COVID-19 Guide
This is not an all encompassing guide to COVID. The purpose of this is to give a launching pad on the critical things that an ID physician needs to know whenever you are guiding colleagues on the inpatient setting. As such, I will not be going over thing such as disaster preparedness, critical care issues (with some caveats), and PPE. Updated as of 12/29/2020.
As the state of COVID (at least in terms of therapeutics, diagnosis, and presentation) has not changed much, this page will not be updated for the foreseeable future. I realize it has been over half a year since this post is updated, but I think the basics of the post are solid albeit it is a little verbose. I will work on it whenever I can to update it, and will include topics on the origin of SARS-CoV2 (as this is a hot topic), update the section on vaccinations, clotting, streamline some of the therapeutics to highlight the most important points as well as some of the transmission/mask and social distancing stuff. As we head into a state where most of the US population is (hopefully) vaccinated, I think this is a relic of a time of uncertainty and most of the up to date stuff you may find in the article summaries, as I tend to talk about a COVID-related topic there.
BASIC MICROBIOLOGY

While it is believed the virus originated from a bat host (like the other coronaviruses), it seems the intermediate animal was a pangolin.
SARS-CoV and SARS-CoV2 bind to ACE-2 receptors. These are highly expressed throughout the body, but seem to be also expressed in type I and type II pneumocytes, endothelial cells, as well as the nasopharynx (2). This may explain in part the pathophysiology behind COVID-19, including hypoxemia (by taking out type II pneumocytes, this leads to less pulmonary surfactant and thus, atelectasis), and hypercoagulability (see later).
Pathophysiology:
As highlighted above, SARS-CoV2 binds to ACE-2 receptors in the nasopharynx and type 2 pneumocytes.
Infection of type II pneumocytes -> lack of surfactant -> atelectasis
But keep in mind that it also infects type I pneumocytes-> sloughing off of type I pneumoncytes and ultimately organizing pneumonia seen on biopsies (3). In this research letter, 6 French patient’s who died from COVID had autopsied done. The interesting thing here is the difference in pathology, with one who died within 5 days and had lymphocytic viral pneumonia, while the others had what looked like acute fibrinous organizing pneumonia. This may have implications in terms of ventilation/oxygenation for critical care folks. A recent series (218) found that patients with COVID-19 tended to have severe endothelial injury, with histology finding widespread thrombosis with microangiopathy. When compared to influenza patients, these changes were 9 times as prevalent in the COVID-19 population.This is a nice overview of the pathophysiology that is going on in COVID and how to target specific therapies (36).
In terms of oxygenation, the pathophysiology may stem from disruption of lung architecture, since type II pneumocytes, which make surfactant are involved.
In this way, you can see how the “crash and burn” happens. It is a horrible cycle; atelectasis from COVID-19 leads to further atelectasis from lack of normal lung architecture which, in turn, leads to more atelectasis and worsening hypoxemia.
How viral load plays a role here is unclear, but seems the highest viral load tends to be at the beginning of the symptomatic period with a slow decline in viral load over anywhere from 2-8 weeks (198):
Transmission:
Both airborne and mucocutaneous transmission play a role, however airborne seems to be the major driver of outbreaks. Despite this, virus has been isolated from various surfaces (5):
.
The virus titer not only is dependent on the surface, but also the presence of proteins associated with the virus. In one experiment (183), virus was diluted in bovine serum albumin to simulate the environment it would have in droplets. Overall, the presence of protein did not result in a significant drop in viral titers across 3 surfaces (glass, polystyrene plastic, and aluminum):
Regardless, the main route of transmission has been argued to be airborne. Viruses are released during exhalation, talking, and coughing in microdroplets and at typical indoor velocities, a 5 um droplet will travel tens of meter (184). Further, RNA has been found in droplets smaller than this. Authors have argued that airborne transmission to be the only plausible explanation for superspreading events in crowded and inadequate ventilated environments. As such, the recommendation has been to:
- Provide sufficient and effective ventilation
- Supplement ventilation with airborne infection control measures such as local exhaust, high efficiency air filtration, and germicidal UV light
- Avoid overcrowding
Some examples of transmission:
- Person to person transmission (7,8) including transmission of the virus during the incubation period (7), as well as fairly high attack rates varying from 20%-80% in those who had close contact with index cases (9, 10) in settings such as choir practices and church gatherings. These may have been airborne transmission events
- Reports have also suggested asymptomatic transmission, including a traveler to China, who infected his neighbor and said neighbor was responsible for a cluster of >71 cases (162). Another study from Vietnam identified 2 clusters of patients who were in contact with asymptomatic patients, despite the fact these asymptomatic carriers had lower viral loads per PCR (163).
- Secondary attack rate during the pre symptomatic period ranges from 3-16%, with higher rates the older you are (8%), if the index case is symptomatic (~6%), and if you live with the index case (16%, 11). Although workplace attack rate in this study was 0 (woot woot). As below, further data (164) suggest that close contacts (i.e. family members living in the same household) tend to have a fairly high secondary attack rate, reaching up to nearly 17% (in the figure below, residential address are those people that live in the same household but are not related i.e roommates. Why not clump them together…idk).
- SARS-CoV2 have been isolated from surfaces. For instance, one publication (12) noted isolation of virus in a hotel when swabbing areas such as sheets and duvet covers. How clinically significant is this is unclear.
- .A publication from early on during the pandemic identified an index case who had returned from Wuhan and had dinner with his family at the third floor of a restaurant next to 2 other families (166). As shown below, this lead to an outbreak of 9 total cases:
Other potential areas of virus shedding:
- GI tract: including biopsy specimens throughout via C-scope where virus was isolated (13)
- Stool and rectum (14, 16)
- Blood, plasma (14,15,16)
- Semen (17)
- Nothing has been isolated from urine
- What does this mean? Most likely nothing. Given the superspreading events highlighted above, doubt that shedding virus from the stool is going to be a main driver of infection
Presymptomatic transmission – as noted above, this seems to be a huge problem. In one of the SnFs in Washington where this pandemic started in the US, there was a high proportion of pre-symptomatic and asymptomatic spread (18)
R ⌀ is the average number of people that an infected person transmits the virus to. Essentially, it is an estimate of how infectious an agent is:
- If <1: epidemic will burnout
- If =1: epidemic will continue at a steady pace
- If >1: it will increase
Right now, this number is ~2.5-3, however this can changed based on the location (i.e. cruise ships, it can get as high as ~13, see 19)
Superspreaders – the concept that the disease is spread by a handful of individuals for several reasons (20, 21). See below:
Viral Shedding: really depends on disease severity. In reality, we do not know if the detection of virus in PCR represents infectious particles or rather just dead virus (22). Korea’s CDC did not find evidence of spread from those who “re-tested” positive.
- Though there is some evidence there is shedding via PCR anywhere from 16-22 days (23), and up to 41 days in some cases (24)
- o In a recent study of symptomatic and asymptomatic COVID cases (182), it was found that even asymptomatic patients could continue to test positive up to 44 days:
- While asymptomatic patients tended to convert to a negative PCR faster than symptomatic patients, a significant portion were testing positive at days 7-14 (199). The difference did not reach statistical significance, however.
- How clinically significant this is, is unknown.
Mask and Social Distancing:
Most of what is known about mask usage comes from prior viral pandemics and other bat coronaviruses. Having said that, there is fairly good evidence that masks do decrease the amount of droplets that go into the air from one person. For instance (156), in a study comparing the efficacy of a surgical mask vs 2 other types of mask for prevention of viral diseases (won’t really go over the details here since these types of masks are not freely available), the found that in a simulation of viral load (using fluorescent KCl) that surgical masks only allowed roughly 3-4.5% of aerosolized contents (compared to almost none in the test masks).
Of course this is only for one individual wearing a surgical mask, as opposed for the individual spreading the “droplets” wearing one as well.
For instance, Singapore was one of the hardest hit areas during the original SARS outbreak (157) and thus, implemented aggressive methods to avoid propagation of the virus. During the first 100 days after the initial warning of the virus, roughly 95% of people were adherent to mask wearing policies. During this time, they recorded 961 cases of COVID-19, able to track them down to 11 clusters of 113 people engaged in mask off activities and only 3 clusters involving 11 people with mask- on settings. They further compared their number of cases when compared to other countries:
This suggest the impact of wide mask wearing policies, especially compared to the rest of the world
Another meta-analysis (158) evaluated mask wearing in the healthcare setting, non-healthcare setting, and the impact of social distancing. Most of the data presented involved the bat coronaviruses (SARS, MERS, SARS-CoV2), with most of the data obtained pertaining to the original SARS outbreak. In this meta-analysis, they included 44 observational studies (no RCTS or prospective data). Pooled analysis of these studies suggested the benefit of distance and risk of infection:
The adjusted OR overall was 0.18 (95% CI 0.09 to 0.38) AR 12.8% with shorter distance vs 2.6% with further distance, risk difference -10.2% 95% CI -11.5 to -7.5. This suggest there is benefit to social distancing in terms of infection rates.
The degree of distancing played a bigger role in those who where at a higher risk of infection:
In terms of mask, they evaluated the use of N95 respirators and face masks. They found a significant reduction in the risk of infection (aOR 0.15, 95% CI 0.07 to 0.34) with an absolute risk of 3.1% with face mask vs 17.4% without face mask, risk difference -14.3% (95% CI -15.9 to 10.7). This association was stronger in healthcare settings (RR 0.3, 95% CI 0.22 to 0.41) vs non-healthcare settings (RR 0.56, 95% CI 0.4 to 0.79, p-value 0.049).
The above pulls together N95 and face mask. Splitting them up:
Seems that the benefit tends to be driven by the use in health care settings. This holds true (even more so) with N95 when compared to face mask. For non-health care settings, the overall results do not seem to be impressive. Indeed the authors mention “Our unadjusted analyses might, at first impression, suggest use of face masks in the community setting to be less effective than in the health-care setting, but after accounting for differential N95 respirator use between health-care and non-health-care settings, we did not detect any striking differences in effectiveness of face masks between settings.” While the effect may be weak, one can see how translating this into the mass populations can have its benefits (due to the sheer size of..you know the entire global population).
Further US data comes from a data set (159) that evaluated states that underwent either employee-only mask mandates and community wide mask mandates. They estimate that, 21 days after the mandate went into effect, there was a 2% drop in the new daily case rate:
This did not hold true for states with employee-only mask wearing mandates:
They estimate that roughly 230k to 450k cases were avoided by the mask-mandates. Of course, it is difficult to say how many of those people in said states wore the mask and if there were other mandates, such as stay at home orders, that may have accounted for such a drop.
Do non-surgical masks work? Yes, but not all of them are created equal.
One study (126) took 211 volunteers (208 with flu and 6 with suspected COVID) and they evaluated the efficacy in preventing particles from both a surgical mask and a triple layer cotton mask. They measured the background number of particles for all participants in a bedroom and in a car and both the surgical mask and the three layer cotton mask had fairly comparable results in the amount of measured particles, suggesting that both coverings were protective.
Another study (185) evaluated a method to measure droplets while wearing different mask or mask alternatives. Over a 40s period (10s of baseline, 10s of speaking, and an additional 20s of baseline), absolute droplet count was 960 (control). Droplet transmission ranged from 0.1% for the N95 to up to 110% for the fleece mask, which was likely due to dispersing of the largest droplets into multiple smaller ones:
An earlier RCT in healthcare workers in high risk wards (186) evaluated surgical masks with cloth masks (two-layer made of cotton) during a 4 week period. Cloth masks actually resulted in a higher attack rate in all primary outcomes of the trial:
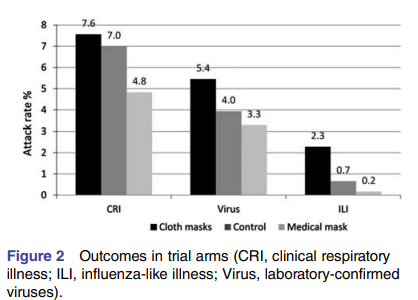
Overall, cloth masks had a RR of 13.25 (95% CI 1.74-100.97) for influenza like illness in comparison to medical masks. This suggest that while masks are incredibly helpful for disease prevention, not all masks are made equal. Having said that, in low risk areas certain cotton masks but surgical masks may be the best bet.
Of course, what about the recent study published in the Annals of Internal Medicine, the DANMASK study (219)? This was a Danish study that evaluated the recommendation to wear a surgical mask outside of home. This was a nationwide, unblinded, RCT where those who were spending more than 3 hours per day outside of the home were encouraged to wear a mask. Primary endpoint was SARS-CoV2 infection in the mask wearer at one month. Over 6000 patients were randomized, however only 46% of participants in the mask group wore the mask as recommended. 1.8% of patients in the mask group and 2.1% in the no mask group were infected at the end of the month, for an absolute difference of -0.3% (95% CI -1.2 to 0.4%, p-value 0.38; OR 0.82, 95% CI 0.51 to 1.23, p-value 0.33) in the ITT group. So it appears that mask wearing did not help with infection prevention. There are a few issues here to mention:
- It is unclear if other prevention methods were followed (social distancing, handwashing, no gathering of large crowds)
- The adherence to mask testing was fairly low
- The incidence of SARS-CoV2 in this population was 2%, so if masks did have an effect, it would be unlikely for us to see it here. It is unclear the relative risk reduction of masks in SARS-CoV2 infection but low-balling it at 1% RRR with a 2% incidence, lets just say you’re going to need way more than 6000 people to detect a difference.
- COVID tests were self-administered; while this is usually a good method, the sensitivity may be lower due to lack of proficiency in performing the test (i.e. you won’t be able to get the tip into the nasopharynx)
- At the end of the day, the clinical question asked was if the recommendation decreased incidence of SARS-CoV2 infection.
At the end of the day, I do not think the trial answers the question of masks reducing transmission of SARS-CoV2 infection but rather ended up studying the compliance rates of the recommendation. All that to say, just wear a mask.
In terms of stay-at-home orders, a recent retrospective study (167) looked at the effectiveness of such orders in 42 states and DC. They took a look at the rate of infection pre-implementation and post-implementation of these orders and found the rate of increase of infection pre-order was 0.113 (or 11.3%, 95% CI 0.11-0.115) per day and post-order was 0.047 (95% CI 0.045-0.048) per day. A pooled-meta analysis of the data by number of cases suggested a mean difference of 6.847 (95% CI 6.831 to 6.863 p-value 0.0001):
In other words, stay-at home orders have reduced the infection rate from 12% per day to less than 5% per day. This essentially increases the doubling rate of infected patients from ~5-6 to about 2 weeks.
CLINICAL CHARACTERISTICS:
Symptoms:
I could say this looks like either acute influenza, or prolonged ARDS but I know you want more. In general:
- Most tend to be middle age (~55 years old), and around 50% have an underlying condition, mostly hypertension (40%) and some have diabetes (4)
- Incubation: 5 to 7 days, up to 14 days in some cases (35)
- What to look for:
- Fever: usually present in anywhere from 52% of patients up to 100% depending on the case series you look at (23, 25, 26, 27).
