Candida and staphylococcus are two fairly common isolates of bloodstream infections, as well as part of our own microbiome. These two organisms also instill the fear of God into me, given how sticky they can be. I’ve talked a lot about staphylococcus and endocarditis in general, so candida endocarditis gets the spotlight for this post. I’ll focus on antifungal therapy, since its a bit more interesting.
Let’s look at the guidelines as a launching point. The IDSA guidelines mention that optimum therapy for both native and prosthetic valve endocarditis is that of valve replacement and a long-course of antifungal therapy (1). They advocate the use of lipid formulations of amphotericin B and echinocandins, with higher doses of echinocandins being “thought necessary” to treat endocarditis. The source of this recommendation is a 48 patient case series, of which those with endocarditis and osteomyelitis could get 100mg of caspofungin daily up to 150mg (usual dose is 50mg, 2). While the overall success rate was 81%, it should be noted that there were a total of 7 patients in the higher dose group, of which only 3 had endocarditis, and treatment success for endocarditis was 33%. Looking at the guidelines from the AHA gives you an insight into the lack of data with regards to medical management (3): “Historically, 2 treatment doctrines have prevailed in fungal IE despite the lack of prospective trials conducted to define the most appropriate therapy: Fungal IE is a “stand-alone indication” for surgical management of an infected valve; and amphotericin B, a fungicidal agent, is the initial drug of choice for fungal IE. Because of the alarming mortality rate associated with fungal IE and the availability of newer antifungal drugs, in particular fungicidal drugs like the echinocandins, a re-evaluation of these principles seems to be in order.” In other words, not terribly helpful since no one really knows what they’re doing. Part of the issue with therapy deals with the risk of relapse, and biofilm formation. As some people are aware, the production of biofilms makes penetration of antibiotics incredibly difficult, which is why endocarditis tends to be treated for such a long time; good luck trying to sneak a high amount of antibiotic into an area that is difficult to reach.
This phenomenon seems to have some clinical implications. An interesting paper describes a patient with fungal endocarditis with C. parapsilosis who underwent valve replacement (4). The infected valve was incubated with two different concentrations of amphotericin B. Candida sampled from the excised vegetation continued to grow up to a week later:

