6 weeks following lamivudine withdrawal (p-value 0.04) despite the lack of reversion to WT genotype in these subjects. Following resumption of lamivudine, plasma HIV RNA levels decreased to baseline in 3 subjects.
A retrospective study (9) evaluated time to virological failure or treatment discontinuation between M184V negative and positive groups when getting lamivudine based dual therapy (either protease inhibitors or integrase inhibitor). While patients with M184V were more likely older, have HCV co-infection, had had HIV for a lot longer and been on ART longer than the negative mutation patients, the estimated proportion of patients free from virological failure was similar between groups:
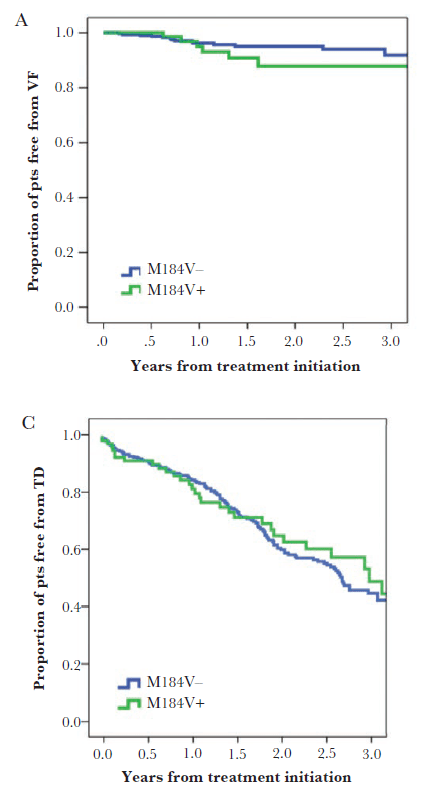
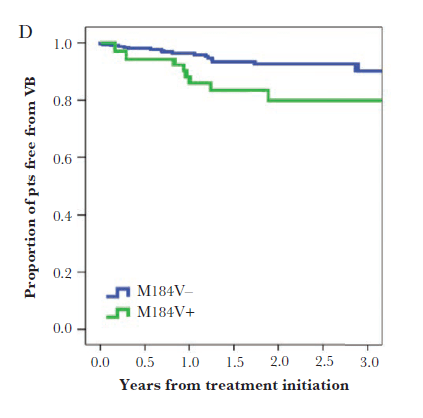
Though notably, M184V patients were more likely to have viral blips when compared to those without the mutation:
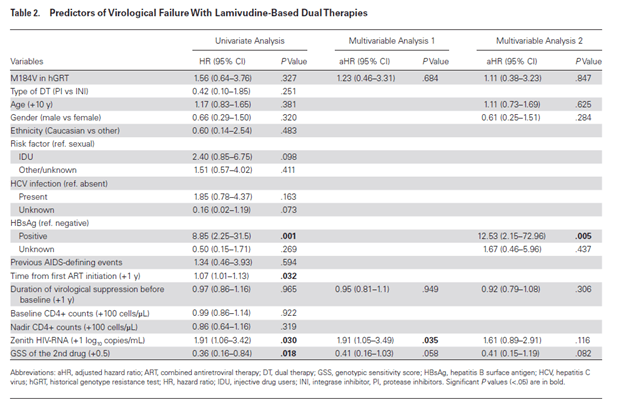
In multivariate analysis, presence of M184V mutation was not a predictor for virological failure with lamivudine based dual therapy:
Further data comes from an abstract presented at IDSA in 2015 (10), that evaluated transitioning virally suppressed patients with 2 prior failed regimens to FTC/TAF/EVG/c + DRV. Patients were randomized in a 2:1 fashion to open label FTC/TAF/EVG/c + DRV or their baseline regimen. 85% of patients in the experimental group and 78% of the baseline group had an M184V mutation. Outcomes were comparable between these 2 groups:
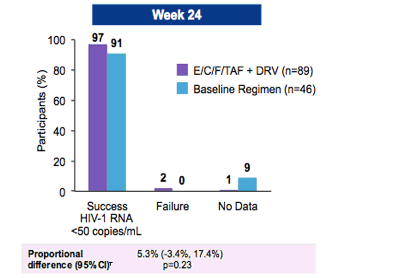
So these data suggest that, in isolation, M184V mutation in those who are virally suppressed may not lead to virological failure. So what can you do in terms of what regimen to pick in those with an isolated M184V mutation? The concern is, of course, the lack of an active “third agent” which may be addressed, as highlighted in the above study (10) by adding another agent. But given that there is some activity of FTC against the virus despite the mutation, there may not be a need to add another agent to the mix. In other words, can we give a regimen that involves something like FTC/TAF + DTG or FTC/TAF/BIC?
