Robert Koch is probably one of the most important people in all of medicine. Arguably one of the fathers of microbiology, he popularized the idea of his “postulates” which are a set of criteria designed to assess whether a microorganism causes disease (1). His most well known work is with the bacteria bacillus anthracis aka anthrax. His work with the organism established many of his postulates. For instance, in Wollstein, modern-day Poland, he established a practice and noted that anthrax had killed roughly 530 people and 56,000 livestock in a time span of around 4 years (2). He designed inoculation studies, where he would inoculate mice with blood from a sheep that had died from anthrax. At autopsy, he found rod-shaped structures in blood, lymph nodes, and spleen. Inoculating a second mouse with blood from the spleen of the first mouse yielded similar results. Of course, blood laden with bacteria did not cause disease after some time. So Koch had to figure out a way to culture bacteria.
He inoculated the cornea of a rabbit with bacteria, and noted the aqueous humor turned turbid; in short, he turned aqueous humor into a culture medium. He would place splenic tissue into this aqueous humor to create a living environment and then seal bacteria in a concave slide, allowing him to study it. Moreover, he also saw that in optimal conditions (warm, moist, aerated environment) allowed bacteria to swell, elongate, and form long filaments which would acquire granules that developed spheres. When the filaments decompose, the spheres remain. When the fluid was dried and reconstituted with aqueous humor, bacteria emerged from the spheres. Koch had discovered spores, and figured this is how spores in the absence of bacteria, can cause disease (2). It is from Koch we get one of the most famous illustrations, anthrax at various stages (3):
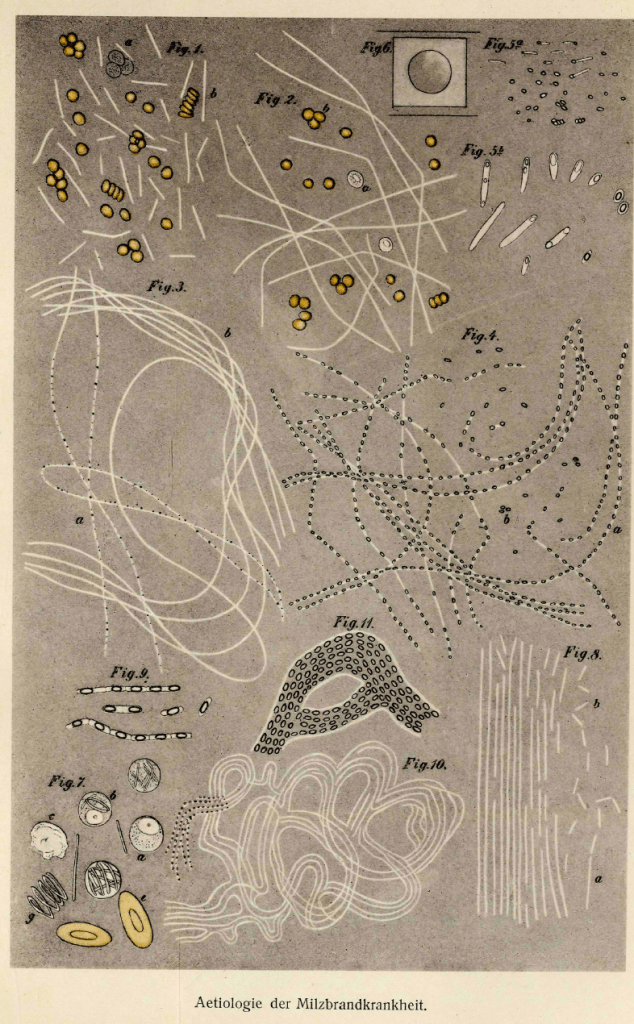
Microbiology
Besides being the name of a famous heavy metal band and a terrifying series of events in an already alarming year, anthrax is a gram-positive rod that exists in the environment as a spore and can remain viable in the soil for decades (4, 5). It is associated with both domesticated and wild animals, as well as animal products such as wool, hair, bone, meat, horns, and hides. Indeed, wool was such a common exposure it was known as “woolsorters disease.” In the environment, it survives as a spore, allowing it to survive for decades, including in bones of an archeological site or in the sugar cube of a world war 1 terrorist (6). Spores ingested by herbivores germinate within the animal to produce the virulent vegetative forms that replicate. As highlighted above, spores are generated from the bacterium and can remain behind even without live bacteria, allowing the infection to propagate (4). In humans, spores are either inhaled or introduced subcutaneously (see later). This organism obtains its virulence from the presence of a poly-D-glutamic acid capsule and the production of its infamous exotoxins. The capsule allows the organism to escape immune clearance (7, 8) while the toxins wreck everything else. These two toxins, the lethal toxin and edema toxin, are made up each of two different proteins; lethal factor-protective antigen, and edema factor-protective antigen (9):