One study evaluated the impact of biofilms on antifungal susceptibility in a bioprosthetic biofilm model (5). Compared to the planktonically grown candida, the MIC in the biofilm Candida for the azole medications was significantly higher:

Notably, the echinocandins and the liposomal versions of amphotericin B retained significant activity against candida.
The best example comes from an animal study that compared the efficacy of amphotericin B, fluconazole, or fluconazole + flucytosine for candida endocarditis, pyelonephritis, and endophthalmitis (6). A relatively high percentage of amphotericin B was able to penetrate vegetation compared to fluconazole:
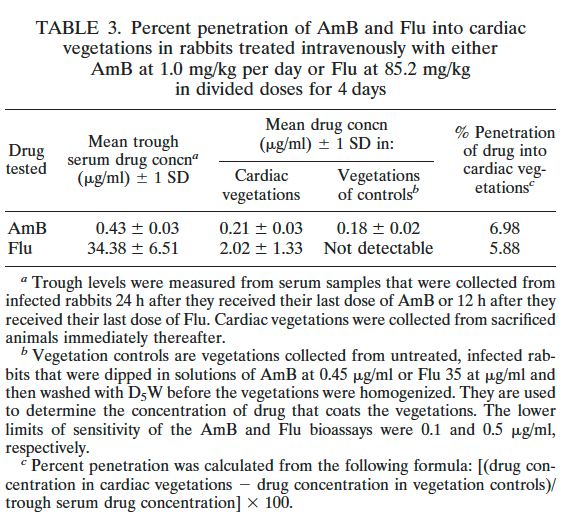
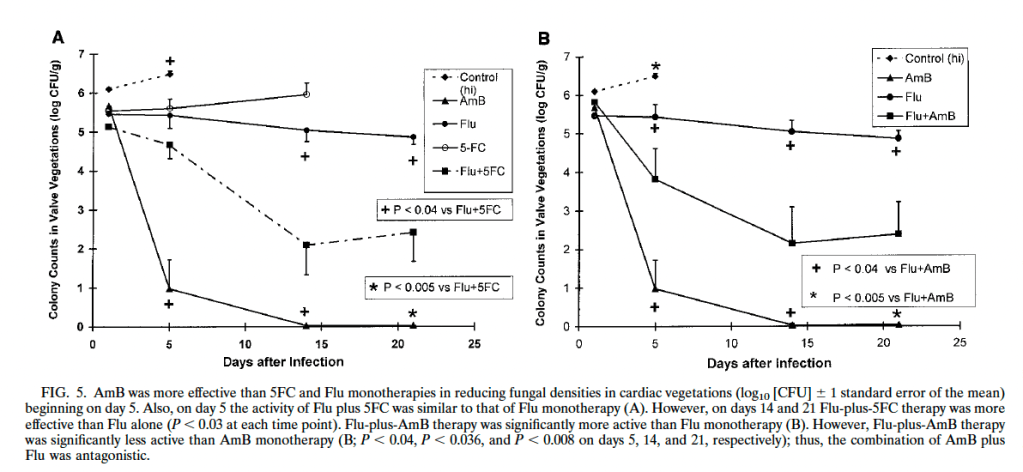
When looking at colony counts within vegetations, amphotericin B deoxycholate tended to decrease colony counts faster than fluconazole and flucytosine, and it was more effective throughout the days of treatment. Further, when combining fluconazole and amphotericin B, it seemed to have an antagonistic effect as the combination performed worse than amphotericin B alone:
The best clinical data comes from a retrospective cohort of 294 patients with candida bloodstream infection, where isolates were compared based on their ability to form biofilms (7). Multivariate analysis found that infection by biofilm-producing candida species as an independent risk factor for mortality:

Indeed, the raw mortality rate was much higher in the biofilm producing strains, 70%, compared to 45.7% in the rest of the strains.
Surgery:
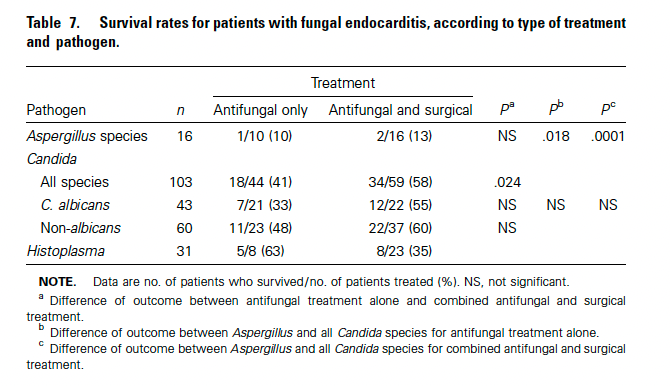
Given the biofilm issue and high mortality, it is no wonder why fungal endocarditis is thought of as being an absolute indication for valve replacement. A historical analysis of 270 patients with fungal endocarditis found a crude mortality of 72% (8). When looking at those patients with Candida endocarditis, antifungal and surgical intervention was associated with improved odds of mortality:
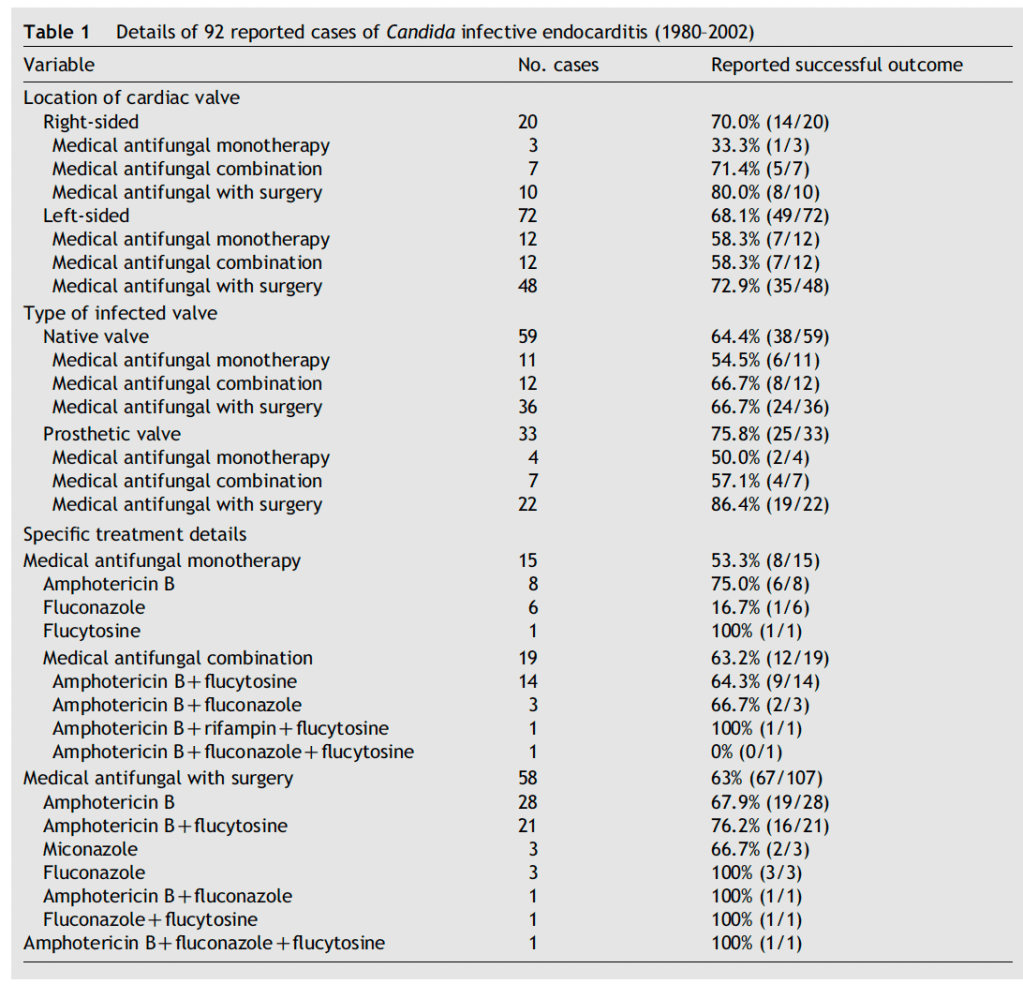
A meta-analysis of 105 patients (9) found that monotherapy was associated with the lowest odds of success, although this was mostly seen in the fluconazole group. Surgical intervention plus antifungal therapy yielded a success rate of 67%, which increased to 76% if amphotericin was combined with flucytosine:
Using 22 observational case-series, meta-regression analysis found that adjunctive therapy trended towards mortality benefit, while antifungal monotherapy trended towards increased mortality though these values did not reach statistical significance:

An interesting review of 140 cases found no difference in mortality between any administered antifungal regimens, however a binomial logistic regression found that cardiac surgery and effective anti-biofilm agents were associated with reduced mortality (10):
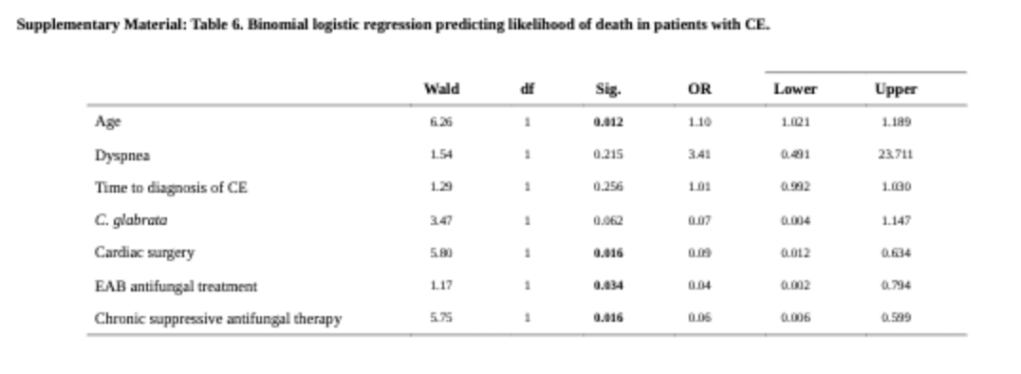
Medical Therapy
There is a benefit from surgery, however it should come to no surprise that mnay of the risk factors for candidemia and endocarditis (i.e. central line placement, TPN, antibiotic therapy) tend to occur in patients who are not ideal surgical candidates. If you look through the literature, you will no doubt find case reports highlighting how some authors managed to cure one patient with fluconazole or some combination of agents. For instance, a combination of fluconazole and caspofungin was successful in curing a case of PVE with C. glabrata without surgery after a 9 day course of amphotericin B (11). In another case, a combination of liposomal amphotericin B and caspofungin for three weeks was used for cure in native valve C. glabrata endocarditis (12). Moreover, caspofungin by itself has been used to cure mitral PVE with C. glabrata without suppressive therapy (13). Candida parapsilosis mural endocarditis has been cured with a combination of caspofungin and voriconazole for a total of 60 days with subsequent suppressive voriconazole (14). A summary of several papers is highlighted below (11-22):
| Age | Gender | Comorbidities | Candida spp | Type | Therapy | Surgery | Citation |
| 72 | male | Diabetes, CKD | C. glabrata | Prosthetic valve | fluconazole 400mg +amphotericin B -> fluconazole + Caspofungin | No | Lye et al 2005 |
| 84 | female | relapsing UTI | C. glabrata | Native valve | LAm-B + caspofungin for 3 weeks -> caspofungin x 8 weeks -> caspofungin thrice weekly | No | Jimenez-Esposito et al 2004 |
| 64 | female | Atrial fibrillation, mitral valve replacement | C. glabrata | Prosthetic valve | Fluconazole for 6 days -> caspofungin for 6 weeks | No | Rajendram et al 2004 |
| 59 | female | EtOH cirrhosis, pancreatitis | C. parapsilosis | Mural | fluconazole for 17 days, caspofungin for 72hrs, then voriconazole + caspofungin for 60 days and 9 months of suppressive voriconazole | No | Lopez-Ciudad et al 2006 |
| 68 | Male | AAA, ICM, sleep apnea, aortic valve replacement | C. albicans | Prosthetic valve | LAm-B for 47 days after surgery, fluconazole suppressive therapy | No | Aaron et al 2003 |
| 58 | male | aortic dissection | C. parapsilosis | Prosthetic valve | Amphotericin B for 27 days, fluconazole suppression | No | Czwerwiec et al 1992 |