- Cough: 48%-85%, usually dry
- Dyspnea: 37%-76% (this is important, see the section on “happy hypoxemia).
- Abdominal symptoms: typically diarrhea in ~20% and nausea (13)
- A proportion of patients may report symptoms up 60 days after diagnosis and 36 days after discharge from the hospital (187). In one retrospective study, only 12.6% of patients from Italy surveyed reported no symptoms, 53.1% reported fatigue, 43.4% reporting dyspnea, and 21.7% reporting chest pain.
- Loss of smell and taste has been reported widely. In one prospective multicenter cohort (188), loss of smell was the initial sign of infection in 29.1% of patients, and occurred shortly after other symptoms in 70.9%. This occurred suddenly and within a median of 3 days of infection. Roughly 20% lacked other symptoms of COVID-19. In this cohort, 72.9% of patients had some sort of recovery at 15 days with 27.1% of these having complete recovery.
- Unfortunately, using these signs as a diagnostic modality is not superior to RT-PCR:
- Labs: aside from the standard CMP, CBC w diff, you should get the following:
- LDH – this is one of the disease groups that tends to raise your LDH with pulmonary infiltrates. Increased severity increases the LDH (26, 28) and is associated with mortality (29)
- Procalcitonin, but may not be terribly helpful
- D-dimer: see later, however higher levels seem to correlate with disease severity and mortality. Whether this can be used to guide intensity of anticoagulation remains to be seen (26-29)
- CRP: this seems to correlate with disease severity. Those who tended to have worse outcomes or required a higher level of oxygen had higher CRP (26,28). Higher CRPs are also associated with bad outcomes (29).
- Ferritin: as above, higher levels = more badness
- Lymphocytes – usually about <1500, but ranges from 800-1700 and higher. Not very specific or sensitive, but in the right clinical setting, it can be helpful. If you want more details, look here (30), the crux here is that, in general, there tends to be lower lymphs (esp both CD4 and CD8) in COVID, especially in severe COVID and the lymphocyte level may track with improvement.
Imaging: TL;DR -> most patients tend to have some sort of findings in CT, (even when pre-symptomatic). You won’t see just one consolidation, but rather peripheral, patchy infiltrates and perhaps diffuse consolidation.
- CXR: in general, you will see something on the CXR, depending on severity. Literature suggests that anywhere from 25% of cases in mild disease up to 100% of cases in severe ICU cases will have some sort of infiltrate, usually bilateral (23, 26, 28). One study found that roughly 54% of patients with non-severe disease had abnormal CXR, compared to 76% of those with severe disease, ranging from GGO, patches, bilateral interstitial abnormalities (31).
- Pleural effusions do not seem to be common, though one case series in Seattle, WA this find this in ~28% of its patients (27).
- CT scanning:
- Ground glass opacities or consolidation is typically seen in most, usually involving more than 2 lobes in about 70% of the time without nodules or lymphadenopathy (32).
- In general, lesions tend to be bilateral as well and by far, these tend to be peripheral. In this one study, there was a few pleural effusions and some lymphadenopathy, but this seems to be rare (33).
- In general, the pattern seems to be as follows: early onset you have ground glass opacities (usually in the first 5 days) which progress to more consolidative at around the 2 week mark (34).
To summarize: someone with fevers, dry cough, and clinical history suggestive of possible viral pneumonia with elevated LDH, high CRP, high D-dimer and imaging on CXR with peripheral ground glass opacities and CT similar as above -> Think COVID.
Pneumothorax: one of the interesting things I have witnessed with several COVID patients is the high rate of pneumothorax. Whether this is my own personal bias or if there is some increased risk due to COVID in relation to other diseases, I do not know (seriously, not even with severe pneumocystis pneumonia did I see so many).
Unfortunately, most of the reports on the topic are limited to case series or case reports. One retrospective review of 3000 patients admitted with COVID identified only 6 patients with pneumothorax, yielding an incidence of 0.66 (244). Another described 3 cases of pneumothorax in non-intubated patients, with no risk factors for pneumothorax (245). One of these actually had bilateral pneumothoraces that occurred sequentially. The largest case series evaluated 71 patients who presented with either pneumothoraces or pneumomediastinum (246). Notably, 9 of these (15%) actually presented to the ED with pneumothoraces and 20 (32%) were not on NIPPV or on mechanical ventilation when diagnosed. This suggests that a mechanism other than barotrauma may be at work (indeed, I have seen several cases of pneumothorax in someone a few weeks into their illness not on CPAP).
The mechanism behind pneumothoraces in context of COVID remains to be elucidated, however it is believed to be related to rupture of alveoli and formation of cystic lesions, along with ischemic parenchymal damage (247). Despite my own personal bias, it seems the incidence is low overall at 1% for all patients, and 2% for those who are in the ICU (246). The take-home message is that COVID patients who do not have risk factors for pneumothorax and who are not on any positive pressure ventilation can have a pneumothorax. Indeed, if you see someone get spontaneously hypoxic, think of a pulmonary embolism (see later) or a pneumothorax.
Diagnostics:
- COVID Specific PCR:
A few things to keep in mind:
- As of now, we have no gold-standard to compare PCRs to. That means, that the test characteristics will chance as we learn more
- The sensitivity of each test will depend on the quality of the specimen. Just like a putting a needle to collect blood cultures will lead to contaminated specimens, so will an inappropriately specimen will lead to a high false negative rate
- Further, the timing of when you get the sample (i.e getting it at day 1 vs day 7 of illness), where it comes from (mouth, BAL) and the severity of disease also plays a role in the sensitivity.
- By in large, all of these assays are super specific, meaning you won’t find MERS when you want to find SARS-CoV2
- There are a few things that you will be asked, especially as it relates to the interpretation of testing. A few concepts to keep in mind
How does it work?
Remember that cycle threshold depends on the fluorescence. They’re not very good at giving you an absolute number i.e. if the number of cycles crosses a threshold = positive fluorescence and therefore = positive test. This is not terribly important clinically, just as an aside since you may encounter this in the literature
- Limit of detection (LOD): the minimum amount of RNA needed for the test to flag “positive” in 95/100 samples. The higher this is, the higher the rate of false positives. Conversely, the lower it is, the higher the rate of false negatives.
- Assays available:
RT-PCR Sensitivity:
- -This depends on the location of the swab (nasopharyngeal, oropharyngeal, BAL), and how may days since symptom onset. These assays actually have high analytical sensitivities and specificities under ideal circumstances, but real world is not ideal. At the end of the day, method of acquisition (i.e. if the person doing the swab doesn’t know what they’re doing), days from infection (higher viral load = higher sensitivity) play a crucial role here.
- In general, when compared to CT scanning (showing peripheral opacities), the sensitivity of throat swab was around 65-71% (38, 39). One re-analysis of this data suggested the actual sensitivity of about 77% (40).
- Specificity tends to be fairly high, around 97%.
- A recent analysis of RT-PCR in Chicago found the sensitivity ranged from 82%-97% (189). This tested various tests (Cepheid Xpert and Xpress, Roche cobas, Abbott ID now, BD Max System, BioFire, Labcorp).
- The 2 ends were calculated differently, however. One calculation assumed all positives were true positives while if a negative was followed by a positive, this was a false negative. However, only a small percentage had repeat testing so those who only tested negative once were true negatives. The second calculation included patients that tested twice.
- It is safe to assume that in real life and outside of hospitals or situations where you can get back to back RT-PCR testing is roughly 80% sensitive.
- When comparing throat swabs and nasal swabs, nasal swabs tend to do much better (41), though if possible, BAL tends to have the best sensitivity of anywhere from 93-100% (41, 42).
- Though the sensitivity depends on how long into the illness you get the sample from, with throat samples going down in sensitivity after one week, and dropping to a sensitivity of 36% after 2 weeks for throat swabs and 50% in nasal swabs (41).
- A recent study showed (161) that while there was significant concordance between OP and NP swabs, the NP swabs tended to have a lower Ct, which meant higher viral load.
- The figure below (43) highlights the differences in throat and nose swabs in an ideal scenario where you get an adequate sample (i.e someone did their job correctly when collecting the swab). Shows 2 things, A) that these tests can be highly sensitive in the right setting (aka not real life) and B) the fact that probability of detecting SARS-CoV2 decreases the further down the disease you are at.
- This next figure (44) is important for screening healthcare workers or high risk exposures. Bottom line, the probability of detecting the virus a few hours after exposure is really low. The sweet spot as to when to screen someone would be roughly 5-7d after exposure.
Saliva vs nasopharyngeal sampling: there seems to be a movement towards using saliva as a screening measure,. For instance, one review highlighted that deep throat salivary samples tend to have higher sensitivity (87-92%) when compared to oral cavity samples (50%) and those expressed from the salivary glands (12%; 45)
Another study suggests there is a higher titer of viruses detected in the salivary samples of a small series of inpatients (46). Some high risk healthcare workers tested positive via salivary samples but not nasopharyngeal samples. The numbers are small, but this could prove another testing modality. A retrospective study of 91 patients (211) with paired NP and OP samples found the sensitivity of NP PCR to be 89% and that of OP samples to be 72%, with the highest sensitivity being between days 8-14 from illness onset. A larger study evaluated 229 paired OP and NP samples (212) and found that both options were viable with high sensitivity as long as the OP sample was taken within 7 days of symptom onset, achieving a positive percent agreement between the 2 samples of 96.6% (95% CI 87.3-99.4). The degree to which there is viable virus may play a role in terms of transmission:
Can patients self-collect a sample? Absolutely. An FDA report (214) found that the number of samples that resulted indeterminate or failures were identical between self-collected and healthcare collected:
2. Serology
As it stands, this is not going to be a major up front diagnostic tool. The utility is going to be more in terms of epidemiological purposes and for convalescent plasma. Let’s review the actual structure, as there are 3 antigens that are potential targets (47).
- N protein, conserved in all other coronaviruses
- S1 protein, also seen in SARS-CoV2
- Receptor binding domain (RBD) of SARS-CoV2 S1 protein, which is more specific.
There are obvious issues with the use of the first 2 antigens in terms of false positives, especially as SARS-CoV2 enters the pool of respiratory viruses. Some assays actually use both N protein and S1 protein, so this may not be much of an issue (49).
There are3 general assays that are used:
- The ELISA – standard, uses color change to detect the antibody
- Chemiluminescence immunoassay – similar to ELISA but uses light
- Lateral flow assay – works like an OTC pregnancy test, see below
I say all this because this plays a role in how to interpret the test one sends and how you see the reports of sensitivity and specificity. Not all assays use the same targets and not all assays have the same performance characteristics. Further, lateral flow assay is one modality that may be easier to employ en masse, and may be more commonly used as a POC rapid test.
In general, it seems that ELISA tends to perform the best out of all. Sensitivity is around 73-74% in confirmed COVID infection by PCR up to 87% if adding suspected COVID patients that tested negative by PCR (48, 50). IgG tends to perform similarly, has a sensitivity of ~71-87% (48,50). Combined antibody testing tends to be best, having a higher sensitivity by ELISA up to 78% (50). CMIA tends to do a bit worse, sensitivities ranging from 48% up to 69% for IgM and 70% for IgG (49, 50).
The sensitivity varies as to when you get it i.e getting it early will lead to a false negative. For instance, ELISA IgM sensitivity tends to be ~33% if obtained within 7 days, and increases up to 96% about 3 weeks out (50). Some people will seroconvert within 5 days, but usually this doesn’t happen for most until roughly 13-15 days and up to 19 days for IgM and up to 20 days in IgG (50). Usually, more people seroconvert their IgG than they do IgM by day 20 (51, 52, 53).
Example of the pattern of Ab and PCR dynamics. Note that antibodies tend to rise after 7 days. A study evaluating 4 commercial serological assays and one local ELISA (232) found fairly high sensitivities. Two assays evaluated IgG while 2 others evaluated total antibodies. In general, 2 assays targeted the nucleocapsid protein and 2 target spike protein. 1000 pre-pandemic samples and 769 COVID samples were analyzed. The overall sensitivity and specificity for all assay taken >20 days after symptom onset was higher than 92%:
Adjusting the threshold could achieve a sensitivity/specificity of 98% at around 30 days after symptoms. PPV and NPV were also evaluated for each assay, with the Abbott assay having the highest rate of false negatives:
It also seems that the titer can be a marker of disease severity, with more severe cases having higher titers (54):
How long do antibodies last is a question that remains up in the air. A recent serological study (182) evaluated 37 symptomatic and 37 asymptomatic patients and tracked the level of IgM and IgG at 2 points in time, in the acute phase and in the convalescent phase 8 weeks after discharge from the hospital:
They found that 81.1% of asymptomatic patients tested positive for IgG compared to 83% of those who were symptomatic in the acute phase. Not surprisingly, those patients who were symptomatic had higher levels of IgG. When tested early in convalescence, however, the found that IgG levels decreased in 93.3% of asymptomatic patients and in 96.8% of the symptomatic patients. The median percentage decrease was 71.1% (range 32.8-88.8%) in the asymptomatic group vs 76.2% (range 10.9-96.2%) in the symptomatic group. Further, the levels of neutralizing antibodies were also decreased in both groups, but the change was not as dramatic (8.3% in the asymptomatic group vs 11.7% in the symptomatic group).
In a larger study from Iceland (213), 1107 of 1215 patients who were tested were found to be seropositive, with titers increasing over a period of 2 months and remaining consistent for the remaining 2 months of the study:
What this means at this juncture is unclear, but hopefully more data comes around suggesting that antibody titers remain elevated.
3. Antigen Testing
These are usually rapid, POC testing that uses a lateral flow testing format (see above; remember this is similar to an OTC pregnancy test, making it a simple POC test). A review by WHO shows that the sensitivity of this modality compared to NAAT tends to range from 0 to 94% (215) with specificity remaining >97%. The Forrest Plot in a Cochrane review reflects this wide margin when testing either NP or OP samples (216):
Given this wide variability in sensitivity, recommendations for this modality are as follows:
- Use in symptomatic patients, as the higher viral load increases the sensitivity
- Do not use if there is low prevalence of COVID-19
- Follow up with NAAT if feasible. While low false positivity, as noted above, low prevalence will result in a poor PPV.
- Would follow up with NAAT even if the Ag test is negative, if there is a high index of suspicion.
Here is a proposed flow chart for the use of Antigen testing in settings of widespread community transmission where there is no NAAT capacity (215):
Approach to Initial Labs:
In general, I advocate the following upfront:
- BMP + LFTS
- CBC w diff
- Inflammatory markers: CRP, ferritin
- LDH
- D-dimer, fibrinogen, PT/PTT, INR
- Blood cultures and respiratory viral panel if warranted
As far as what to do once they are admitted, I would trend LDH, Ferritin, CRP, and D-dimer for prognostication purposes. You could argue you do not need these, but if you need to make clinical decisions (or discuss goals of care), using these against data can give you a sense of prognostication when discussing with family members
For daily rounding:
- Some people advocate not using stethoscope. In the beginning, I was on this camp, but given disposable stethoscopes, these are fine.