The PA mediates cell binding and uptake of edema factor and lethal factor within the cell, usually by binding to tumor endothelial marker 8 or capillary morphogenesis 2. After binding, the N-terminal piece is cleaved and the PA piece remains cell-bound, allowing the binding site to bind either EF or LF and undergo endocytosis (7). The lethal factor is a zinc metalloproteinase enzyme that inactivates MAPKK, which impairs both innate and adaptive immune functions (8):

ET causes tissue edema while EF causes the alteration of transcriptional programs of target cells, leading to hypotension, vasodilatation, tachycardia, and reduced myocardial performance.
Manifestations:
The vast majority of anthrax cases tend to be cutaneous anthrax, where fever, regional lymphadenopathy occur with a vesicle rupturing and forming an ulcer and black eschar that is pathognomonic for anthrax (7). A more recent manifestation has been injection anthrax, similar to cutaneous anthrax but associated with IV drug abuse. The largest (and most detailed) description of cutaneous anthrax can be found in a paper by Herman Gold, where he highlights 117 cases of mostly the cutaneous variety (10; the pictures are worth the read). While common, I’d like to focus on inhalational anthrax, given the bioterrorism concerns. The largest outbreak occurred in the city of Sverdlovsk, where windborne spread of an aerosol of anthrax from a microbiological facility led to infection and the opportunity to figure out the pathophysiology behind inhalational anthrax (11). 42 autopsies were performed, with all having prominent and consistent lesions of hemorrhagic thoracic lymphadenitis and hemorrhagic mediastinitis (12). Moreover, spread of anthrax to the peribronchial lymph nodes by the lymphatic vessels was seen by the presence of organisms in the marginal sinus. Indeed, it is believed that upon inhalation, spores that are smaller than 5um are deposited into the alveoli and phagocytosed by alveolar macrophages, and subsequently transported to mediastinal lymph nodes (7). One interesting bit is that disseminated disease was seen in 39 cases, with gastrointestinal lesions or meningeal edema seen. Another autopsy report notes that the entire mediastinum was occupied by hemorrhagic edema infiltrating soft tissues (13). The lung parenchyma had a slightly increased consistency due to congestion and edema rather than pneumonitis. Moreover, diffuse hemorrhagic leptomeninigitis was noted in another specimen, with gram positive bacilli seen in lungs, meninges, liver, kidney, spleen, lung, and brain.
The accepted staging for inhalational anthrax is that proposed by Lucey and found in the latest edition of Mendel. Here, an early prodromal stage leads to an intermediate stage, followed by a late fulminant phase. In the literature, this is described as a “biphasic illness” with a flu-like illness occurring after an incubation of around 4 days, followed by rapid deterioration manifested by shock, hypoxemia, and death within 24hrs (7, 13). Indeed, the fulminant imaging changes are seen in this sequence, with widened mediastinum and pleural effusion progressing within 32 hours of admission:

One case series of 5 patients found all died within 5 days of illness onset (14):

The average duration of the acute stage is less than 24 hours. In a cohort study, fevers, dyspnea, fever, and disorientation were seen, with several pathological findings seen on autopsy reports:

A review of a century’s worth of inhalational anthrax mounting to a whooping 82 cases in the literature gives us the most detailed clinical description (15). Fevers (81%), abnormal lung findings (80%), chills (67%), tachycardia (66%), malaise (64%), cough (62%), and dyspnea (52%) were the most commonly cited symptoms. CXR was usually abnormal, with pleural effusions (69%) and widened mediastinum (51%) being present on admission, and 38% having developed meningoencephalitis. In this historical cohort, the prodromal phase lasted a mean of 4.1 days, while the fulminant phase lasted 1.1 days. A case series of 10 patients exposed to anthrax during the 2001 attack found the median time from exposure to onset of symptoms ranged from 4 to 6 days after exposure (16). All had fevers and chills, fatigue, malaise, and lethargy, as well as cough, dyspnea, nausea, vomiting, and pleuritic chest pain.