| 47 | female | HIV, IVDU | C. albicans | Native valve | IV fluconazole for 4 weeks, 8 months oral fluconazole | No | Westling et al, 2004 |
| 47 | female | major depressive disorder | C. albicans | native valve | Fluconazole IV for 14 days -> 6 weeks | No | Wells et al, 1995 |
| 62 | female | Rheumatic heart disease | C. parapsilosis | Prosthetic valve | Amphotericin B followed by fluconazole for 8 months | No | Lejko-Zupanc and Kozelj, 1996 |
| 65 | female | HCV-cirrhosis, autoimmune hemolytic anemia | C. glabrata | Prosthetic valve | Anidulafungin for 2 weeks prior to surgery, and 4 weeks after | Yes | De Rosa et al, 2012 |
| 48 | male | CAD, mitral valve replacement | C. parapsilosis | Prosthetic valve | Amphotericin B + flucytosine for 5 weeks; amphotericin B + flucytosine during relapse, followed by fluconazole/ketoconazole | No | Zahid et al, 1994 |
| 21 | male | Diffuse polyposis, colectomy | C. albicans | Native valve, folliculitis | Fluconazole for 28 days, intravitreal amphotericin B | No | Venditti et al, 1992 |
The point is there is no great data source for antifungal therapy. And most of it comes from retrospective data, with the commonly used antifungals being echinocandins and amphotericin B.
One retrospective cohort of candida endocarditis compared the impact of either an amphotericin B based or echinocandin-based antifungal therapy in 70 patients (23). Around 46% of patients underwent surgical intervention, with overall in-hospital and one-year mortality being 36% and 59%, respectively. When comparing amphotericin B therapy and echinocandin therapy, mortality at 3 different time points was not statistically different (45% in-hospital mortality for amphotericin B vs 29% for echinocandin, 64% for amphotericin B at one year vs 69% at one year for echinocandins) despite the higher rate of surgical intervention in the amphotericin B group (55% vs 35%). A meta-analysis of 62 patients with candidal endocarditis found that 58% of patients who received only fluconazole survived compared to 84% in those who had an additional antifungal along with fluconazole (24). Moreover, the type of amphotericin B had an impact on mortality. Those who received amphotericin B had a 35% failure rate, compared to liposomal amphotericin which had a 22%. When the authors looked at 23 historical controls, they found that fluconazole-containing regimens plus valvular surgery had a survival rate of 91%. This suggests that fluconazole alone is likely not enough and combination therapy is the name of the game. It could also be that those who received azoles had a less toxic therapy compared to older amphotericin B formulations and had some degree of susceptibility to azoles.