- Daily CXR are not needed
- Keep a close eye on the oxygenation; for some reason (see later), these patients tend to deteriorate quickly and the major tip off is their oxygenation. They will look great and have no tachypnea but they will sat in the 70s.
- DVT PPx is a must for everyone (see later)
- Use Acetaminophen for fevers. There is no clear data for NSAID use in COVID-19 (55), but there is some concern about possible upregulation of ACE-2 receptors. Until there is more data, I would advocate Acetaminophen here.
- Progression of oxygen requirements: low flow oxygen -> Non-Rebreather (call ICU) -> High flow nasal cannula -> CPAP
- If you are at the point of Non-rebreather -> awake proning (see later)
Triage: this tends to be fairly simple. Aside from the obvious (hypotension, severe hypoxemia) I would focus on a few things.
- Degree of hypoxemia – even if they look great (aka have their phone out), if they are requiring a non rebreather to maintain their saturations, think about ICU
- Resp rate >30. That is bad
- Altered mental status
- Multilobar involvement
I would call the ICU about these patients if you encounter them.
THERAPIES:
This is a very fast moving subject. As we have seen, the hype for hydroxychloroquine has died down, ivermectin and colchicine never really got off the ground, and there may be some doubts about remdesivir. Prior to this, lets move to another hot topic that has been in the news lately.
- Vaccines:
This has been getting a lot of press lately, with the approval of both the Pfiezer vaccine as the Moderna vaccine in the US. As of now, the COVE trial for the Moderna vaccine has not been released yet, so I won’t really discuss this one. This is a randomized, placebo controlled, phase 3 trial that enrolled over 30 thousand patients and demonstrated 94.5% efficacy in preventing COVID.
Both of these are mRNA vaccines. The BNT162b2, the Pfizer vaccine, is a lipid nanoparticle-forumlated, modified RNA that encodes the full-length spike protein (the thing that the virus uses to bind to ACE-2 receptors). This is modified to lock the protein in the pre-fusion conformation. This essentially allows our body to encode this mRNA into the spike protein (without any virus around) and the immune system gets to work in producing antibodies. The safety and efficacy of this vaccine was evaluated in a large trial (220), where patients were randomized in a 1:1 ratio to receive a 30ug of the vaccine or placebo. The 2 doses were separated by 21 days. Primary end points for safety were systemic or local adverse events, while for efficacy it was the number of confirmed COVID cases at least 7 days after the second dose in those who had no evidence of prior infection. 43448 patients were randomized, with 18566 receiving at least 2 doses of the vaccine. The most common adverse event was pain at the site, 83% after the first doe, and 78% after the second. Systemic symptoms were also more common in the vaccine group, with fatigue and headache being the most common (58 vs 23% and 52 vs 24% respectively). No deaths were attributed to the vaccines. Efficacy in the 36,523 without prior evidence of COVID demonstrated a 95% vaccine efficacy (95% CI 90.3 to 97.6):
There was no difference in efficacy in the subgroup analysis, which included groups stratified by age, sex, race, and country did not show any differences either.
There are also adenovirus-vector vaccines, however these have not really gotten off the ground as of yet. One small trial sponsored by Cansino found it induced immunogenicity, though an accompanying editorial did raise some concerns about using adenovirus as the vector given prior experiences with a similar vaccine for HIV (221, 222).
2. Remdesivir – Think of it as the pip-tazo of the antivirals. It is a nucleotide prodrug that has broad spectrum antiviral activity against Filoviridae (Ebola and Marburg), SARS, MERS, RSV, Nipah, and Hendra viruses (73). It has fantastic activity for MERS when compared to Kaletra and ribavirin (73) and it has been the subject of intense interest every since it failed to make any meaningful impact on Ebola. By far, the data is not terribly impressive. To summarize, it may have an impact in shortening the duration of illness in mild to moderate disease, but it may not have any meaningful effect in severe disease. There have been several studies, including a compassionate use case series that was, for some reason, published in NEJM. There are several issues with this paper, so I will not discuss it here.
The first major RCT was the ACTT-1 trial (74). To summarize, this was a multi-center, placebo-controlled trial comparing remdesivir vs placebo with primary outcome being time to recovery at day 28 based on an 8-category ordinal scale. 1062 patients were randomized, with those in the remdesivir group having a shorter time to recovery (median 10 days vs 15 days, RR 1.29, 95% CI 1.12-1.49). This was primarily driven by patients who were on oxygen, while the difference in those who were on HFNC/NIPPV, on vent/ECMO, or no oxygen having no statistically significant difference.
Another multicenter, open-label RCT (205) evaluated a 10d course of Remdesivir vs 5d course vs standard of care in patients with moderate COVID-19. Primary endpoint was change in clinical status on a 7-point ordinal scale by day 11. The 5-day remdesivir group had higher odds of better clinical status when compared to standard care (OR 1.65, 95% CI 1.09-2.48). There was no difference in clinical improvement between the standard group and 10d Remdesivir group.
Most recently, the SOLIDARITY trial (223) was released. This was a multi-center, open-label, pragmatic trial that compared a 10d course of remdesivir vs standard of care. Primary endpoint was in-hospital mortality at day 28. 5475 were randomized in the remdesivir arm of the trial (note this trial was huge, and it had an HCQ arm, kaletra arm, and IFN arm). There was no mortality benefit between the groups (not surprising), though one thing hidden in the supplemental figures is the increased length of stay, which was seen across all arms of the trial:
This may be due to patients staying in-house to finish their therapy in comparison to those in the ACCT-1 trial, where they merely looked at the improvement, not necessarily length of stay. Further, the paper also included a meta-analysis pooling together both trials:
So it doesn’t seem remdesivir improves mortality, though in an ideal world, you can get patients recovered quickly. This effect holds, even 10 days into the symptomatic stage, which is surprising for an antiviral. That being said, if cost is not an issue at your facility and this is available, I think a reasonable approach is to give this to anyone who requires a facemask or HFNC and opt for the 5 day course. If the patient recovers to the point they can be discharged and they have not finished their remdesivir, I see no point in keeping them for the 5 day course.
An attempt to improve upon Remdesivir has been to add another drug to create a cocktail. A continuation of the ACTT-1 trial (231) evaluated the combination of remdesivir and baricitinib. The latter is a JAK 1 and 2 inhibitor that inhibits intracellular signaling of cytokines (including IL-2, 6, 10, IFN-gamma). This was a multi-center, double-blind, placebo controlled trial. Patients were randomized in a 1:1 fashion to either remdesivir + baricitinib or remdesivir + placebo. Primary outcome was time to recovery at day 28 on an eight-point ordinal scale. 1033 patients were randomized with around 10% of patients in both groups were on invasive mechanical ventilation or ECMO, while over 50% in each group were requiring some sort of supplemental oxygen. Overall patients in the combination group recovered one day faster than patients in the placebo group (median 7 days vs 8 days, RR 1.16, 95% CI 1.01 to 1.32).
The highest rate of recovery was seen in patients who were on non-invasive ventilation or high-flow oxygen, with a median time to recovery in the combination group of 10 days vs 18 days (RR 1.51, 95% CI 1.10 to 2.08).
There was no statistical difference in mortality, though notably the highest difference in survival was seen in those with a baseline score of 5 or higher. There was no difference in outcomes when stratified by duration of symptoms:
For now, until more data comes out, I would stick to remdesivir alone or with steroids.
TL;DR – if you have it available, go for it. Would use it for 5 days, but if a patient is feeling better to the point of discharge prior to the 5 days, I would stop it and discharge the patient.
3. Steroids – This has been a game changer since the release of the RECOVERY trial. While there was some concern early on in the pandemic about using steroids early in the game due to the risk of prolonging viral shedding, it has been shown that using them in a careful fashion it can actually improve mortality in some of the more severe COVID cases. Early data can be found here: 75, 76, 77, 78, 79, 80, 81, 82. I’ll be focusing on the larger COVID-19 specific trials. The rationale is below:
The RECOVERY trial is a large UK trial that has enrolled and randomized patients to either standard of care, dexamethasone, HCQ, azithromycin, Kaletra, Toculizumab, or convalescent plasma. As far as the dexa arm is concerned (139), patients admitted with COVID-19 were randomized in a 2:1 fashion to either standard of care, or dexamethasone 6mg for 10 days or until discharge. Primary endpoint was 28 day all cause mortality. 6425 patients were randomized, with mortality at 28 days being lower overall in the dexamethasone group (22.9% vs 25.7%, RR 0.83, 95% CI 0.75 to 0.93, p <0.001). The greatest benefit stemmed from patients on mechanical ventilation (29.3% vs 41.4% in dexa and standard care group, respectively, RR 0.64, 95% CI 0.51 to 0.81) as well as those who got oxygen without mechanical ventilation (23.3% vs 26.2%, RR 0.82, 95% CI 0.72 to 0.94).
These are encouraging results. One of the ramifications of this study, however, was that many of the other trials evaluating the use of steroids in COVID patients were stopped once this trial was published. This was seen in the October 6, 2020 issue of JAMA, which published 3 different trials of steroids in COVID patients.
For instance, the CoDEX trial (224) was a multicenter, open label, RCT from Brazil that evaluated the use of IV dexamethasone 20mg for 5 days followed by 10mg for another 5 days in patients with COVID who were mechanically ventilated. Patients were randomized in a 1:1 fashion to intervention and standard of care. Primary outcome was ventilator-free days during the first 28 days, with mortality being a secondary outcome. The initial power analysis required 350 patients, but this was stopped after 299 were recruited following the publication of the RECOVERY trial. Patients in the dexamethasone group had a higher mean number of days free from mechanical ventilation (6.6, 95% CI 5-8.2 days vs 4, 95% CI 2.9-5.4 days; difference 2.26, 95% CI 0.2-4.38, p=0.04), with no significant mortality differences at day 28 (56.3% vs 61.5% in the dexamethasone group and standard of care group, respectively; HR 0.97, 95% CI 0.72 to 1.31).
While this study did reach its primary outcome, the early termination may have prevented other secondary outcomes from reaching statistical significance. In comparison to the recovery trial, it seems that higher doses of dexamethasone may not add much (6mg vs 20mg), though there were no significant differences in terms of infections between groups, which is reassuring.
The REMAP-CAP trial (225), which is a multicenter, open label trial that randomizes patients to multiple interventions within multiple domains, evaluated the use of hydrocortisone in COVID-19 patients. Patients were randomized in a 1:1:1 fashion if they were admitted to the ICU to fixed-dose hydrocortisone (50mg q6h for 7 days), shock-dependent dosing (50mg q6h for 28 days), and no hydrocortisone. Primary outcome was respiratory and cardiovascular organ support-free days at day 21 and this was analyzed with a bayesian cumulative logistic model to estimate posterior probability distributions. 403 patients were randomized, with a median organ-support free days of 0 for each study group, likely reflecting prolonged critical illness. The bayesian model found that fixed dose steroids (93%) and shock-dependent steroids (80%) were likely superior to no steroids. Notably, this study was terminated early as well and the probabilities did not meet the prespecified probabilities (99%).
The CAPE-COVID trial (226) is a placebo controlled, double blind, randomized controlled trial that was attached to the parent CAPE COD trial which was designed to determine the superiority of hydrocortisone in reducing 28 day mortality in ICU patients compared to placebo. Patients admitted with severe COVID to any one of 9 French ICUs were eligible, with patients randomized in a 1:1 fashion to receive hydrocortisone 200mg/day for 7 days followed by 100mg/day for 4 days and 50mg/day for 3 days (total 14) versus placebo. Primary outcome was treatment failure at day 21 (defined as death or persistent dependency on mechanical ventilation or high-flow oxygen therapy). The calculated sample size was 290, however the trial was terminated early when enrollment slowed down towards the end of the first wave and the anticipated release of RECOVERY. 149 patients were randomized, with treatment failure occurring in 42.1% of patients in the hydrocortisone group compared to 50.7% in the placebo group (difference -8.6%, 95% CI -24.9% to 7.7%). Post-hoc outcomes did not differ between groups either, though patients in the hydrocortisone group were less likely to die at day 21 (14.7% vs 27.4%) and were more likely to have been discharged from the ICU (57.3% vs 43.8%). This suggest a trend towards the benefit of steroids but this is likely underpowered to show this effect.
A meta-analysis of 7 studies (227) evaluated the mortality benefit of several steroids (dexamethasone, hydrocortisone, and methylprednisone) in severe COVID patients. The vast majority of these patients were either on mechanical ventilation or admitted the ICU. The overall fixed-effect meta-analysis demonstrated an absolute mortality risk of 32% with corticosteroids compared to 40% in the usual group (OR 0.66, 95% CI 0.53-0.82). Most of these results were driven by the RECOVERY trial, with the analysis that excluded these patients having an OR of 0.77 (95% CI 0.56-1.07), suggesting a trend towards benefit here.
Subgroup analysis suggest major benefit for those who have been symptomatic >7 days and on mechanical ventilation:
4. Monoclonal antibodies – these are some up and coming therapies that are in early development. One of the first developed monoclonal antibodies, bamlanivimab, is an anti-spike antibody created to neutralize SARS-CoV2. It first showed promise in monkeys with significant reduction in viral loads when given prior to infection, so not terribly helpful (228):
The ACTIV-3 trial, unfortunately, was stopped early due to futility. The BLAZE-1 trial (229) is a phase II trial evaluating the use of bamlanivimab in outpatients. 452 patients were randomized to either placebo or 3 different doses of the MAB. Primary endpoint was change in viral load at 11 days. Primary outcome was achieved in the 2800mg group
Though patients in the treatment group were overall less likely to have been hospitalized (secondary outcome):
And seem to have improved symptoms at day 11 in comparison to placebo:
A few things to note here, though. First, the trial was designed by the manufacturer of the drug, Eli Lilly. So take that as you will. Second, they define hospitalizations as a composite of both ED visits and in-patient admissions, based on the rationale that most ED visits lead to hospitalizations. I do not know how I feel about that. Third, on the symptom improvement, they lump all other doses, though viral load reduction was only achieved with the 2800mg reduction. All seems very strange to me, almost as if they were trying to get some positive secondary outcome. Either way, I do not think the data supports the use of this -mab yet. We’ll wait and see. Seems the next step is to create a cocktail using this -mab and another one and try that one. Either way, as of writing this both NIH and CDC guidelines recommend against the use of bamlanivimab, though some centers are using it as it was granted Emergency Use Authorization (EUA).