A cohort of 47 historical cases of anthrax was compared to the 11 cases of bioterrorism-related cases and matched to four controls with influenza and four controls with community-acquired pneumonia (17). Mortality was significantly higher for the naturally occurring cases (94% vs 45%), and signs such as vomiting, nausea, pallor, cyanosis, altered mental status, and raised hematocrit were more predictive of inhalational anthrax:
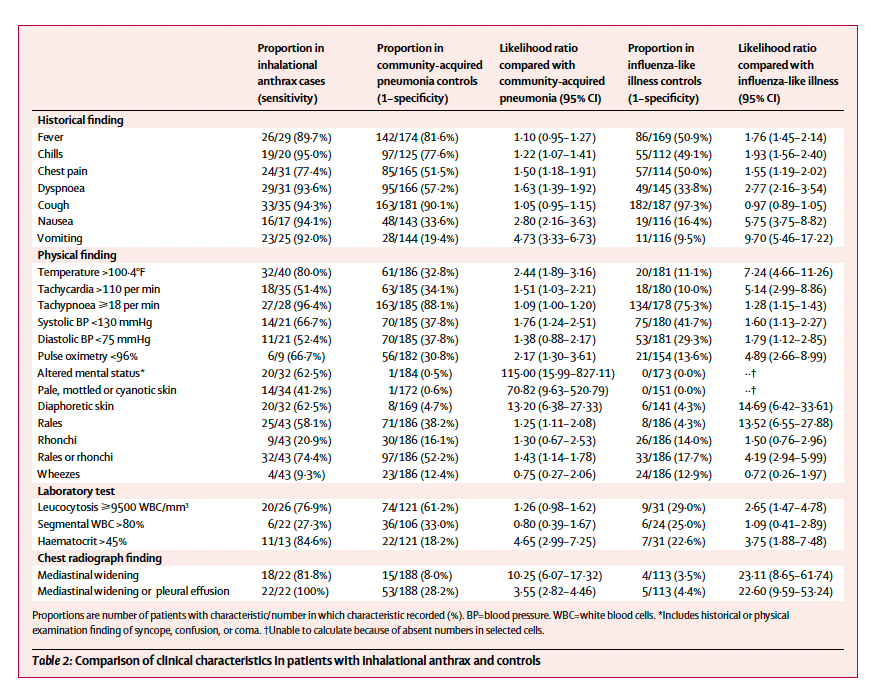
As seen above, the presence of mediastinal widening or pleural effusion yielded a 100% sensitivity for anthrax.
The interesting thing about anthrax is how uncommon the inhalational form is. Considering how both the US and Soviet Union were working on using it as a weapon, it’s surprising how little it is seen. This may be due to the influence of particle size on infectivity. Particles larger than 5um are captured in the upper airways and transported out, while those smaller than 5um reach the terminal bronchioles and alveoli, where they are phagocytosed inside macrophage and drain into mediastinal lymph nodes (5):

Individual anthrax spores range from 1.5 to 3um, however in nature they can clump and form aggregates up to 100um in diameter. As a result, these large particles do not make it into the alveoli and thus, no disease is seen. This may be the reason why inhalational anthrax is not seen in nature, but rather in situations where spores may be disrupted and disseminated in smaller particles.
Therapy:
The recommended antibiotic therapy includes a regimen of “two plus one” consisting of two bactericidal antibiotics of different classes, one of which is a beta-lactam, and a toxin inhibitor. This can be either a 1) quinolone + 2) carbapenem + 3) clindamycin or linezolid (8). Other options include doxycycline, piperacillin-tazobactam, other carbapenems, or rifampin. The guidelines by the CDC recommend intravenous ciprofloxacin, meropenem, and linezolid in the event the disease is complicated by meningitis, as these can all cross the CNS barrier (18). If no meningitis, then at least 2 drugs given IV for at least 2 weeks or the patient is stable, whichever is longer, is the recommendation. Beyond this, 60 days is the recommended duration of therapy to avoid relapse due to germination. Despite my assumption, most of the recommendations come from expert opinion and retrospective data. Regardless, there is ample in vitro data to guide antibiotic therapy. A review of 22 isolates found that most beta-lactams, quinolones, and tetracyclines were susceptible, however one glaring exception is any beta-lactam that is a second through third generation cephalosporin (19):
This was also seen in a study of 65 isolates tested against several antibiotics via broth microdilution method (20):
96 isolates of anthrax obtained from France between 1994 and 2000 also show that higher generation cephalosporins do not perform well here (21):

Similar results were obtained from a cohort of 110 strains ranging from vaccine strains to animal strains (22):

Moreover, time-kill studies suggest that quinolones, rifampin, vancomycin, and amoxicillin had similar kill activity (23), though it also shows ceftriaxone killing this particular strain though the MIC for this particular combination was 8 (i.e. not something I would use clinically).