A retrospective cohort of 46 cases of prosthetic valve candida endocarditis found 67% had combined antifungal therapy (usually liposomal amphotericin B and flucytosine), and 41% underwent surgical intervention (25). Relapse rates were 25% in the combined surgery-medical cohort who got maintenance compared to 50% in those who got only surgical therapy without maintenance therapy, 50% in those who got treated medically but got no maintenance therapy, and 15% in those who got medical therapy and maintenance therapy. One interesting bit was that, by multivariate analysis, there was a higher odds of 6-month survival if liposomal amphotericin B was used when compared to echinocandin therapy (aOR 13.52, 95% CI 1.03-838). A review of 140 cases of endocarditis found no difference in mortality between any administered antifungal regimens (10):
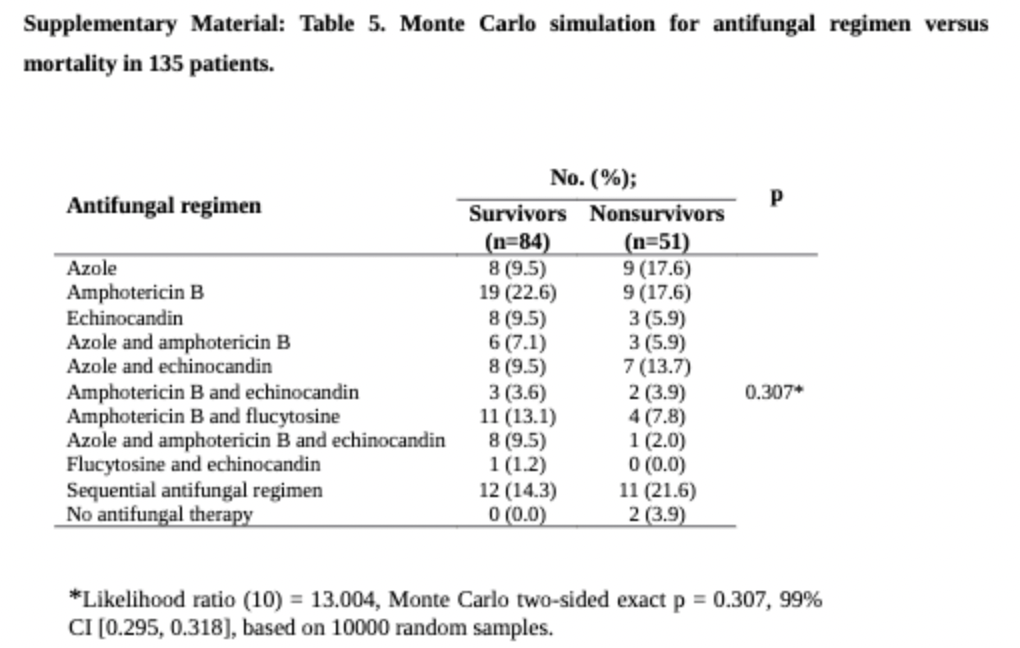
Moreover, chronic suppressive therapy was associated with reduced risk of death for both prosthetic and native valve candida endocarditis (RR 0.30, 95% CI 0.17-0.55). A monte-carlo simulation found that antifungal regimens that contained fluconazole showed improved survival in those who did not undergo surgery:

How long to treat remains a difficult question to answer, and it seems that long-term suppression with something oral such as fluconazole is a reasonable option, given the high relapse rates. For the most part, I’ve seen at least 6 weeks based on UpToDate and guidelines, but it may depend on whether source control is achieved or not. For now, either liposomal amphotericin B with or without flucytosine or high dose echinocandins followed by suppressive azole therapy. Unfortunately, the question of the efficacy of combined antifungal therapy remains to be answered. One review notes that for candida spp, amphotericin B and flucytosine have synergy in vivo and improved survival. This is in contrast to echinocandins and amphotericin B, which had an indifferent pattern in vivo but has had some improved tissue burden in combination (26):