What about REGN-CoV2, the combination of casirivimab and imdevimab? A multicenter, RCT, placebo-controlled double blind trial (230) evaluated its use in symptomatic, non-hospitalized patients with COVID-19. This was the first 2 phases of the trial, were patients were randomized in a 1:1:1 fashion to get placebo, dose of 2.4g or 8g. They evaluated patients who had a positive result within 72hrs prior to randomization and had been symptomatic for no longer than a week. Primary end point was average change in viral load at day 7 as well as one COVID-19 related visit through day 29. They also stratified patients into patients who had antibodies to the virus already and those who did not (to assess their hypothesis that those who did not have antibodies yet would get the most benefit from the cocktail).
30 of 275 patients were PCR negative for COVID, and 123/275 were antibody positive. For all groups, REGN-CoV2 had a greater drop in viral load by day 7 compared to placebo, mostly driven by the 8g dose (2.4g dose did not achieve statistical significance):
Those who were antibody negative yielded the most benefit here, with antibody positive patients not achieving a statistically significant result at either dose:
The difference was also more pronounced in those with higher baseline viral loads:
For COVID-19 related visits, there was no difference between the cocktail or placebo at day 29:
Notably, this study was not powered given the lack of information going into the study, though this trial enrolled healthy patients. While the cocktail seems to improve viral loads in those without underlying immunity against SARS-CoV2, the lack of significant clinical endpoints leaves a lot to be desired. Hopefully, phase III will bring us more info regarding this cocktail.
5. Convalescent plasma – The idea behind this is that you give someone passive immunity with neutralizing antibodies from someone who has recovered. This has been used in the past for the 1918 pandemic, MERS, SARS, and Ebola (101). Initial data came from case series and case reports (102, 103, 104, 210). Other data came from compassionate use trials (208) and smaller open label trials (209). One of the interesting tidbits of early case series was the fact treatment success was seen even when given late in the treatment. How can this be, as neutralizing antibodies tackle the virus while late-stage pathophysiology is driven by the cytokine storm.
It may be that convalescent plasma has more than neutralizing antibodies (101). In other words, it may be that it also plays a role in controlling the cytokine storm.
Despite early case series raving about the effects of convalescent plasma, later trials have not yielded favorable results. The first randomized trial came out of China (105) where the time to clinical improvement at 28 days did not differ between convalescent plasma group and standard of care group. In subgroup analysis, there was a statistical difference in time to improvement in the severe disease group (4.94 days) and clinical improvement at 28 days occurred in 91% of those given convalescent plasma when compared to 68% of those who didn’t. This did not hold up for the life-threatening group. There was also no statistical difference in mortality overall, but a trend towards improved mortality in those who received convalescent plasma (15.7% vs 24.0%, respectively; P = .30). It must be noted this is an underpowered study, since the investigators were unable to enroll enough patients due to the pandemic being under control.
The story is true with other larger trials. A double blind, placebo controlled, multicenter trial from Argentina (233) evaluated convalescent plasma in patients with O2 sats <93%, P/F ratio <300. Patients were randomized in a 2:1 fashion to treatment:placebo, with total neutralizing antibody titers goal being 1:800 for one total dose. Primary endpoint was clinical status 30 days based on a WHO ordinal categories. 334 patients were enrolled, with no difference noted between treatment group and placebo group in the distribution of clinical outcomes (OR 0.83, 95% CI 0.52 to 1.35), with no statistical significance after controlling for sex, history of COPD and tobacco abuse (OR 0.92, 95% CI 0.59 to 1.42). 30 day mortality was similar between groups (10.96% vs 11.43, risk difference -0.46, 95% CI -7.8 to 6.8)
The PLACID trial (234), the open label, parallel arm, phase II multicenter RCT evaluated convalescent plasma in patients with moderate to severe illness, though they excluded patients with P/F ratio <200 or shock requiring vasopressors. Patients were randomized in a 1:1 ratio to receive 2 doses of plasma 24 hours apart or standard of care. Primary outcome was composite of progression to severe disease (P/F ratio <100) or all-cause mortality at 28 days. 464 patients were randomized, with the composite outcome occurring in 44 (19%) of patients in the intervention arm and 41 (18%) in the control arm (unadjusted risk difference 0.008, 95% CI -0.0062 to 0.078, risk ratio 1.04, 95% CI 0.71 to 1.54).
There were also no difference in patients who had neutralizing antibodies prior to plasma or in those who had no neutralizing antibodies prior to therapy. There were also no differences in serum ferritin, CRP, LDH, or D-dimer levels between groups. It is unclear of where to go from here. For instance, CDC guidelines to not mention convalescent plasma as of November while the IDSA guidelines do not recommend its use outside of a clinical trial. As such, this may fall out of favor soon
TL;DR – given the state of the evidence, it may be that convalescent plasma will be phased out. CDC makes no mention of it and IDSA does not recommend its use.
ANTI-COAGULATION:
Clotting in COVID
It seems that COVID-19 tends to cause a predilection towards thrombosis, that is different from DIC (i.e. consumptive coagulopathy). This is not unusual, as there are many viruses that cause some sort of coagulopathy (88), however this differs from others in that it is more pro thromobotic than the original SARS (88). In general, it stands to reason that patients with septic shock have a higher incidence of DVT than those without septic shock. In one instance, the rate of PE was much higher in those with COVID-related ARDS when compared to matched controls (11.7% vs 2.1%, 89), though the rate of overall thrombotic events was not different. Whether or not this is because it was not looked for, it is difficult to tell.
One thing we do know is that those patients who had higher D-dimer and lower Fibrinogen tended to have higher mortality (23, 90).
Another retrospective study (91) found that a cutoff value for d-dimer of 2000ng/mL (this will depend on the lab) will have a sensitivity/specificity of 93% and 83% respectively for mortality:
In a recent meta analysis (92), found that the mean D-dimer (in mg/dL) for those who died was 6.13, and those who had a positive D-dimer had a relative risk of 4.11 for all cause mortality (21% vs 4.9%).
Not only that, but for those patients who had COVID, those with positive D-dimer were twice as likely to have severe disease.
The significance of the elevated D-dimer is difficult to discern here. It may be a marker of just “extreme inflammation” mote than anything, and hence it carries prognostic value. Since patients are know to be prothrombotic, the recommendation has been to at least give patients prophylactic anticoagulation. This may not be enough, however. For instance a retrospective study of 184 Dutch patients, 31% of patients with COVID-19 in their ICU were found to have thrombotic events (either PE, DVT , or arterial embolus ) despite prophylactic anticoagulation (93). Similarly, in another study (in Padua University, no less), found that those with COVID-19 admitted to the ICU had coagulation profiles that fit a hypercoagulable profile that is different from consumptive coagulopathy (94). They also found that about 23% of their small sample had a DVT despite prophylaxis.
Why is this? It may be due to heparin resistance. Recall that heparin binds to anti-thrombin III, activating it, and inhibiting factor Xa. AT III deficiency can occur in settings such as liver failure, DIC, or acute thrombosis, or binding to acute phase reactants that increase during intense inflammation. Heparin is a negatively charged molecule that can bind to positively charged plasma proteins. This phenomena was demonstrated in a retrospective study (235), where 10 ICU patients who were getting UFH infusions. 8 of these required >35,000 units/day and 3 required >50,000 units/day (heparin resistance is defined as requiring >35,000 units/day). Another letter (236) described 4 patients requiring a mean of 48,708 units/day of UFH.
Another retrospective study (237) of 42 patients admitted to a critical care area and the ward are in COVID patients. anti-Xa levels were evaluated (0.6-1.0 IU/mL for BID LMWH and 1-2IU/mL for daily LMWH). Mean anti-Xa levels were significantly low in both groups:
A study of 56 ICU patients (238) evaluated anti-Xa levels and found that 41% of the time, they were out of target range, usually subtherapeutic. And yet another study of 31 ICU patients evaluated the dose of dalteparin prophylaxis and anti-Xa levels (239). Notably, most patients received dose >75U/Kg (prophylaxis dose for dalteparin is usually 5000 U/day). Despite this, many patients fell below the therapeutic anti-Xa level.
The implications of this is unclear. Despite this, anticoagulation has been associated with improved mortality. For instance, a retrospective study (95) evaluated the use of therapeutic anticoagulation in those with severe COVID-19 (RR>30, P/F <300, O2 <93%) vs no AC or use <7d. All cause mortality was the same in both groups but a closer look at the subgroup analysis demonstrated a mortality benefit in those with a D-dimer >6x the upper limit of normal. Unfortunately, there is no report as to if there was an increase in bleeding Or tracking of fibrinogen.
Another retrospective analysis (96) from NYC evaluated the impact of therapeutic anticoagulation on mortality. There was significant difference in the in-hospital mortality in the AC group at 21 days (22.5%) vs in the non-AC group at 14d (22.8%). The more impressive bit was on those who were mechanically ventilated, as shown below (29% at 21d vs 63% at 9d):
Smaller data has also suggested improvement in P/F ratios (240).
A small, open-label, randomized trial evaluated therapeutic vs prophylactic anticoagulation in 20 patients. Patients in the therapeutic anticoagulation group were improved their P/F ratios by day 14 compared to the prophylactic group, and significant drop in the D-dimer:
Further, more patients in the therapeutic group were liberated from the ventilator by day 28 (HR 4, 95% CI 1.035-15). These results are promising, but the incredibly small size of the trial limits the validity of its conclusion.
So what to do in this situation? Initially during the pandemic, D-dimer levels were used to guide intensity of anticoagulation. This was based on the previously cited study, however a review of the guidelines (241) did not recommend using this approach. From the review, “the ACF states that biomarker thresholds such as D-dimer for guiding anticoagulation management should not be done outside the setting of a clinical trial. Similarly, the SCC-ISTH remarks that D-dimer levels should not be used to solely guide anticoagulation regimens…The ACC mentioned that D-dimer >2 times ULN may suggest that patient is at high risk for VTE and to consider extended prophylaxis (up to 45 days) in patients with low risk of bleeding. “
When to Anticoagulate?
At the end of the day, the data for anticoagulation is all over the place and I do not think anyone really has an answer here. Some generalizations here:
- VTE prophylaxis with heparin for all patients is reasonable
- Measuring anti-Xa levels if using LMWH is also reasonable to avoid subtherapeutic anticoagulation
- If there is any other risk factors for increased risk of thrombosis in a COVID patient who is critically ill, it may be reasonable to proceed with therapeutic anticoagulation provided there is no risk of bleed.
Again, guidelines do not help much. A recent review from JACC (242) with regards to the question of full dose anticoagulation in patients without VTE did not reach a consensus, with “the majority of panel members consider prophylactic anticoagulation, although a minority consider intermediate-dose or therapeutic dose to be reasonable.” With regards to extended anticoagulation post-discharge, they recommend against it unless someone is at the highest risk for VTE, including co-morbidities like active cancer, elevated D-dimer >2 times ULN, and low risk of bleeding.
Heparin seems to be the prefer drug for anticoagulation. A nice review (243) highlights some non-anticoagulation benefits of the drug. For instance, it highlights its ability to bind inflammatory cytokines, inhibit neutrophil chemotaxis and leukocyte migration, and neutralizing positively charged C5a. Moreover, it may also act as an inhibitor of viral attachment. Regardless, heparin may be more than just an anticoagulant (i.e. may have magical properties like azithromycin).
COVID and Stroke
There has been a significant amount of papers written about the incidence of stroke in COVID infected patients. For instance, in one NEJM report (179), 5 patients who where under the age of 50 presented with CVA symptoms. One thing to note in this paper was these patients had few or no respiratory symptoms, and 3 of these patients had D-dimers >1500, suggesting COVID can induce a hypercoagulable state despite the lack of symptoms!
Further data suggest that this phenomena is not typical of respiratory viruses. For instance, a retrospective study from NY compared data from COVID patients and influenza patients and found that those with COVID had a higher incidence of stroke (180).
This suggest that, compared to other respiratory viruses, COVID induces a special “hypercoagulable” state that is not seen in other respiratory viruses. Indeed, even when compared with patients with stroke, those with COVID tend to be more hypercoagulable, with higher CRP and D-dimers seen in those who end up with a stroke. A retrospective study (181) evaluated 3556 patients with COVID and found that 32 of them had suffered a stroke. When evaluating it with another set of CVA patients without COVID and a historical control, those with COVID tended to be younger, have higher NIHSS score, have higher D-dimer and inflammatory markers. In hospital death was also significantly higher in this population:
TL;DR – patients with COVID are already hypercoagulable and are at a higher risk of CVA. This can occur even before someone has respiratory symptoms.
PULMONARY ISSUES:
Prehospital monitoring:
There has been a movement towards outpatient monitoring of COVID patients with pulse oximeters. For instance, one editorial advocated for their use in the pre-hospital setting as a triage tool that can be use to expedite admissions for those who develop worsening hypoxemia (168).
Given that those patients who become hypoxemic deteriorate quickly (see below), this makes sense. In a retrospective study (169) from France, it was found there was a huge discrepancy between SpO2 and respiratory rate, essentially noting that, when compared to prior years, there was a higher proportion of people with low SpO2 but normal respiratory rate (aka happy hypoxemia, see below):
Because of this, programs (170) have launched for at-home monitoring of patients that have tested positive for COVID. Indeed, this seems to be a reasonable approach with findings that smart phone pulse oximeters correlating fairly well, up to 96-99%, with standard oximeters (171), however a review by CEBM does not support the use of app-based pulse ox (172). Having said that, if a patient is able to get an at-home pulse ox, this may be a reasonable option for outpatient monitoring.
Happy Hypoxemia
This is essentially someone who is profoundly hypoxic despite being on high oxygen and not tachypnic. Why is this? Remember, that oxygenation depends on inspired oxygen ensuring alveoli are not collapsed. Ventilation (CO2) depends on respiratory rate and minute ventilation. The change in rate of breathing occurs largely to correct ventilation issues, not oxygenation issues. There is a bit more that goes into this.
Since the initial pathophysiology involves infection of type II pneumocytes and lack of surfactant -> alveoli collapse -> V/Q mismatch and at the most extreme -> shunt.
It is not into the later stages where there is fibrosing of alveoli and possible pulmonary emboli where increased work of breathing occurs. Having said that, this “happy hypoxemia” may look innocent enough, but may lead to the sudden “crash and burn” that is seen in some COVID patients. To explain why this may be the case, lets look back at the pathophys:
So what to do here? Awake proning! The landmark trial here was the 2013 PROSEVA trial (106), a French multicenter open label trial where patients with severe ARDS were randomized to standard of care or proning >16h for >20d. Primary outcome was 28d mortality (32.8% in the standard care group vs 16% in the proning group).
Early on during the COVID pandemic in China, a team used an early warning system to identify patients who could decompensate from a respiratory standpoint (107). They found that early awake proning (while on HFNC and NC) could be used to avoid intubation and improve mortality.
One of the issues of awake proning is selection. In one small retrospective study, 24 patients were recruited and out of these, only 15 were able to maintain prone positioning for >3 hours (108). OF these, only 6 responded favorably (representing a PaO2 increase by >/=-20), or 25% of the overall sample. Another case series from Singapore (109) evaluated a protocol of awake proning of one hour per session, 5 sessions a day, separated by 3 hours. They saw favorable results in 10 patients (9 weaned off O2, one deceased), however there was no control group and the sample size was small.