A phase 3, open label, parallel-group trial (11) evaluated the efficacy and safety of DTG + 2 NRTIs vs LPV/r + 2 NRTIs in patients who had failed therapy with 2 NRTIs + 1 NNRTI. Patients were randomized to either DTG or LPV/r and 2 investigator-selected NRTIs (at least one being active by genotype testing). Primary outcome was the proportion of patients archiving viral suppression at 48 weeks. 84% of patients in the DTG group and 81% of patients in the LPV/r group had an M184V mutation on at randomization.

Overall, 84% of the patients in the DTG group achieved viral suppression compared to 70% of those in the LPV/r group (adjusted difference 13.8%, 95% CI 7.3-20.3). DTG achieved higher response rates regardless of the presence or absence of M184V and the use of either 3TC/FTC.

This suggest that in patients who failed therapy with at least isolated M184V, using DTG + an active NRTI (something like tenofovir) will achieve viral suppression.
In patients who are already suppressed, switching regimens is also a reasonable approach. A multicenter, double blind, non-inferiority trial (12) evaluated virologically suppressed patients with NRTI resistance (including M184V) who were on DTG + either FTC/TAF or FTC/TDF. Patients were randomized in a 1:1 fashion to switch to BIC/FTC/TAF or DTG + FTC/TAF for 48 weeks plus a matching placebo. Primary endpoint was percentage of patients with viral load of >50 copies/mL at week 48. 284 patients were randomized to BIC/FTC/TAF and 281 were randomized to DTG + FTC/TAF. Overall, 0.4% of patients in the BIC/FTC/TAF group had viral load of >50 copies/mL compared to 1.1% of the patients in the DTG + FTC/TAF group:
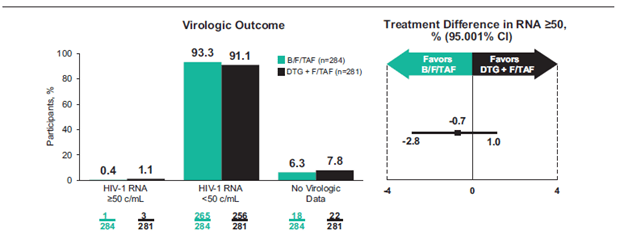
Further, the results were similar despite the baseline resistance, including M184V:
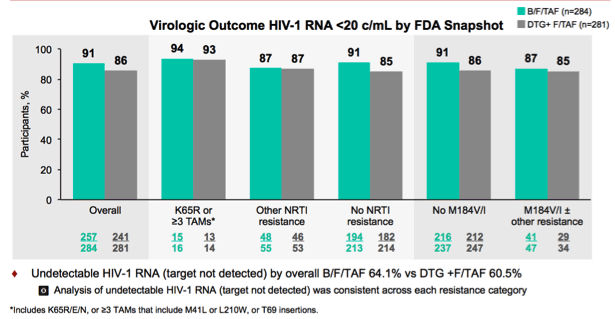
One notable thing was that 21 patients had both resistance to tenofovir (by either K65R mutation or lots of TAMs) or M184V (13 in the BIC arm and 8 in the DTG arm). No treatment failure occurred at 48 weeks.
TL;DR
- M184V mutation confers resistance to 3TC and FTC (lamivudine, emtricitabine)
- Viruses with this mutation are less fit
- It causes hypersusceptibility to tenofovir, zidovudine, and stavudine
- 3TC/FTC retains some activity against viruses with these mutations
- If a virus only has M184V mutation, an integrase-based regimen that includes another active NRTI is a reasonable approach for those who are suppressed or those who fail another first line therapy (i.e. can use FTC/TAF + DTG, BIC/FTC/TAF, or EVG/c/FTC/TAF) without necessarily adding another agent
References:
- Diallo K, Götte M, Wainberg MA. Molecular impact of the M184V mutation in human immunodeficiency virus type 1 reverse transcriptase. Antimicrob Agents Chemother. 2003;47(11):3377-3383. doi:10.1128/aac.47.11.3377-3383.2003
- Schuurman R, Nijhuis M, van Leeuwen R, et al. Rapid changes in human immunodeficiency virus type 1 RNA load and appearance of drug-resistant virus populations in persons treated with lamivudine (3TC). J Infect Dis. 1995;171(6):1411-1419. doi:10.1093/infdis/171.6.1411
- Gallant JE. The M184V mutation: what it does, how to prevent it, and what to do with it when it’s there. AIDS Read. 2006;16(10):556-559.