A study evaluating different C. parapsilosis strains found that all combinations in the echinocandin resistant strains had some degree of antagonism, with the exception of the amphotericin B and fluconazole group (27). Moreover, flucytosine + amphotericin B OR voriconazole and fluconazole and voriconazole did not demonstrate antagonism:
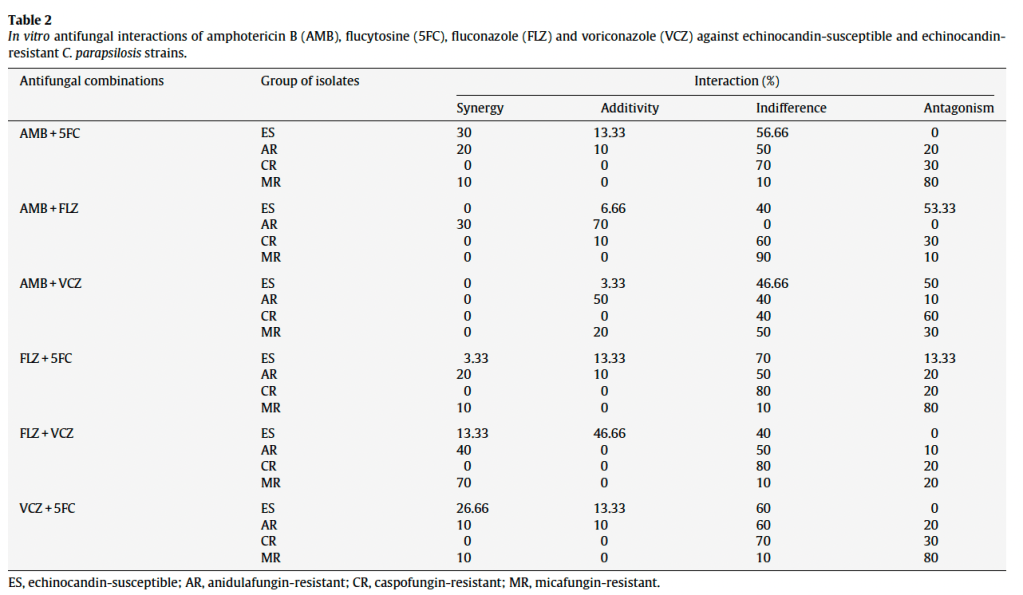
One letter also notes micafungin has some degree of synergy with amphotericin B (28):
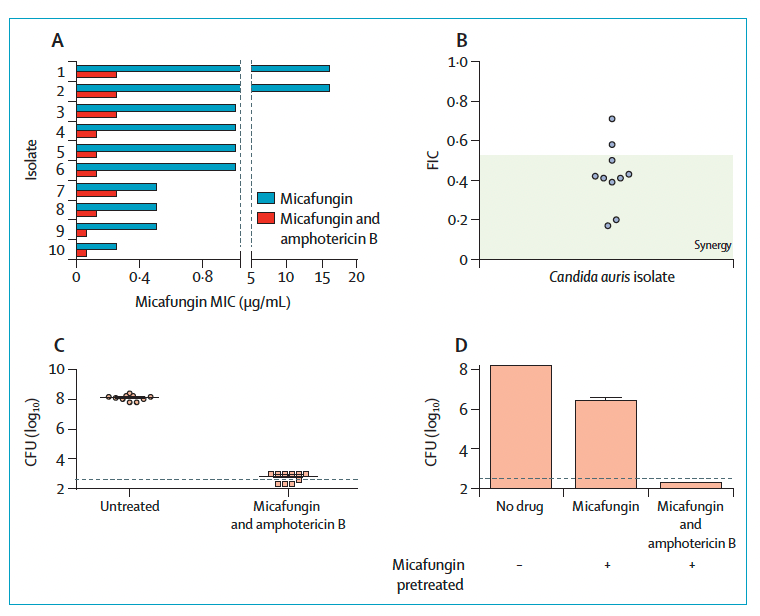
How this applies clinically remains to be seen, however given the lack of good data for dual-therapy in staph aureus endocarditis, I am not terribly optimistic.
References:
- Pappas PG, Kauffman CA, Andes DR, Clancy CJ, Marr KA, Ostrosky-Zeichner L, Reboli AC, Schuster MG, Vazquez JA, Walsh TJ, Zaoutis TE, Sobel JD. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2016 Feb 15;62(4):e1-50. doi: 10.1093/cid/civ933. Epub 2015 Dec 16. PMID: 26679628; PMCID: PMC4725385.
- Cornely OA, Lasso M, Betts R, Klimko N, Vazquez J, Dobb G, Velez J, Williams-Diaz A, Lipka J, Taylor A, Sable C, Kartsonis N. Caspofungin for the treatment of less common forms of invasive candidiasis. J Antimicrob Chemother. 2007 Aug;60(2):363-9. doi: 10.1093/jac/dkm169. Epub 2007 May 26. PMID: 17526917.
- Baddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Tleyjeh IM, Rybak MJ, Barsic B, Lockhart PB, Gewitz MH, Levison ME, Bolger AF, Steckelberg JM, Baltimore RS, Fink AM, O’Gara P, Taubert KA; American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young, Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and Stroke Council. Infective Endocarditis in Adults: Diagnosis, Antimicrobial Therapy, and Management of Complications: A Scientific Statement for Healthcare Professionals From the American Heart Association. Circulation. 2015 Oct 13;132(15):1435-86. doi: 10.1161/CIR.0000000000000296. Epub 2015 Sep 15. Erratum in: Circulation. 2015 Oct 27;132(17):e215. Erratum in: Circulation. 2016 Aug 23;134(8):e113. Erratum in: Circulation. 2018 Jul 31;138(5):e78-e79. PMID: 26373316.
- Rubinstein E, Noriega ER, Simberkoff MS, Rahal JJ Jr. Tissue penetration of amphotericin B in Candida endocarditis. Chest. 1974 Oct;66(4):376-7. doi: 10.1378/chest.66.4.376. PMID: 4416656.