Another case series from NYC (110) evaluated the first 50 patients that came in during the prespecified period. They evaluated the difference in SpO2 before and after proning. They saw a significant increase in SpO2 from 80% to 94% about 5 minutes after self proning, but about 24% of these patients ultimately ended up intubated after 24hrs. Lastly, another case series from China evaluated the use of prone and lateral positioning in 48 patients (111). Of these, 13 actually did well and were discharged (!) and were not included in the analysis. 7 did not tolerate and died. The rest seem actually had a drop in their P/F Ratio and improvement in their tachypnea (a lot of data was missing, so take this with a grain of salt).
In another data set from Columbia in NYC, 29 patients that came in to the step down unit were proned on admission (165) for at least an hour. Primary end point here was change in SpO2 within the hour. A good percentage of those patients had a rise in their SpO2 without changing their FiO2:
However 6 patients whose SpO2 did not rise above 95% within the hour ended up intubated
This suggest that awake proning can get you to avoid intubation in some patients, but those who do not respond within a few hours, it may be best to tube them after they fail awake proning.
TL;DR – early awake proning in someone who is not in respiratory distress (i.e .tachypnea aka the happy hypoxemia) may be a reasonable thing to try. Protocols are up in the air, but if patients are able to tolerate, the PROSEVA protocol (16hrs proned, 8 recumbent) seems reasonable.
Assorted Items:
Children:
Kids tend to have a different response to SARS-CoV2 when compared to adults. Indeed, a retrospective study sought to analyze children with COVID (115), at that time, the number of pediatric cases made up about 2.66% of all the cases in China. We do know children tend to get more affected by other viral respiratory diseases differently from adults, with RSV being an obvious example. There seems to be more data surrounding the idea that children are not involved in super spreading events given their more indolent course and lack of symptoms. Part of this may be due to the degree of which nasal gene expression of ACE-2 varies in adults and children (116). A retrospective study of nasal epithelium from Mount Sinai conducted from 2015-2018 demonstrated a significantly reduced ACE-2 gene expression in those age <10. This went up exponentially, reaching near “normal” levels at around age 18.
Whether or not this means that the lower respiratory tract has less ACE-2 levels is unclear, however if there is less ACE-2 to go around in the nasopharynx, then the probability of being infected in the first place is much lower. This was confirmed in a recent study evaluating viral loads and ACE levels in a pediatric population from MGH (204). However, children with SARS-CoV2 infection were found to have significantly elevated viral loads, comparable to those of adult patients.
Further, kids who end up having up infected tend to have higher ACE-2 expression, but this did not correlate with viral load.
This means that while kids can have lower ACE-2 levels, once an infection sets up the viral load is comparable to that of adults. What this means in terms of transmission and super-spreading events in children is unknown.
The clinical presentation seems to be much milder in children as well. For instance, an observational cohort study from the Zhejian Providence in China found that roughly 19% of cases had cough, while only 36% had fever (117). A MMWR evaluated the pediatric cases from 2/12 to 4/2 (118). They found that out of 149K cases, only 1.7% of those were made up by children <18. The breakdown by age bracket was as follows
- 15-17 years: 813 (32%)
- 10-14 years: 682; 27%
- 5-9 years: 388; 15%
- 1-4 years: 291; 11%
- <1 years: 398: 15%
It is also noted that fever, cough, and shortness of breath were less frequently reported in the pediatric population. A summary comparing both adults and children with COVID-19 is highlighted below:
A relatively large review (119) evaluated 406 pediatric patients, aged 2 days to 16 years. They found that about 50.7% demonstrated fever and 42.4% had cough. Much less included shortness of breath (14%), sputum production (3%), runny nose (5.7%).
In terms of imaging (118, 119), it seems pediatric patients tend to have similar findings on CT, including consolidation and ground glass opacities. However, the degree to which imaging findings are seen on children tend to be associated with severity.
Role of Children in Transmission:
There has been a lot of opinion in the pediatric circles about letting young kids back to school, with the idea that children are not the super spreaders or main drivers of the pandemic.
Initially, there was some concern about children having attack rates similar to the older population, in one early study from China suggesting an attack rate of 7.4% for those under 9, while an attack rate for those 40-49 being reported as 4.9% (120). However a study from Vo, Italy (who was able to test roughly 80% of their population shortly after their first COVID related death), found that no children <10 tested positive for the virus, despite 10 of them living with someone who was positive (121). In a study from Iceland looking at the rate of positivity between targeted and screening testing, 6.7% of children <10 years of age tested positive in the targeted testing group in comparison to none in the screening testing (122). And in households with a COVID patient, the attack rate for those <20 years of age was roughly 5.26% (95% CI 2.43, 9.76%), as compared to 13.72% (95% CI 10.68, 17.24%) and 17.69% (95% CI: 11.89, 24.83%) in 20-59 year olds and ≥60 year olds, respectively (p-values <0.005; 123)
A Japanese study found that attack rates were significantly lower in people <20, with a peak roughly at around age 50 for both males and females (124):
All this suggests that children are less likely to be infected than adults when in the same household, however many of these reports do not suggest that children are the index case in their household. Indeed, a recent report from Ireland found that 3 children with COVID-19 did not result in any secondary cases, despite being with a total of 894 contacts (125). Only one child was symptomatic (fever)
In New South Wales (126), a total of 18 COVID cases were found in 15 schools (9 students and 9 staff). 863 close contacts in these 15 schools were identified and only 2 students were identified as secondary cases. A retrospective study of 31 family clusters identified only 3 pediatric index cases (127) and data from the Neatherlands suggests that in 10 patients with COVID-19 who were <18yo, the 43 close contacts identified did not become ill when compared to 8.3% of the close contacts of the 221 adult patients who were ill (128).
All told, it seems that children are less susceptible to COVID-19 infection due to their lower levels of ACE2 expression in the nasopharynx and this may, in turn, having a less chance of being superspreaders. Further, there is no evidence of child to child transmission in areas of high person to person contact (i.e. schools). Finally, it also seems that in family clusters, it is the adult that serves as the index case rather than the child. Whether this is related to a higher level of viremia in the adult vs a child is unknown. This may change, given that children have been found to have viral loads that match that of adults with severe infection (204). However, epidemiological data thus far suggest this is not the case. This is an evolving topic, however.
Transplant:
This is a very broad topic and I do not think I can do it enough justice, as there are numerous nuances and as more data emerges, we will learn more. What am I not going to cover?
- Immunosuppression management immediately post-transplantation
- Which induction agent to use if there is suspicion of COVID (of course, I doubt anyone would transplant here).
- Immunosuppression following treatment of rejection
Needless to say, this may end up being out of date. Further, a lot the data coming up are essentially case series and mostly descriptive experiences of certain transplant centers around the world. Most of the experience comes from, not surprisingly, the kidney transplant population. In an attempt to piss off transplant people and alienate them, allow me to tell you what I have found.
There are some generalizations that can be made:
- The clinical and lab characteristics mirror those of the immunocompetent
- The highest mortality patients seem to be those immediately post transplant or those far removed (>3yrs)
- Most of the immunosuppression strategies involved either reducing or stopping antimetabolites and increasing steroids to pulsed-dose in some cases.
I. Kidney Transplant – The largest case series comes from Columba University (129), where they evaluated 15 patients with COVID. Median time since transplant was 49 months. Symptoms mirror those of the immunocompetent population
The management was as follows:
One of the things the authors noted is the following “We should note that the majority of the patients in our cohort were on prednisone at the time of diagnosis despite our status as an early steroid withdrawal center for most kidney transplant recipients, perhaps suggesting that the greater immunosuppression associated with the use of an immunosuppressive three-drug regimen may predispose patients to a more severe infection requiring hospitalization.”
And just like in immunocompetent folks, CRP can be used to track badness.
Another case series from the Netherlands (130) describes 23 SOT patients, 15 of which were kidney transplants. Most of the symptoms mirror what has been reported, including fever in 81%, cough in 71% and dyspnea in 59%. They also report similar lab findings:
The majority of the patients had mild symptoms and they made no changes in the immunosuppression of 13/23. 14 patients were discharged, 2 remained in the ICU at the time of publication, and 5 had died. Those who had a more severe outcome scored higher in the clinical frailty scale (CFS; 5.8 vs 1.92).
More recently, Montefiore reported 36 consecutive cases (131) of COVID in kidney transplant cases. At a median follow up of 21 days, 10 out of 36 transplant patients had died, including 7/11 of those who were intubated. This also included 2 recent transplant recipients who had received anti-thymocyte induction in the preceding 5 weeks.
II. Lung Transplant – A case series from Philadelphia (132) highlighted 8 COVID patients in transplant patients. Like previously, the symptoms mirrored to those of immunocompetent patient as did labs:
All patients were admitted and 6 of them ended up having their antimetabolite stopped. At 24 days, 2 of the patients had died, both of which had undergone lung transplant in the prior 2 weeks. The authors note the infection was likely hospital acquired.
A group from Italy (133) reported on 4 patients who presented with COVID following lung transplantation (range 43-72 months). 3 patients survived with a combination of cessation of antimetabolite and increased steroids (IV pulsed solu-medrol). The 4th patient died following complications with ESBL K pneumonia off her BAL and aspergillosis. This leads me to:
Aspergillus Co-Infection
Just like influenza, it seems that SARS-CoV2 infection is a risk factor for aspergillus co-infection. Of course, the difficulty here, as with influenza, is diagnosis. The EORTC/MSG criteria caters to the neutropenic populations, but the AspICU (134) algorithm has been proposed to circumvent this, with sensitivities hoovering around 60% and specificity of ~90% for the ICU populations:
This essentially relaxes some of the microbiology requirements for putative invasive aspergillosis.
In one retrospective analysis of 27 consecutive ICU mechanically ventilated patients found that a third of them fit criteria for putative IPA (135). They didn’t find a difference in mortality between the mechanically ventilated IPA patients and those that did not meet criteria for IPA; four of nine [44%] vs seven of 18 [39%], p=0·99. 6 of the 9 diagnosed were given glucocorticoids prior to the diagnosis for ARDS/Pneumonia. Many of these were diagnosed with a combination of BAL GM, serum GM, and serum BD glucan:
Another retrospective case series found 5 patients with severe ARDS with mycology consistent with IPA (136). These patient would not have met criteria by the original EORTC/MSG criteria and they also point that most of them had imaging findings consistent with viral pneumonia. Only one was using IV steroids (2 were on inhaled steroids for COPD), and 3/5 had died at the time of publication.
A retrospective series of 104 COVID patients admitted to a hospital in China identified 7 patients with IPA (137). These patients tended to be older (mean 73yo vs 53yo) and all of the IPA patients required ICU admission. Mead glucocorticoid dosage (solu-medrol) was 70mg vs 40mg per day, though it was not statistically significant. They were diagnosed on a median of 19 days after COVID diagnosis. In the Neatherlands, 6 patients with IPA were identified in a cluster of 31 patients with COVID that had been admitted to the ICU (138). Patients were diagnosed at around day ~12 of their illness, and 4 of the 6 patients died at around hospital day 12. They also note these patients did not have classic risk factors for IPA.
TL;DR: seems that COVID, just like influenza, is a risk factor for invasive pulmonary aspergillosis. Given this, a lot of these patients will be missed since they do not fit the classic “EORTC/MSG” risk factors such as liquid malignancies and prolonged steroid use. Have a high index of suspicion; cut off BAL GM of >1 and off serum of 0.5 seems to be what has been used. This further complicates the matter of steroid use (see above).
Animals/Pets
There was recently a report from the Netherlands (140) that suggested a farmer was infected by exposure to a mink, suggesting that several animals can be infected. Indeed, there was been reports from Europe suggesting that domestic cats and dogs have been found to harbor the virus as well other feline animals, but the impact of this in human transmission remains to be elucidated.
One of the first cases of domestic animal SARS-CoV2 infection occurred in Hong Kong (141). An elderly lady was diagnosed with COVID-19 and her dog (a 17yo Pomeranian) was swabbed at least 3 times and had a low level PCR positivity on both salivary and nasal swabs. He was also found to have low anti-SARS-CoV2 antibodies, but never displayed symptoms suggestive of the disease.
Further cases have been identified, but seems that transmission occurs from Humans to Pets (142):
– A German shepherd in Hong Kong became ill after its owner tested positive for COVID. The owner actually had 2 dogs, one of which actually tested negative. The Germen shepherd tested positive for the same virus as the owner, however the PCR was low positive and the dog never developed symptoms
– A pet cat tested positive for PCR from feces and gastric fluids after its owner developed GI and respiratory symptoms of COVID-19.
– Another pet cat from Hong Kong was tested after its owner came down with COVID. They tested its oral cavity, and obtained nasal and rectal samples, all of which tested positive. He did not develop any symptoms
Other reports of animals in the Bronx Zoo (143) testing positive for SARS-CoV2 were making news in the early part of the pandemic:
– 4 Siberian Tigers and 3 African lions who are house in 2 enclosures in the Bronx Zoo tested positive for SARS-CoV2, however none of the animals were found to be in respiratory distress.
By far, seems like animals are getting the virus from humans, and there has been no report of a pet infecting a human. Further, given the low viral load found in some of the aforementioned pets, it seems the risk of transmission at this time from pet to human is low.
Vitamin D:
The immunomodulatory role of vitamin D in pulmonary infections has been described in tuberculosis (144) and other types of pneumonia (145). For instance, vitamin D deficiency has been associated with progression of pulmonary tuberculosis (146) and a meta analysis also found that the risk of pulmonary tuberculosis increased with vitamin D deficiency. Further, another meta analysis of 25 RCTs found that vitamin D supplementation reduced the risk of acute respiratory tract infection (adjusted OR 0.88, 95% CI 0.81-0.96), with the highest effects been seen on those with severe vitamin D deficiency (<25; 145).
While there have been no papers published on Vitamin D supplementation and treatment in COVID-19, there is some retrospective data coming out in terms of how deficiency affects clinical progression. For instance, in southeast Asia, those patients with critical COVID-19 were found to have significantly lower vitamin D levels (147).
Another study of cases from Europe found a correlation (albeit a weak one) between vitamin D levels and both cases per 1 million as well as mortality (148). This correlation held true when analyzing the average vitamin D levels in COVID cases throughout Europe, with the observation that those countries with higher mortality rates (i.e. Italy and Spain), had worse COVID-related mortality (149):
Further, another study found an inverse correlation between vitamin D deficiency and CRP levels (150), with the correlation coefficient ® ranging from -0.93 to -1.0 among different age groups (and a difference of 12.1% between vitamin D deficient and non-deficient group). The other interesting tid bit here is the following. They compared the CRP levels (using this as a surrogate of inflammation) to vitamin D levels as well as income level. As shown below, they found that throughout all vitamin D levels and throughout most ages (except the very young), those with lower income levels had higher CRP levels, suggesting there was more inflammation in that population that was independent of vitamin D levels.