Anthrax antiserum, based on the idea that the toxins wreck everything, have also been utilized. A review of both animal and human studies evaluated antitoxin therapy for inhalational anthrax and found that nonhuman primates treated with antitoxin had a higher likelihood of survival, even when serum protective antigen was detected up to 31 hours after exposure (24). Anthrasil, Thravixa, Raxibacumab, and anthrivig were all associated with improved survival. Antibiotic and antitoxin combination therapy was evaluated in 8 animal models, with survival rates being high up to 42 hours postexposure. Raxibacumab, a fully human monoclonal antibody directed against the protective antigen, was evaluated in a open-label, parallel-group, randomized animal trial in escalating doses of 1, 5, 10, or 20mg/kg of body weight on day -2 and 40mg/kg after spore challenge (25). Survival at day 14 was exclaimed, and in monkeys and rabbits exposed to spores, a dose of 10mg/kg improved survival at 14 days when compared to placebo.

For systemic illness, both 20mg/kg and 40mg/kg yielded improved mortality at day 28, however the difference between the two doses was not statistically significant (p=0.30).

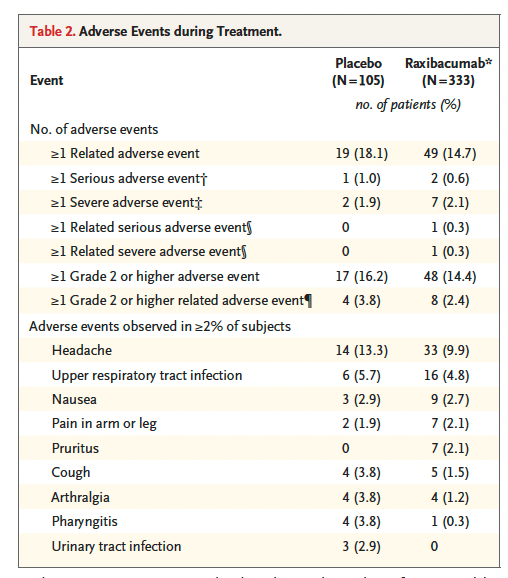
This antibody was tested in 333 human subjects at a dose of 40mg/kg, without any significant adverse events.
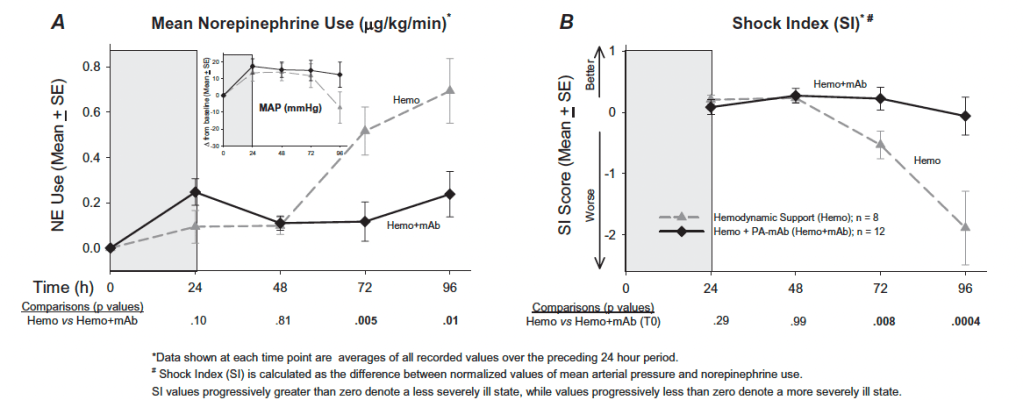
The benefit from antitoxin therapy may stem from the hemodynamic improvements it provides. One study infected beagles with lethal toxin and evaluated the impact of hemodynamic support alone, monoclonal antibody alone, or both together (26). The combination of antibody and hemodynamic support was more effective than antibody alone when therapy started at time 0, at 9 hours or 12 hours after infection (p = 0.004). Interestingly, however, then looking at hemodynamic parameters such as MAP or LVEF, the combination of hemodynamic support and antibodies yielded better parameters:

Shock index and norepinephrine use was also better in the combination cohort:
When looking at survival, the combination cohorts tended to have better outcomes across experiments:

Overall, combination therapy with multiple antibiotics and anthrax antiserum tends to provide the best benefit overall. This was seen in a review that evaluated both the pre-2001 and 2001 inhalational anthrax cases (15). Treatment with antibiotics or anthrax antiserum started during the prodromal phase (HR 0.09, p < 0.001), given antibiotics or combination antibiotic-antiserum therapy (HR 0.02, p < 0.001), or if they were among the US 2001 cases (HR 0.3, p=0.034) were associated with improved mortality. Increasing time to either antibiotic or antiserum therapy (HR 4.5, p=0.002), increasing time to pleural fluid drainage (HR 4.5, p=0.010), advancing age (HR 1.04, p=0.003), and development of meningoencephalitis (HR 7.5, p < 0.001) were all associated with higher mortality. Notably, mortality increased significantly with each day of delayed antibiotic therapy:

Historical data suggests that those patients that received both a bactericidal and protein synthesis inhibitor antibiotic therapy (27) had higher chances of survival (45% vs 28%, p =0.07), and combination therapy tended to have more favorable outcomes (mortality for one antibiotic, 70%, for 2, 25%, for three or more 5%). Two animal studies evaluated the combination of antibiotics and AIGIV+ at a 15U/kg dose (28). In the first study, either placebo + levofloxacin or AIGIV+ levofloxacin was studied either at 30, 36, 48, or 60hrs after spore exposure. The second study was similar, but the delay was at 60, 72, 84, and 96 hours. In the first study, none of the animals in the placebo-levofloxacin group died with the exception of one in the 60 hour group. In the second study, most of the animals died due to the extreme delay to therapy initiation. Despite this, even when the delay was up to 96 hours, the combination of AIGIV+ levofloxacin yielded a higher survival (71% vs 25%):

Interestingly, in both groups the presence of levofloxacin cleared anthrax bacteremia, suggesting its bactericidal effect.

Moreover, therapy with AIGIV leads to a reduction in the presence of toxins in the animals.