- Kuhn DM, George T, Chandra J, Mukherjee PK, Ghannoum MA. Antifungal susceptibility of Candida biofilms: unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrob Agents Chemother. 2002 Jun;46(6):1773-80. doi: 10.1128/AAC.46.6.1773-1780.2002. PMID: 12019089; PMCID: PMC127206.
- Louie A, Liu W, Miller DA, Sucke AC, Liu QF, Drusano GL, Mayers M, Miller MH. Efficacies of high-dose fluconazole plus amphotericin B and high-dose fluconazole plus 5-fluorocytosine versus amphotericin B, fluconazole, and 5-fluorocytosine monotherapies in treatment of experimental endocarditis, endophthalmitis, and pyelonephritis due to Candida albicans. Antimicrob Agents Chemother. 1999 Dec;43(12):2831-40. doi: 10.1128/AAC.43.12.2831. PMID: 10582868; PMCID: PMC89573.
- Tumbarello M, Posteraro B, Trecarichi EM, Fiori B, Rossi M, Porta R, de Gaetano Donati K, La Sorda M, Spanu T, Fadda G, Cauda R, Sanguinetti M. Biofilm production by Candida species and inadequate antifungal therapy as predictors of mortality for patients with candidemia. J Clin Microbiol. 2007 Jun;45(6):1843-50. doi: 10.1128/JCM.00131-07. Epub 2007 Apr 25. PMID: 17460052; PMCID: PMC1933062.
- Ellis ME, Al-Abdely H, Sandridge A, Greer W, Ventura W. Fungal endocarditis: evidence in the world literature, 1965-1995. Clin Infect Dis. 2001 Jan;32(1):50-62. doi: 10.1086/317550. Epub 2000 Dec 12. PMID: 11118386.
- Steinbach WJ, Perfect JR, Cabell CH, Fowler VG, Corey GR, Li JS, Zaas AK, Benjamin DK Jr. A meta-analysis of medical versus surgical therapy for Candida endocarditis. J Infect. 2005 Oct;51(3):230-47. doi: 10.1016/j.jinf.2004.10.016. Epub 2004 Dec 1. PMID: 16230221.
- Giuliano S, Guastalegname M, Russo A, Falcone M, Ravasio V, Rizzi M, Bassetti M, Viale P, Pasticci MB, Durante-Mangoni E, Venditti M. Candida endocarditis: systematic literature review from 1997 to 2014 and analysis of 29 cases from the Italian Study of Endocarditis. Expert Rev Anti Infect Ther. 2017 Sep;15(9):807-818. doi: 10.1080/14787210.2017.1372749. Epub 2017 Sep 14. PMID: 28903607.
- Lye DC, Hughes A, O’Brien D, Athan E. Candida glabrata prosthetic valve endocarditis treated successfully with fluconazole plus caspofungin without surgery: a case report and literature review. Eur J Clin Microbiol Infect Dis. 2005 Nov;24(11):753-5. doi: 10.1007/s10096-005-0038-2. PMID: 16283214.
- Jiménez-Expósito MJ, Torres G, Baraldés A, Benito N, Marco F, Paré JC, Moreno A, Claramonte X, Mestres CA, Almela M, García de la María C, Pérez N, Schell WA, Corey GR, Perfect J, Jiménez de Anta MT, Gatell JM, Miró JM. Native valve endocarditis due to Candida glabrata treated without valvular replacement: a potential role for caspofungin in the induction and maintenance treatment. Clin Infect Dis. 2004 Oct 1;39(7):e70-3. doi: 10.1086/424018. Epub 2004 Sep 8. PMID: 15472836.
- Rajendram R, Alp NJ, Mitchell AR, Bowler IC, Forfar JC. Candida prosthetic valve endocarditis cured by caspofungin therapy without valve replacement. Clin Infect Dis. 2005 May 1;40(9):e72-4. doi: 10.1086/429322. Epub 2005 Mar 23. PMID: 15825018.
- López-Ciudad V, Castro-Orjales MJ, León C, Sanz-Rodríguez C, de la Torre-Fernández MJ, Pérez de Juan-Romero MA, Collell-Llach MD, Díaz-López MD. Successful treatment of Candida parapsilosis mural endocarditis with combined caspofungin and voriconazole. BMC Infect Dis. 2006 Apr 11;6:73. doi: 10.1186/1471-2334-6-73. PMID: 16608509; PMCID: PMC1458348.
- Aaron L, Therby A, Viard JP, Lahoulou R, Dupont B. Successful medical treatment of Candida albicans in mechanical prosthetic valve endocarditis. Scand J Infect Dis. 2003;35(5):351-2. doi: 10.1080/00365540310008357. PMID: 12875529.