Another retrospective (200) study of 7807 individuals with confirmed COVID were more likely to have vitamin D deficiency (<20ng/mL; OR 1.50 95% CI 1.13-1.98). There was a trend towards higher rates of hospitalization in those who had low vitamin D, but this did not reach statistical significance in multivariate logistic regression analysis (aOR 1.95, 95% CI 0.99-4.78). RCTs on the use of vitamin D to prevent respiratory infections are not terribly impressive. In one RCT for adolescents and kids (201), the use of weekly vitamin D did result in a 24% reduction of developing non-influenza infection (HR 0.76 95% CI 0.61-0.94), but not for influenza, though vast majority of the patients had adequate vitamin D levels. In another RCT (202) for older adults (50-84), monthly vitamin D was not associated with difference of time to an acute respiratory infection (74.7% in vitamin D group vs 73.7% in placebo group, aHR 1.01 95% CI 0.94-1.07). Subgroup analysis of those who had vitamin D <25 found they had a higher risk of respiratory tract infection overall. In another study of 322 patients using monthly vitamin D or placebo (203), there was no difference in the overall number of upper respiratory tract infections in either group (593 in vitamin D group vs 611 in placebo group).
It may be reasonable to supplement weekly vitamin D if there is any deficiency, however the benefit of further supplementation beyond repletion is uncertain. Its use in treatment is also questionable at best. Currently, there is a pilot RCT that was published (217) which demonstrated benefit to vitamin D supplementation as therapy, however the trial is still ongoing and this pilot was done to power the study.
References:
1. Chen Y, Liu Q, Guo D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J Med Virol. 2020;92(4):418‐423. doi:10.1002/jmv.25681
2. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203(2):631‐637. doi:10.1002/path.1570
3. Copin, Marie-Christine, et al. “Time to Consider Histologic Pattern of Lung Injury to Treat Critically Ill Patients With COVID-19 Infection.” Intensive Care Medicine, 2020.
4. Chen, Nanshan, et al. “Epidemiological and Clinical Characteristics of 99 Cases of 2019 Novel Coronavirus Pneumonia in Wuhan, China: a Descriptive Study.” Lancet (London, England), vol. 395, no. 10223, 2020, pp. 507-513.
5. van Doremalen, Neeltje, et al. “Aerosol and Surface Stability of SARS-CoV-2 as Compared With SARS-CoV-1.” The New England Journal of Medicine, vol. 382, no. 16, 2020, pp. 1564-1567.
6. Chan JFW, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020; 395:514–23
7. Yu, Ping, et al. “A Familial Cluster of Infection Associated With the 2019 Novel Coronavirus Indicating Possible Person-to-Person Transmission During the Incubation Period.” The Journal of Infectious Diseases, vol. 221, no. 11, 2020, pp. 1757-1761.
8. Ghinai, Isaac, et al. “First Known Person-to-person Transmission of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) in the USA.” Lancet (London, England), vol. 395, no. 10230, 2020, pp. 1137-1144.
9. Hamner L, Dubbel P, Capron I, et al. High SARS-CoV-2 Attack Rate Following Exposure at a Choir Practice — Skagit County, Washington, March 2020. MMWR Morb Mortal Wkly Rep 2020;69:606–610. DOI:
10. James A, Eagle L, Phillips C, et al. High COVID-19 Attack Rate Among Attendees at Events at a Church — Arkansas, March 2020. MMWR Morb Mortal Wkly Rep 2020;69:632–635. DOI
11. Zhang W, Cheng W, Luo L, Ma Y, Xu C, Qin P, et al. Secondary transmission of coronavirus disease from presymptomatic persons, China. Emerg Infect Dis. 2020 Aug
12. Jiang FC, Jiang XL, Wang ZG, Meng ZH, Shao SF, Anderson BD, et al. Detection of severe acute respiratory syndrome coronavirus 2 RNA on surfaces in quarantine rooms. Emerg Infect Dis. 2020 Sep
13. Lin, Lu, et al. “Gastrointestinal Symptoms of 95 Cases With SARS-CoV-2 Infection.” Gut, vol. 69, no. 6, 2020, pp. 997-1001.
14. Young, Barnaby Edward, et al. “Epidemiologic Features and Clinical Course of Patients Infected With SARS-CoV-2 in Singapore.” JAMA, 2020.
15. Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus–Infected Pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. doi:10.1001/jama.2020.1585
16. Wei Zhang, Rong-Hui Du, Bei Li, Xiao-Shuang Zheng, Xing-Lou Yang, Ben Hu, Yan-Yi Wang, Geng-Fu Xiao, Bing Yan, Zheng-Li Shi & Peng Zhou (2020) Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes, Emerging Microbes & Infections, 9:1, 386-389, DOI: 10.1080/22221751.2020.1729071
17. Li D, Jin M, Bao P, Zhao W, Zhang S. Clinical Characteristics and Results of Semen Tests Among Men With Coronavirus Disease 2019. JAMA Netw Open. 2020;3(5):e208292. doi:10.1001/jamanetworkopen.2020.8292
18. Arons, Melissa M., et al. “Presymptomatic SARS-CoV-2 Infections and Transmission in a Skilled Nursing Facility.” The New England Journal of Medicine, vol. 382, no. 22, 2020, pp. 2081-2090.
19. J Rocklöv, PhD, H Sjödin, PhD, A Wilder-Smith, MD, COVID-19 outbreak on the Diamond Princess cruise ship: estimating the epidemic potential and effectiveness of public health countermeasures, Journal of Travel Medicine, Volume 27, Issue 3, April 2020, taaa030, https://doi.org/10.1093/jtm/taaa030
20. Frieden, Thomas R., and Christopher T. Lee. “Identifying and Interrupting Superspreading Events-Implications for Control of Severe Acute Respiratory Syndrome Coronavirus 2.” Emerging Infectious Diseases, vol. 26, no. 6, 2020, pp. 1059-1066.
21. Beldomenico, Pablo M.. “Do Superspreaders Generate New Superspreaders? a Hypothesis to Explain the Propagation Pattern of COVID-19.” International Journal of Infectious Diseases : IJID : Official Publication of the International Society for Infectious Diseases, 2020.
22. https://www.cdc.go.kr/board/board.es?mid=a30402000000&bid=0030
23. Zhou, Fei, et al. “Clinical Course and Risk Factors for Mortality of Adult Inpatients With COVID-19 in Wuhan, China: a Retrospective Cohort Study.” Lancet (London, England), vol. 395, no. 10229, 2020, pp. 1054-1062.
24. Zhou, Bo, et al. “The Duration of Viral Shedding of Discharged Patients With Severe COVID-19.” Clinical Infectious Diseases : an Official Publication of the Infectious Diseases Society of America, 2020.
25. Huang, Chaolin, et al. “Clinical Features of Patients Infected With 2019 Novel Coronavirus in Wuhan, China.” Lancet (London, England), 2020.
26. Young, Barnaby Edward, et al. “Epidemiologic Features and Clinical Course of Patients Infected With SARS-CoV-2 in Singapore.” JAMA, 2020.
27. Arentz, Matt, et al. “Characteristics and Outcomes of 21 Critically Ill Patients With COVID-19 in Washington State.” JAMA, 2020.
28. Wang, Dawei, et al. “Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China.” JAMA, 2020.
29. Ruan, Q., Yang, K., Wang, W. et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med 46, 846–848 (2020). https://doi.org/10.1007/s00134-020-05991-x
30. Wang, Fan, et al. “Characteristics of Peripheral Lymphocyte Subset Alteration in COVID-19 Pneumonia.” The Journal of Infectious Diseases, vol. 221, no. 11, 2020, pp. 1762-1769.
31. Guan, Wei-Jie, et al. “Clinical Characteristics of Coronavirus Disease 2019 in China.” The New England Journal of Medicine, vol. 382, no. 18, 2020, pp. 1708-1720.
32. Chung, Michael, et al. “CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV).” Radiology, vol. 295, no. 1, 2020, pp. 202-207.
33. Song, Fengxiang, et al. “Emerging 2019 Novel Coronavirus (2019-nCoV) Pneumonia.” Radiology, vol. 295, no. 1, 2020, pp. 210-217.
34. Pan, Feng, et al. “Time Course of Lung Changes at Chest CT During Recovery From Coronavirus Disease 2019 (COVID-19).” Radiology, vol. 295, no. 3, 2020, pp. 715-721.
35. Lauer, Stephen A., et al. “The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application.” Annals of Internal Medicine, vol. 172, no. 9, 2020, pp. 577-582.
36. Siddiqi, Hasan K., and Mandeep R. Mehra. “COVID-19 Illness in Native and Immunosuppressed States: a Clinical-therapeutic Staging Proposal.” The Journal of Heart and Lung Transplantation : the Official Publication of the International Society for Heart Transplantation, vol. 39, no. 5, 2020, pp. 405-407.
37. Bhatraju, Pavan K., et al. “Covid-19 in Critically Ill Patients in the Seattle Region – Case Series.” The New England Journal of Medicine, vol. 382, no. 21, 2020, pp. 2012-2022.
38. Fang, Yicheng, et al. “Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR.” Radiology, 2020, p. 200432.
39. Ai, Tao, et al. “Correlation of Chest CT and RT-PCR Testing in Coronavirus Disease 2019 (COVID-19) in China: a Report of 1014 Cases.” Radiology, 2020, p. 200642.
40. Padhye, Nikhil S. “Reconstructed Diagnostic Sensitivity and Specificity of the RT-PCR Test for COVID-19.” 2020, doi:10.1101/2020.04.24.20078949.
41. Yang, Yang, et al. “Evaluating the Accuracy of Different Respiratory Specimens in the Laboratory Diagnosis and Monitoring the Viral Shedding of 2019-NCoV Infections.” Dec. 2020, doi:10.1101/2020.02.11.20021493.
42. Wang, Wenling, et al. “Detection of SARS-CoV-2 in Different Types of Clinical Specimens.” JAMA, 2020.
43. Wikramaratna, Paul, et al. “Estimating False-Negative Detection Rate of SARS-CoV-2 by RT-PCR.” July 2020, doi:10.1101/2020.04.05.20053355.
44. Kucirka, Lauren M., et al. “Variation in False-Negative Rate of Reverse Transcriptase Polymerase Chain Reaction-Based SARS-CoV-2 Tests By Time Since Exposure.” Annals of Internal Medicine, 2020.
45. Xu, Ruoshi, et al. “Saliva: Potential Diagnostic Value and Transmission of 2019-nCoV.” International Journal of Oral Science, vol. 12, no. 1, 2020, p. 11.
46. Wyllie, Anne L, John Fournier et al. “Saliva Is More Sensitive for SARS-CoV-2 Detection in COVID-19 Patients than Nasopharyngeal Swabs.” MedRxiv, 2020, doi:10.1101/2020.04.16.2006783.
47. Zhao Rongqing et al Early detection of SARS-CoV-2 antibodies in COVID-19 patients as a serologic marker of infection, Clinical Infectious Diseases, , ciaa523, https://doi.org/10.1093/cid/ciaa523
48. Xiang, Fei, et al. “Antibody Detection and Dynamic Characteristics in Patients With COVID-19.” Clinical Infectious Diseases : an Official Publication of the Infectious Diseases Society of America, 2020.
49. Jin, Yujiao, et al. “Diagnostic Value and Dynamic Variance of Serum Antibody in Coronavirus Disease 2019.” International Journal of Infectious Diseases : IJID : Official Publication of the International Society for Infectious Diseases, vol. 94, 2020, pp. 49-52.
50. Lou, Bin, et al. “Serology Characteristics of SARS-CoV-2 Infection Since Exposure and Post Symptom Onset.” The European Respiratory Journal, 2020.
51. Long, Quan-Xin, et al. “Antibody Responses to SARS-CoV-2 in Patients With COVID-19.” Nature Medicine, 2020.
52. Zhang, Guoxin, et al. “Longitudinal Change of Severe Acute Respiratory Syndrome Coronavirus 2 Antibodies in Patients with Coronavirus Disease 2019.” The Journal of Infectious Diseases, Feb. 2020, doi:10.1093/infdis/jiaa229.
53. Xiao, Ai Tang, et al. “Profile of Specific Antibodies to SARS-CoV-2: the First Report.” The Journal of Infection, 2020.
54. Zhao, Juanjuan, et al. “Antibody Responses to SARS-CoV-2 in Patients of Novel Coronavirus Disease 2019.” Clinical Infectious Diseases : an Official Publication of the Infectious Diseases Society of America, 2020
55. Yousefifard, Mahmoud, et al. “Non-steroidal Anti-inflammatory Drugs in Management of COVID-19; a Systematic Review On Current Evidence.” International Journal of Clinical Practice, 2020, pp. e13557.
56. Gautret et al. (2020) Hydroxychloroquine and azithromycin as a treatment of COVID‐19: results of an open‐label non‐randomized clinical trial. International Journal of Antimicrobial Agents – In Press 17 March 2020 – DOI : 10.1016/j.ijantimicag.2020.105949
57. Million, Matthieu, et al. “Early Treatment of COVID-19 Patients With Hydroxychloroquine and Azithromycin: a Retrospective Analysis of 1061 Cases in Marseille, France.” Travel Medicine and Infectious Disease, 2020, p. 101738.
58. Tang, Wei, et al. “Hydroxychloroquine in Patients With Mainly Mild to Moderate Coronavirus Disease 2019: Open Label, Randomised Controlled Trial.” BMJ (Clinical Research Ed.), vol. 369, 2020, pp. m1849.
59. Geleris, Joshua, et al. “Observational Study of Hydroxychloroquine in Hospitalized Patients With Covid-19.” The New England Journal of Medicine, 2020.
60. Mahévas, Matthieu, et al. “Clinical Efficacy of Hydroxychloroquine in Patients With Covid-19 Pneumonia Who Require Oxygen: Observational Comparative Study Using Routine Care Data.” BMJ (Clinical Research Ed.), vol. 369, 2020, pp. m1844.
61. Chen, Zhaowei, et al. “Efficacy of Hydroxychloroquine in Patients with COVID-19: Results of a Randomized Clinical Trial.” 2020, doi:10.1101/2020.03.22.20040758.
62. Tang, Wei, et al. “Hydroxychloroquine in Patients With Mainly Mild to Moderate Coronavirus Disease 2019: Open Label, Randomised Controlled Trial.” BMJ (Clinical Research Ed.), vol. 369, 2020, pp. m1849
63. Borba, MS et al “Effect of High vs LowDoses of Chloroquine Diphosphate as Adjunctive Therapy for Patients Hospitalized With Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection A Randomized Clinical Trial. JAMA Network Open. 2020;3(4):e208857. doi:10.1001/jamanetworkopen.2020.8857
64. Magagnoli J, et al “Outcomes of hydroxychloroquine usage in the United States Veterans hospitalized with Covid-19” medRxiv 2020.04.16.20065920; doi: https://doi.org/10.1101/2020.04.16/20065920
65. Shittu, Mujeeb Olushola, and Olufemi Ifeoluwa Afolami. “Improving the Efficacy of Chloroquine and Hydroxychloroquine Against SARS-CoV-2 May Require Zinc Additives – a Better Synergy for Future COVID-19 Clinical Trials.” Le Infezioni in Medicina, vol. 28, no. 2, 2020, pp. 192-197.