TL;DR
- Robert Koch’s tinkering with anthrax led to modern microbiology
- The cutaneous form is the most common, with the most lethal being inhalational
- The toxins, both lethal and edema toxins, are responsible for the pathophysiology and hemodynamic issues with anthrax
- Biphasic illness; prodromal flu-like illness followed by a fulminant course with people dying within 24hrs
- Mediastinal widening + pleural effusions
- Quinolones + carbapenems + linezolid + antitoxin is the therapy of choice.
References:
- Segre JA. What does it take to satisfy Koch’s postulates two centuries later? Microbial genomics and Propionibacteria acnes. J Invest Dermatol. 2013 Sep;133(9):2141-2. doi: 10.1038/jid.2013.260. Epub 2013 Jul 11. PMID: 23842116; PMCID: PMC3775492.
- Blevins SM, Bronze MS. Robert Koch and the ‘golden age’ of bacteriology. Int J Infect Dis. 2010 Sep;14(9):e744-51. doi: 10.1016/j.ijid.2009.12.003. Epub 2010 Apr 21. PMID: 20413340.
- Koch R. Die Ätiologie Der Milzbrand-Krankheit : Begründet Auf Die Entwicklungsgeschichte Des Bacillus Anthracis 1876. Leipzig: Barth; 1910.
- Sweeney DA, Hicks CW, Cui X, Li Y, Eichacker PQ. Anthrax infection. Am J Respir Crit Care Med. 2011 Dec 15;184(12):1333-41. doi: 10.1164/rccm.201102-0209CI. Epub 2011 Aug 18. PMID: 21852539; PMCID: PMC3361358.
- [edited by] Gerald L. Mandell, John E. Bennett, Raphael Dolin. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. Philadelphia, PA :Churchill Livingstone/Elsevier, 2010.
- Redmond C, Pearce MJ, Manchee RJ, Berdal BP. Deadly relic of the Great War. Nature. 1998 Jun 25;393(6687):747-8. doi: 10.1038/31612. PMID: 9655389.
- LaForce FM. Anthrax. Clin Infect Dis. 1994 Dec;19(6):1009-13; quiz 1014. doi: 10.1093/clinids/19.6.1009. PMID: 7888525.
- Artenstein AW, Opal SM. Novel approaches to the treatment of systemic anthrax. Clin Infect Dis. 2012 Apr;54(8):1148-61. doi: 10.1093/cid/cis017. PMID: 22438345.
- Sweeney DA, Hicks CW, Cui X, Li Y, Eichacker PQ. Anthrax infection. Am J Respir Crit Care Med. 2011 Dec 15;184(12):1333-41. doi: 10.1164/rccm.201102-0209CI. Epub 2011 Aug 18. PMID: 21852539; PMCID: PMC3361358.
- GOLD H. Anthrax; a report of one hundred seventeen cases. AMA Arch Intern Med. 1955 Sep;96(3):387-96. doi: 10.1001/archinte.1955.00250140109012. PMID: 13248274.
- Meselson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, Yampolskaya O. The Sverdlovsk anthrax outbreak of 1979. Science. 1994 Nov 18;266(5188):1202-8. doi: 10.1126/science.7973702. PMID: 7973702.
- Abramova FA, Grinberg LM, Yampolskaya OV, Walker DH. Pathology of inhalational anthrax in 42 cases from the Sverdlovsk outbreak of 1979. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2291-4. doi: 10.1073/pnas.90.6.2291. PMID: 8460135; PMCID: PMC46072.
- Plotkin SA, Brachman PS, Utell M, Bumford FH, Atchison MM. An epidemic of inhalation anthrax, the first in the twentieth century: I. Clinical features. 1960. Am J Med. 2002 Jan;112(1):4-12; discussion 2-3. doi: 10.1016/s0002-9343(01)01050-6. PMID: 11812400; PMCID: PMC7172370.
- Plotkin SA, Brachman PS, Utell M, Bumford FH, Atchison MM. An epidemic of inhalation anthrax, the first in the twentieth century: I. Clinical features. 1960. Am J Med. 2002 Jan;112(1):4-12; discussion 2-3. doi: 10.1016/s0002-9343(01)01050-6. PMID: 11812400; PMCID: PMC7172370.
- Holty JE, Bravata DM, Liu H, Olshen RA, McDonald KM, Owens DK. Systematic review: a century of inhalational anthrax cases from 1900 to 2005. Ann Intern Med. 2006 Feb 21;144(4):270-80. doi: 10.7326/0003-4819-144-4-200602210-00009. PMID: 16490913.
- Jernigan JA, Stephens DS, Ashford DA, Omenaca C, Topiel MS, Galbraith M, Tapper M, Fisk TL, Zaki S, Popovic T, Meyer RF, Quinn CP, Harper SA, Fridkin SK, Sejvar JJ, Shepard CW, McConnell M, Guarner J, Shieh WJ, Malecki JM, Gerberding JL, Hughes JM, Perkins BA; Anthrax Bioterrorism Investigation Team. Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States. Emerg Infect Dis. 2001 Nov-Dec;7(6):933-44. doi: 10.3201/eid0706.010604. PMID: 11747719; PMCID: PMC2631903.