- Ross L, Parkin N, Chappey C, et al. Phenotypic impact of HIV reverse transcriptase M184I/V mutations in combination with single thymidine analog mutations on nucleoside reverse transcriptase inhibitor resistance. AIDS. 2004;18(12):1691-1696. doi:10.1097/01.aids.0000131355.44834.e4
- Whitcomb JM, Parkin NT, Chappey C, Hellmann NS, Petropoulos CJ. Broad nucleoside reverse-transcriptase inhibitor cross-resistance in human immunodeficiency virus type 1 clinical isolates. J Infect Dis. 2003;188(7):992-1000. doi:10.1086/378281
- Miller V, Stürmer M, Staszewski S, et al. The M184V mutation in HIV-1 reverse transcriptase (RT) conferring lamivudine resistance does not result in broad cross-resistance to nucleoside analogue RT inhibitors. AIDS. 1998;12(7):705-712. doi:10.1097/00002030-199807000-00006
- Castagna A, Danise A, Menzo S, et al. Lamivudine monotherapy in HIV-1-infected patients harbouring a lamivudine-resistant virus: a randomized pilot study (E-184V study). AIDS. 2006;20(6):795-803. doi:10.1097/01.aids.0000218542.08845.b2
- Campbell TB, Shulman NS, Johnson SC, et al. Antiviral activity of lamivudine in salvage therapy for multidrug-resistant HIV-1 infection. Clin Infect Dis. 2005;41(2):236-242. doi:10.1086/430709
- Gagliardini R, Ciccullo A, Borghetti A, et al. Impact of the M184V Resistance Mutation on Virological Efficacy and Durability of Lamivudine-Based Dual Antiretroviral Regimens as Maintenance Therapy in Individuals With Suppressed HIV-1 RNA: A Cohort Study. Open Forum Infect Dis. 2018;5(6):ofy113. Published 2018 May 15. doi:10.1093/ofid/ofy113
- Gregory Huhn, Pablo Tebas, Joel Gallant, Timothy Wilkin, Andrew Cheng, Mingjin Yan, Christian Callebaut, Marshall Fordyce, Moupali Das, Scott Mccallister, Strategic Simplification: The Efficacy and Safety of Switching to Elvitegravir/Cobicistat/Emtricitabine/Tenofovir Alafenamide (E/C/F/TAF) plus Darunavir (DRV) in Treatment-Experienced HIV-1 Infected Adults (NCT01968551), Open Forum Infectious Diseases, Volume 2, Issue suppl_1, December 2015, 726, https://doi.org/10.1093/ofid/ofv131.79
- Aboud M, Kaplan R, Lombaard J, et al. Dolutegravir versus ritonavir-boosted lopinavir both with dual nucleoside reverse transcriptase inhibitor therapy in adults with HIV-1 infection in whom first-line therapy has failed (DAWNING): an open-label, non-inferiority, phase 3b trial. Lancet Infect Dis. 2019;19(3):253-264. doi:10.1016/S1473-3099(19)30036-2
- Paul E Sax, MD, Jürgen K Rockstroh, MD, Anne F Luetkemeyer, MD, Yazdan Yazdanpanah, MD, Douglas Ward, MD, Benoit Trottier, MD, Armin Rieger, MD, Hui Liu, MD PhD, Rima Acosta, BS, Sean E Collins, MD MS, Diana M Brainard, MD, Hal Martin, MD MPH, 380-4030 Investigators, Switching to bictegravir, emtricitabine, and tenofovir alafenamide in virologically suppressed adults with HIV, Clinical Infectious Diseases, , ciaa988, https://doi.org/10.1093/cid/ciaa988

1 comments on “M184V – The Friendly Mutation?”