66. Derwand, R, and M Scholz. “Does Zinc Supplementation Enhance the Clinical Efficacy of Chloroquine/hydroxychloroquine to Win Today’s Battle Against COVID-19?” Medical Hypotheses, vol. 142, 2020, p. 109815.
67. Carlucci, PM et al “Hydroxychloroquine and azithromycin plus zinc vs hydroxychloroquine and azithromycin alone: outcomes in hospitalized COVID-19 patients” medRxiv preprint doi: https://doi.org/10.1101/2020.05.02.20080036
68. Boulware, David R., et al. “A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19.” The New England Journal of Medicine, 2020.
69. Chu, C M., et al. “Role of Lopinavir/ritonavir in the Treatment of SARS: Initial Virological and Clinical Findings.” Thorax, vol. 59, no. 3, 2004, pp. 252-6.
70. Chan, K S., et al. “Treatment of Severe Acute Respiratory Syndrome With Lopinavir/ritonavir: a Multicentre Retrospective Matched Cohort Study.” Hong Kong Medical Journal = Xianggang Yi Xue Za Zhi, vol. 9, no. 6, 2003, pp. 399-406.
71. Cao, Bin, et al. “A Trial of Lopinavir-Ritonavir in Adults Hospitalized With Severe Covid-19.” The New England Journal of Medicine, vol. 382, no. 19, 2020, pp. 1787-1799.
72. Hung, Ivan Fan-Ngai, et al. “Triple Combination of Interferon Beta-1b, Lopinavir-ritonavir, and Ribavirin in the Treatment of Patients Admitted to Hospital With COVID-19: an Open-label, Randomised, Phase 2 Trial.” Lancet (London, England), vol. 395, no. 10238, 2020, pp. 1695-1704.
73. Wang, Yeming, et al. “Remdesivir in Adults With Severe COVID-19: a Randomised, Double-blind, Placebo-controlled, Multicentre Trial.” Lancet (London, England), vol. 395, no. 10236, 2020, pp. 1569-1578.
74. Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, Hohmann E, Chu HY, Luetkemeyer A, Kline S, Lopez de Castilla D, Finberg RW, Dierberg K, Tapson V, Hsieh L, Patterson TF, Paredes R, Sweeney DA, Short WR, Touloumi G, Lye DC, Ohmagari N, Oh MD, Ruiz-Palacios GM, Benfield T, Fätkenheuer G, Kortepeter MG, Atmar RL, Creech CB, Lundgren J, Babiker AG, Pett S, Neaton JD, Burgess TH, Bonnett T, Green M, Makowski M, Osinusi A, Nayak S, Lane HC; ACTT-1 Study Group Members. Remdesivir for the Treatment of Covid-19 – Final Report. N Engl J Med. 2020 Nov 5;383(19):1813-1826. doi: 10.1056/NEJMoa2007764. Epub 2020 Oct 8. PMID: 32445440; PMCID: PMC7262788.
75. Lee, Nelson, et al. “Effects of Early Corticosteroid Treatment On Plasma SARS-associated Coronavirus RNA Concentrations in Adult Patients.” Journal of Clinical Virology : the Official Publication of the Pan American Society for Clinical Virology, vol. 31, no. 4, 2004, pp. 304-9.
76. Chen, Rong-Chang, et al. “Treatment of Severe Acute Respiratory Syndrome With Glucosteroids: the Guangzhou Experience.” Chest, vol. 129, no. 6, 2006, pp. 1441-52.
77. https://emcrit.org/pulmcrit/steroid-covid/https://emcrit.org/pulmcrit/steroid-covid/ (tried to get the original article but was unable to find it; it may be missing somewhere in my hard drive but for now, this will do).
78. Li H, Yang SG, Gu L, et al. Effect of low-tomoderate- dose corticosteroids on mortality of hospitalized adolescents and adults with influenza A(H1N1)pdm09 viral pneumonia. Influenza Other Respir Viruses 2017; 11: 345–54.
79. Fang X, Mei Q, Yang T, et al. Low-dose corticosteroid therapy does not delay viral clearance in patients with COVID-19. J Infect. April 2020. doi:10.1016/j.jinf.2020.03.039
80. Wang, Y et al “Early, low-dose and short-term application of corticosteroid treatment in patients with severe COVID-19 pneumonia: single-center experience from Wuhan, China” medRxiv preprint doi: https://doi.org/10.1101/2020.03.06.20032342
81. Wu C, Chen X, Cai Y, et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. March 2020. doi:10.1001/jamainternmed.2020.0994
82. Fadel R et al “Early Short Course Corticosteroids in Hospitalized Patients with COVID-19” medRxiv preprint doi: https://doi.org/10.1101/2020.05.04.20074609
83. Xu X et al “Effective treatment of severe COVID-19 patients with tocilizumab” Proceedings of the National Academy of Sciences May 2020, 117 (20) 10970-10975; DOI: 10.1073/pnas.2005615117
84. Luo, Pan, et al. “Tocilizumab Treatment in COVID-19: a Single Center Experience.” Journal of Medical Virology, 2020.
85. Ip A et al “Hydroxychloroquine and Tocilizumab Therapy in COVID-19 Patients – An Observational Study” medRxiv preprint doi: https://doi.org/10.1101/2020.05.21.20109207
- Huet T, Beaussier H, Voisin O et al.Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol. 2020; (published online May 29.https://doi.org/10.1016/S2665-9913(20)30164-8
- Cavalli, Giulio, et al. “Interleukin-1 Blockade With High-dose Anakinra in Patients With COVID-19, Acute Respiratory Distress Syndrome, and Hyperinflammation: a Retrospective Cohort Study.” The Lancet Rheumatology, vol. 2, no. 6, 2020, pp. e325-e331.
88. Goeijenbier, M, et al. “Review: Viral Infections and Mechanisms of Thrombosis and Bleeding.” Journal of Medical Virology, vol. 84, no. 10, 2012, pp. 1680-96.
89. Helms, Julie, et al. “High Risk of Thrombosis in Patients With Severe SARS-CoV-2 Infection: a Multicenter Prospective Cohort Study.” Intensive Care Medicine, 2020.
90. Tang, Ning, et al. “Abnormal Coagulation Parameters Are Associated With Poor Prognosis in Patients With Novel Coronavirus Pneumonia.” Journal of Thrombosis and Haemostasis : JTH, vol. 18, no. 4, 2020, pp. 844-847.
91. Zhang, Litao, et al. “D-dimer Levels On Admission to Predict In-hospital Mortality in Patients With Covid-19.” Journal of Thrombosis and Haemostasis : JTH, vol. 18, no. 6, 2020, pp. 1324-1329.
92. Shah S et al “Elevated D-dimer levels are associated with increased Risk of Mortality in COVID-19: “A systematic review and meta analysis” medRxiv preprint doi: https://doi.org/10.1101/2020.04.29.20085407
93. Klok, F A., et al. “Incidence of Thrombotic Complications in Critically Ill ICU Patients With COVID-19.” Thrombosis Research, vol. 191, 2020, pp. 145-147.
94. Spiezia, Luca, et al. “COVID-19-Related Severe Hypercoagulability in Patients Admitted to Intensive Care Unit for Acute Respiratory Failure.” Thrombosis and Haemostasis, vol. 120, no. 6, 2020, pp. 998-1000.
95. Tang, Ning, et al. “Anticoagulant Treatment Is Associated With Decreased Mortality in Severe Coronavirus Disease 2019 Patients With Coagulopathy.” Journal of Thrombosis and Haemostasis : JTH, vol. 18, no. 5, 2020, pp. 1094-1099.
96. Paranjpe, Ishan, et al. “Association of Treatment Dose Anticoagulation With In-Hospital Survival Among Hospitalized Patients With COVID-19.” Journal of the American College of Cardiology, 2020.
97. Thachil, Jecko. “The Versatile Heparin in COVID-19.” Journal of Thrombosis and Haemostasis : JTH, vol. 18, no. 5, 2020, pp. 1020-1022.
98. Shi et al “The potential of low molecular weight heparin to mitigate cytokine storm in severe COVID-10 patients: a retrospective clinical study.” medRxiv preprint doi: https://doi.org/10.1101/2020.03.28.20046144
99. Atallah B et al “Anticoagulation in COVID-19” European Heart Journal – Cardiovascular Pharmacotherapy. doi:10.1093/ehjcvp/pvaa036
100. Bikdeli, Behnood, et al. “COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-up.” Journal of the American College of Cardiology, 2020.
101. Rojas, Manuel, et al. “Convalescent Plasma in Covid-19: Possible Mechanisms of Action.” Autoimmunity Reviews, vol. 19, no. 7, 2020, p. 102554.
102. Shen C, Wang Z, Zhao F, et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma [published online ahead of print, 2020 Mar 27]. JAMA. 2020;323(16):1582‐1589. doi:10.1001/jama.2020.4783
103. Duan, Kai, et al. “Effectiveness of Convalescent Plasma Therapy in Severe COVID-19 Patients.” Proceedings of the National Academy of Sciences of the United States of America, vol. 117, no. 17, 2020, pp. 9490-9496.
104. Rajendran, Karthick, et al. “Convalescent Plasma Transfusion for the Treatment of COVID-19: Systematic Review.” Journal of Medical Virology, 2020.
105. Li L, Zhang W, Hu Y, et al. Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening COVID-19: a randomized clinical trial. JAMA. Published online June 3, 2020. doi:10.1001/jama.2020.10044
106. Guérin C, et al. “Prone positioning in severe acute respiratory distress syndrome”. The New England Journal of Medicine. 2013. 368(23):2159-2168.
107. Sun, Qin, et al. “Lower Mortality of COVID-19 By Early Recognition and Intervention: Experience From Jiangsu Province.” Annals of Intensive Care, vol. 10, no. 1, 2020, p. 33.
108. Elharrar, Xavier, et al. “Use of Prone Positioning in Nonintubated Patients With COVID-19 and Hypoxemic Acute Respiratory Failure.” JAMA, 2020.
109. Ng, Ziqin, et al. “Awake Prone Positioning for Non-intubated Oxygen Dependent COVID-19 Pneumonia Patients.” The European Respiratory Journal, 2020.
110. Caputo ND, Strayer RJ, Levitan R. Early Self-Proning in Awake, Non-intubated Patients in the Emergency Department: A Single ED’s Experience During the COVID-19 Pandemic. Acad Emerg Med. 2020;27(5):375‐378. doi:10.1111/acem.13994
111. Dong, W et al “Early Awake Prone and Lateral Position in Non-intubated Severe and Critical Patients with COVID-19 in Wuhan: A Respective Cohort Study” medRxiv preprint doi: https://doi.org/10.1101/2020.05.09.20091454
112. Caly, Leon, et al. “The FDA-approved Drug Ivermectin Inhibits the Replication of SARS-CoV-2 in Vitro.” Antiviral Research, vol. 178, 2020, p. 104787.
113. Sharun, Khan, et al. “Ivermectin, a New Candidate Therapeutic Against SARS-CoV-2/COVID-19.” Annals of Clinical Microbiology and Antimicrobials, vol. 19, no. 1, 2020, p. 23.
114. Choudhary, Renuka, et al. “Potential Use of Hydroxychloroquine, Ivermectin and Azithromycin Drugs in Fighting COVID-19: Trends, Scope and Relevance.” New Microbes and New Infections, 2020, p. 100684.
115. Dong Y, Mo X, Hu Y, et al. Epidemiology of COVID-19 Among Children in China. Pediatrics. 2020;145(6):e20200702
116. Bunyavanich, Supinda, et al. “Nasal Gene Expression of Angiotensin-Converting Enzyme 2 in Children and Adults.” JAMA, 2020.
117. Qiu, Haiyan, et al. “Clinical and Epidemiological Features of 36 Children With Coronavirus Disease 2019 (COVID-19) in Zhejiang, China: an Observational Cohort Study.” The Lancet. Infectious Diseases, vol. 20, no. 6, 2020, pp. 689-696.
118. CDC COVID-19 Response Team. “Coronavirus Disease 2019 in Children – United States, February 12-April 2, 2020.” MMWR. Morbidity and Mortality Weekly Report, vol. 69, no. 14, 2020, pp. 422-426.
119. Zhen-Dong, Yang, et al. “Clinical and Transmission Dynamics Characteristics of 406 Children With Coronavirus Disease 2019 in China: a Review.” The Journal of Infection, 2020.
120. Bi, Qifang, et al. “Epidemiology and Transmission of COVID-19 in 391 Cases and 1286 of Their Close Contacts in Shenzhen, China: a Retrospective Cohort Study.” The Lancet. Infectious Diseases, 2020.
121. Lavezzo E, et al. “Suppression of COVID-19 outbreak in the municipality of Vo, Italy. medRxiv preprint doi: https://doi.org/10.1101/2020.04.17.20053157
122. Gudbjartsson, Daniel F., et al. “Spread of SARS-CoV-2 in the Icelandic Population.” The New England Journal of Medicine, vol. 382, no. 24, 2020, pp. 2302-2315.
123.Jin Q et al, Household secondary attack rates of COVID-19 and Associated Determinants. medRxiv preprint doi: https://doi.org/10.1101/2020.04.11.20056010
124.Mizumoto et al “Age specificity of cases and attack rate of novel coronavirus disease (COVID-19)” medRxiv preprint doi: https://doi.org/10.1101/2020.03.09.20033142
125. Heavey, Laura, et al. “No Evidence of Secondary Transmission of COVID-19 From Children Attending School in Ireland, 2020.” Euro Surveillance : Bulletin Europeen Sur Les Maladies Transmissibles = European Communicable Disease Bulletin, vol. 25, no. 21, 2020.
- National Centre for Immunization and Surveillance COVID-19 in schools – the experience in NSW, 2020. Available: http://ncirs.org.au/sites/default/files/2020-04/NCIRS%20NSW%20Schools%20COVID_Summary_FINAL%20public_26%20April%202020.pd
- Zhu Y et al. “Children are unlikely to be the primary source of household SARS-CoV2 infections” medRxiv preprint doi: https://doi.org/10.1101/2020.03.26.20044826.
- The National Institute for Public Health and the Environment (RVIM). Children and COVID-19, 2020. Available: https://www.rivm.nl/en/novel-coronavirus-covid-19/children-and-covid-19 [Accessed June 18 2020]
- Columbia University Kidney Transplant Program. “Early Description of Coronavirus 2019 Disease in Kidney Transplant Recipients in New York.” Journal of the American Society of Nephrology : JASN, vol. 31, no. 6, 2020, pp. 1150-1156.