- Kyriacou DN, Stein AC, Yarnold PR, Courtney DM, Nelson RR, Noskin GA, Handler JA, Frerichs RR. Clinical predictors of bioterrorism-related inhalational anthrax. Lancet. 2004 Jul 31-Aug 6;364(9432):449-52. doi: 10.1016/S0140-6736(04)16769-X. PMID: 15288744.
- Hendricks KA, Wright ME, Shadomy SV, Bradley JS, Morrow MG, Pavia AT, Rubinstein E, Holty JE, Messonnier NE, Smith TL, Pesik N, Treadwell TA, Bower WA; Workgroup on Anthrax Clinical Guidelines. Centers for disease control and prevention expert panel meetings on prevention and treatment of anthrax in adults. Emerg Infect Dis. 2014 Feb;20(2):e130687. doi: 10.3201/eid2002.130687. PMID: 24447897; PMCID: PMC3901462.
- Doğanay M, Aydin N. Antimicrobial susceptibility of Bacillus anthracis. Scand J Infect Dis. 1991;23(3):333-5. doi: 10.3109/00365549109024319. PMID: 1909051.
- Mohammed MJ, Marston CK, Popovic T, Weyant RS, Tenover FC. Antimicrobial susceptibility testing of Bacillus anthracis: comparison of results obtained by using the National Committee for Clinical Laboratory Standards broth microdilution reference and Etest agar gradient diffusion methods. J Clin Microbiol. 2002 Jun;40(6):1902-7. doi: 10.1128/JCM.40.6.1902-1907.2002. PMID: 12037041; PMCID: PMC130796.
- Cavallo JD, Ramisse F, Girardet M, Vaissaire J, Mock M, Hernandez E. Antibiotic susceptibilities of 96 isolates of Bacillus anthracis isolated in France between 1994 and 2000. Antimicrob Agents Chemother. 2002 Jul;46(7):2307-9. doi: 10.1128/AAC.46.7.2307-2309.2002. PMID: 12069996; PMCID: PMC127281.
- Manzulli V, Fasanella A, Parisi A, Serrecchia L, Donatiello A, Rondinone V, Caruso M, Zange S, Tscherne A, Decaro N, Pedarra C, Galante D. Evaluation of in vitro antimicrobial susceptibility of Bacillus anthracis strains isolated during anthrax outbreaks in Italy from 1984 to 2017. J Vet Sci. 2019 Jan 31;20(1):58-62. doi: 10.4142/jvs.2019.20.1.58. PMID: 30541185; PMCID: PMC6351761.
- Athamna A, Massalha M, Athamna M, Nura A, Medlej B, Ofek I, Bast D, Rubinstein E. In vitro susceptibility of Bacillus anthracis to various antibacterial agents and their time-kill activity. J Antimicrob Chemother. 2004 Feb;53(2):247-51. doi: 10.1093/jac/dkh016. Epub 2003 Dec 19. PMID: 14688054.
- Huang E, Pillai SK, Bower WA, Hendricks KA, Guarnizo JT, Hoyle JD, Gorman SE, Boyer AE, Quinn CP, Meaney-Delman D. Antitoxin Treatment of Inhalation Anthrax: A Systematic Review. Health Secur. 2015 Nov-Dec;13(6):365-77. doi: 10.1089/hs.2015.0032. PMID: 26690378; PMCID: PMC4710135.
- Migone TS, Subramanian GM, Zhong J, Healey LM, Corey A, Devalaraja M, Lo L, Ullrich S, Zimmerman J, Chen A, Lewis M, Meister G, Gillum K, Sanford D, Mott J, Bolmer SD. Raxibacumab for the treatment of inhalational anthrax. N Engl J Med. 2009 Jul 9;361(2):135-44. doi: 10.1056/NEJMoa0810603. PMID: 19587338.
- Barochia AV, Cui X, Sun J, Li Y, Solomon SB, Migone TS, Subramanian GM, Bolmer SD, Eichacker PQ. Protective antigen antibody augments hemodynamic support in anthrax lethal toxin shock in canines. J Infect Dis. 2012 Mar 1;205(5):818-29. doi: 10.1093/infdis/jir834. Epub 2012 Jan 5. PMID: 22223857; PMCID: PMC3274375.
- Pillai SK, Huang E, Guarnizo JT, Hoyle JD, Katharios-Lanwermeyer S, Turski TK, Bower WA, Hendricks KA, Meaney-Delman D. Antimicrobial Treatment for Systemic Anthrax: Analysis of Cases from 1945 to 2014 Identified Through a Systematic Literature Review. Health Secur. 2015 Nov-Dec;13(6):355-64. doi: 10.1089/hs.2015.0033. Epub 2015 Dec 1. PMID: 26623698; PMCID: PMC4710510.
- Kammanadiminti S, Patnaikuni RK, Comer J, Meister G, Sinclair C, Kodihalli S. Combination therapy with antibiotics and anthrax immune globulin intravenous (AIGIV) is potentially more effective than antibiotics alone in rabbit model of inhalational anthrax. PLoS One. 2014 Sep 16;9(9):e106393. doi: 10.1371/journal.pone.0106393. PMID: 25226075; PMCID: PMC4165586.
- Greig SL. Obiltoxaximab: First Global Approval. Drugs. 2016 May;76(7):823-30. doi: 10.1007/s40265-016-0577-0. PMID: 27085536