- Czerwiec FS, Bilsker MS, Kamerman ML, Bisno AL. Long-term survival after fluconazole therapy of candidal prosthetic valve endocarditis. Am J Med. 1993 May;94(5):545-546. doi: 10.1016/0002-9343(93)90092-4. PMID: 8498400.
- Westling K, Thalme A, Julander I. Candida albicans tricuspid valve endocarditis in an intravenous drug addict: successful treatment with fluconazole. Scand J Infect Dis. 2005;37(4):310-1. doi: 10.1080/00365540410021207. PMID: 15804670.
- Wells CJ, Leech GJ, Lever AM, Wansbrough-Jones MH. Treatment of native valve Candida endocarditis with fluconazole. J Infect. 1995 Nov;31(3):233-5. doi: 10.1016/s0163-4453(95)80033-6. PMID: 8586845.
- Lejko-Zupanc T, Kozelj M. A case of recurrent Candida parapsilosis prosthetic valve endocarditis: cure by medical treatment alone. J Infect. 1997 Jul;35(1):81-2. doi: 10.1016/s0163-4453(97)91145-2. PMID: 9279731.
- De Rosa FG, D’Avolio A, Corcione S, Baietto L, Raviolo S, Centofanti P, Pasero D, Rinaldi M, Di Perri G. Anidulafungin for Candida glabrata infective endocarditis. Antimicrob Agents Chemother. 2012 Aug;56(8):4552-3. doi: 10.1128/AAC.00515-12. Epub 2012 May 29. PMID: 22644027; PMCID: PMC3421621.
- Venditti M, De Bernardis F, Micozzi A, Pontieri E, Chirletti P, Cassone A, Martino P. Fluconazole treatment of catheter-related right-sided endocarditis caused by Candida albicans and associated with endophthalmitis and folliculitis. Clin Infect Dis. 1992 Feb;14(2):422-6. doi: 10.1093/clinids/14.2.422. PMID: 1554827.
- Zahid MA, Klotz SA, Hinthorn DR. Medical treatment of recurrent candidemia in a patient with probable Candida parapsilosis prosthetic valve endocarditis. Chest. 1994 May;105(5):1597-8. doi: 10.1378/chest.105.5.1597. PMID: 8181369.
- Arnold CJ, Johnson M, Bayer AS, Bradley S, Giannitsioti E, Miró JM, Tornos P, Tattevin P, Strahilevitz J, Spelman D, Athan E, Nacinovich F, Fortes CQ, Lamas C, Barsic B, Fernández-Hidalgo N, Muñoz P, Chu VH. Candida infective endocarditis: an observational cohort study with a focus on therapy. Antimicrob Agents Chemother. 2015 Apr;59(4):2365-73. doi: 10.1128/AAC.04867-14. Epub 2015 Feb 2. PMID: 25645855; PMCID: PMC4356766.
- Smego RA Jr, Ahmad H. The role of fluconazole in the treatment of Candida endocarditis: a meta-analysis. Medicine (Baltimore). 2011 Jul;90(4):237-249. doi: 10.1097/MD.0b013e3182259d38. PMID: 21694646.
- Rivoisy C, Vena A, Schaeffer L, Charlier C, Fontanet A, Delahaye F, Bouza E, Lortholary O, Munoz P, Lefort A; French Mycoses Study Group and Grupo de Apoyo al Manejo de las Endocarditis en España (GAMES). Prosthetic Valve Candida spp. Endocarditis: New Insights Into Long-term Prognosis-The ESCAPE Study. Clin Infect Dis. 2018 Mar 5;66(6):825-832. doi: 10.1093/cid/cix913. PMID: 29077791.
- Ostrosky-Zeichner L. Combination antifungal therapy: a critical review of the evidence. Clin Microbiol Infect. 2008 May;14 Suppl 4:65-70. doi: 10.1111/j.1469-0691.2008.01983.x. PMID: 18430131.
- Chassot F, Venturini TP, Piasentin FB, Santurio JM, Svidzinski TIE, Alves SH. Activity of antifungal agents alone and in combination against echinocandin-susceptible and -resistant Candida parapsilosis strains. Rev Iberoam Micol. 2019 Jan-Mar;36(1):44-47. doi: 10.1016/j.riam.2018.07.007. Epub 2019 Jan 26. PMID: 30691952.
- Jaggavarapu S, Burd EM, Weiss DS. Micafungin and amphotericin B synergy against Candida auris. Lancet Microbe. 2020 Dec;1(8):e314-e315. doi: 10.1016/S2666-5247(20)30194-4. Epub 2020 Dec 7. PMID: 35544183.