- Hoek, Rogier A S., et al. “Covid-19 in Solid Organ Transplant Recipients: a Single Center Experience.” Transplant International : Official Journal of the European Society for Organ Transplantation, 2020.
- Akalin, Enver, et al. “Covid-19 and Kidney Transplantation.” The New England Journal of Medicine, 2020.
- Myers, Catherine N., et al. “COVID-19 in Lung Transplant Recipients.” Transplant Infectious Disease : an Official Journal of the Transplantation Society, 2020, pp. e13364.
- Morlacchi LC, Rossetti V, Gigli L, et al. COVID-19 In Lung Transplant Recipients: A Case Series From Milan, Italy [published online ahead of print, 2020 Jun 8]. Transpl Infect Dis. 2020;e13356. doi:10.1111/tid.13356
- Blot SI, Taccone FS, Van den Abeele AM, et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients [published correction appears in Am J Respir Crit Care Med. 2012 Oct 15;186(8):808]. Am J Respir Crit Care Med. 2012;186(1):56-64. doi:10.1164/rccm.201111-1978OC
- Alanio, Alexandre, et al. “Prevalence of Putative Invasive Pulmonary Aspergillosis in Critically Ill Patients With COVID-19.” The Lancet. Respiratory Medicine, vol. 8, no. 6, 2020, pp. e48-e49.
- Koehler, Philipp, et al. “COVID-19 Associated Pulmonary Aspergillosis.” Mycoses, vol. 63, no. 6, 2020, pp. 528-534.
- Wang, Jie, et al. “Clinical Characteristics of Invasive Pulmonary Aspergillosis in Patients With COVID-19 in Zhejiang, China: a Retrospective Case Series.” Critical Care (London, England), vol. 24, no. 1, 2020, p. 299.
- Rutsaert, Lynn, et al. “COVID-19-associated Invasive Pulmonary Aspergillosis.” Annals of Intensive Care, vol. 10, no. 1, 2020, p. 71.
- Horby et al. “Effect of Dexamethasone in Hospitalized Patients with COVID-19 – A Preliminary Report.” medRxiv preprint doi: https://doi.org/10.1101/2020.06.22.20137273
- https://www.government.nl/ministries/ministry-of-agriculture-nature-and-food-quality/news/2020/05/19/new-results-from-research-into-covid-19-on-mink-farms
- PRO/AH/EDR> COVID-19 update (56): China (Hong Kong) Animal, Dog, Final Serology Positive. 2020. http://promedmail.org/post/20200326.7146438
- Leroy, Eric M et al. “The risk of SARS-CoV-2 transmission to pets and other wild and domestic animals strongly mandates a one-health strategy to control the COVID-19 pandemic.” One health (Amsterdam, Netherlands), 100133. 13 Apr. 2020, doi:10.1016/j.onehlt.2020.100133
- James A. A Tiger at Bronx Zoo Tests Positive for COVID-19. ABC News. Sunday 5 April, 2020. https://wpde.com/news/coronavirus/a-tiger-at-bronx-zoo-tests-positive-for-covid-19
- Nnoaham, Kelechi E., and Aileen Clarke. “Low Serum Vitamin D Levels and Tuberculosis: a Systematic Review and Meta-analysis.” International Journal of Epidemiology, vol. 37, no. 1, 2008, pp. 113-9.
- Martineau, Adrian R., et al. “Vitamin D Supplementation to Prevent Acute Respiratory Tract Infections: Systematic Review and Meta-analysis of Individual Participant Data.” BMJ (Clinical Research Ed.), vol. 356, 2017, p. i6583.
- Talat, Najeeha et al. “Vitamin d deficiency and tuberculosis progression.” Emerging infectious diseases vol. 16,5 (2010): 853-5. doi:10.3201/eid1605.091693
- Alipio, Mark, Vitamin D Supplementation Could Possibly Improve Clinical Outcomes of Patients Infected with Coronavirus-2019 (COVID-19) (April 9, 2020). Available at SSRN: https://ssrn.com/abstract=3571484 or http://dx.doi.org/10.2139/ssrn.3571484
- Ilie, Petre Cristian, et al. “The Role of Vitamin D in the Prevention of Coronavirus Disease 2019 Infection and Mortality.” Aging Clinical and Experimental Research, 2020.
- Laird et al “Vitamin D and Inflammation: Potential Implications for Severity of COVID-19” Irish Medical Journal. Vol 113; No. 5; p81
- Daneshkhan A et al, “The Possible Role of Vitamin D in Suppressing Cytokine Storm And Associated Mortality in COVID-19 Patients” medRxiv preprint doi: https://doi.org/10.1101/2020.04.08.20058578
- Abdel-Wahab N, Talathi S, Lopez-Olivo M, Suarez-Almazor M. Risk of developing antiphospholipid antibodies following viral infection: a systematic review and meta-analysis. Lupus. 2018;27(4):572-583. doi:10.1177/0961203317731532
- Beyrouti R, Adams ME, Benjamin L, et al. Characteristics of ischaemic stroke associated with COVID-19. J Neurol Neurosurg Psychiatry. Published online April 30, 2020:jnnp-2020-323586. doi:10.1136/jnnp-2020-323586
- Zhang, Yan, et al. “Coagulopathy and Antiphospholipid Antibodies in Patients With Covid-19.” The New England Journal of Medicine, vol. 382, no. 17, 2020, pp. e38.
- Bowles, Louise, et al. “Lupus Anticoagulant and Abnormal Coagulation Tests in Patients With Covid-19.” The New England Journal of Medicine, 2020.
- Cao Y, Wei J, Zou L, et al. Ruxolitinib in treatment of severe coronavirus disease 2019 (COVID-19): A multicenter, single-blind, randomized controlled trial. Journal of Allergy and Clinical Immunology. Published online May 2020. doi:10.1016/j.jaci.2020.05.019
- Li, Yi, et al. “Transmission of Communicable Respiratory Infections and Facemasks.” Journal of Multidisciplinary Healthcare, vol. 1, 2008, pp. 17-27.
- Cheng, Vincent Chi-Chung, et al. “The Role of Community-wide Wearing of Face Mask for Control of Coronavirus Disease 2019 (COVID-19) Epidemic Due to SARS-CoV-2.” The Journal of Infection, vol. 81, no. 1, 2020, pp. 107-114.
- Lyu, Wei, and George L. Wehby. “Community Use of Face Masks and COVID-19: Evidence From a Natural Experiment of State Mandates in the US.” Health Affairs (Project Hope), 2020, pp. 101377hlthaff202000818.
- Chu, Derek K., et al. “Physical Distancing, Face Masks, and Eye Protection to Prevent Person-to-person Transmission of SARS-CoV-2 and COVID-19: a Systematic Review and Meta-analysis.” Lancet (London, England), 2020.
- Ho, Kin-Fai, et al. “Medical Mask Versus Cotton Mask for Preventing Respiratory Droplet Transmission in Micro Environments.” The Science of the Total Environment, vol. 735, 2020, p. 139510.
- Patel, Monita R., et al. “Performance of Oropharyngeal Swab Testing Compared to Nasopharyngeal Swab Testing for Diagnosis of COVID-19 -United States, January-February 2020.” Clinical Infectious Diseases : an Official Publication of the Infectious Diseases Society of America, 2020.
- Liu J, Huang J, Xiang D. Large SARS-CoV-2 Outbreak Caused by Asymptomatic Traveler, China [published online ahead of print, 2020 Jun 30]. Emerg Infect Dis. 2020;29(9):10.3201/eid2609.201798. doi:10.3201/eid2609.201798
- Nguyen Van Vinh Chau et al OUCRU COVID-19 research group, The natural history and transmission potential of asymptomatic SARS-CoV-2 infection, Clinical Infectious Diseases, , ciaa711, https://doi.org/10.1093/cid/ciaa711
- Jing, Qin-Long, et al. “Household Secondary Attack Rate of COVID-19 and Associated Determinants in Guangzhou, China: a Retrospective Cohort Study.” The Lancet. Infectious Diseases, 2020.
- Thompson, Alison E., et al. “Prone Positioning in Awake, Nonintubated Patients With COVID-19 Hypoxemic Respiratory Failure.” JAMA Internal Medicine, 2020.
- Lu, Jianyun, et al. “COVID-19 Outbreak Associated With Air Conditioning in Restaurant, Guangzhou, China, 2020.” Emerging Infectious Diseases, vol. 26, no. 7, 2020.
- Castillo RC, Staguhn ED, Weston-Farber E. The effect of state-level stay-at-home orders on COVID-19 infection rates [published online ahead of print, 2020 May 24]. Am J Infect Control. 2020;S0196-6553(20)30314-X. doi:10.1016/j.ajic.2020.05.017
- Michard, Frederic, et al. “COVID-19: Pulse Oximeters in the Spotlight.” Journal of Clinical Monitoring and Computing, 2020.
- Jouffroy, Romain, et al. “Prehospital Pulse Oximetry: a Red Flag for Early Detection of Silent Hypoxemia in COVID-19 Patients.” Critical Care (London, England), vol. 24, no. 1, 2020, p. 313.
- Annis, Tucker, et al. “Rapid Implementation of a COVID-19 Remote Patient Monitoring Program.” Journal of the American Medical Informatics Association : JAMIA, 2020.
- https://www.cebm.net/covid-19/question-should-smartphone-apps-be-used-as-oximeters-answer-no/
- Arshad et al “Treatment with Hydroxychloroquine, Azithromycin, and Combination in Patients Hospitalized with COVID-19” International Journal of Infectious Diseases. July 1, 2020
- Rajter et al “ICON (Ivermectin in COvid Nineteen) study: Use of Ivermectin is associated with Lower Mortality in Hospitalized Patients with COVID 19” medRxiv preprint doi: https://doi.org/10.1101/2020.06.06.20124461
- Manne, Bhanu Kanth, et al. “Platelet Gene Expression and Function in COVID-19 Patients.” Blood, 2020.
- Zaid Y et al “Platelets can contain SRS-CoV-2 RNA and are hyperactivated in COVID-19”
- http://www.jalali.one/farid/COVID19-Pathology.pdf?fbclid=IwAR2vlTmx5ev4QE9W4fhFt9YhoSdwdlY1UEltk_TZb3_UyONViX2szhTiJ7M
- Dewitte, A., Lepreux, S., Villeneuve, J. et al. Blood platelets and sepsis pathophysiology: A new therapeutic prospect in critical ill patients?. Ann. Intensive Care 7, 115 (2017). https://doi.org/10.1186/s13613-017-0337-7
- Middleton, Elizabeth A., et al. “Neutrophil Extracellular Traps (NETs) Contribute to Immunothrombosis in COVID-19 Acute Respiratory Distress Syndrome.” Blood, 2020.
- Oxley et al “Large-Vessel Stroke as a Presenting Feature of COVID-19 in the Young.” N Engl J Med 2020; 382:e60 DOI: 10.1056/NEJMc2009787
- Merkler, Alexander E., et al. “Risk of Ischemic Stroke in Patients With Coronavirus Disease 2019 (COVID-19) Vs Patients With Influenza.” JAMA Neurology, 2020.
- Yaghi S, Ishida K, Torres J, et al. SARS-CoV-2 and Stroke in a New York Healthcare System. Stroke. 2020;51(7):2002-2011. doi:10.1161/STROKEAHA.120.030335
- Long, Q., Tang, X., Shi, Q. et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat Med (2020). https://doi.org/10.1038/s41591-020-0965-6
- Morawska L, Milton DK. It is Time to Address Airborne Transmission of COVID-19 [published online ahead of print, 2020 Jul 6]. Clin Infect Dis. 2020;ciaa939. doi:10.1093/cid/ciaa939
- Pastorino B, Touret F, Gilles M, de Lamballerie X, Charrel RN. Prolonged Infectivity of SARS-CoV-2 in Fomites. Emerg Infect Dis. 2020;26(9):10.3201/eid2609.201788. doi:10.3201/eid2609.201788
- Fischer et al., Sci. Adv. 10.1126/sciadv.abd3083 (2020).
- MacIntyre CR, Seale H, Dung TC, et al. A cluster randomised trial of cloth masks compared with medical masks in healthcare workers. BMJ Open. 2015;5(4):e006577. Published 2015 Apr 22. doi:10.1136/bmjopen-2014-006577
- Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19 [published online ahead of print, 2020 Jul 9]. JAMA. 2020;324(6):603-605. doi:10.1001/jama.2020.12603
- Salmon Ceron D, Bartier S, Hautefort C, et al. Self-reported loss of smell without nasal obstruction to identify COVID-19. The multicenter Coranosmia cohort study [published online ahead of print, 2020 Jul 7]. J Infect. 2020;S0163-4453(20)30463-1. doi:10.1016/j.jinf.2020.07.005
- Ridgway JP, Pisano J, Landon E, Beavis KG, Robicsek A. Clinical Sensitivity of Severe Acute Respiratory Syndrome Coronavirus 2 Nucleic Acid Amplification Tests for Diagnosing Coronavirus Disease 2019. Open Forum Infect Dis. 2020;7(8):ofaa315. Published 2020 Jul 24. doi:10.1093/ofid/ofaa315
- Scholz, M.; Derwand, R.; Zelenko, V. COVID-19 Outpatients – Early Risk-Stratified Treatment with Zinc Plus Low Dose Hydroxychloroquine and Azithromycin: A Retrospective Case Series Study. Preprints 2020, 2020070025 (doi: 10.20944/preprints202007.0025.v1
- Mitjà O, Corbacho-Monné M, Ubals M, et al. Hydroxychloroquine for Early Treatment of Adults with Mild Covid-19: A Randomized-Controlled Trial [published online ahead of print, 2020 Jul 16]. Clin Infect Dis. 2020;ciaa1009. doi:10.1093/cid/ciaa1009
- Cavalcanti AB, Zampieri FG, Rosa RG, et al. Hydroxychloroquine with or without Azithromycin in Mild-to-Moderate Covid-19 [published online ahead of print, 2020 Jul 23]. N Engl J Med. 2020;NEJMoa2019014. doi:10.1056/NEJMoa2019014
- Horby and Landray et al. Effect of Hydroxychloroquine in Hospitalized Patients with COVID-19: Preliminary results from a multi-centre, randomized, controlled trial. medRxiv preprint doi: https://doi.org/10.1101/2020.07.15.20151852
- Skipper CP, Pastick KA, Engen NW, et al. Hydroxychloroquine in Nonhospitalized Adults With Early COVID-19: A Randomized Trial [published online ahead of print, 2020 Jul 16]. Ann Intern Med. 2020;M20-4207. doi:10.7326/M20-4207
- Boulware DR, Pullen MF, Bangdiwala AS, et al. A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19. N Engl J Med. 2020;383(6):517-525. doi:10.1056/NEJMoa2016638
- Zeng QL, Yu ZJ, Gou JJ, et al. Effect of Convalescent Plasma Therapy on Viral Shedding and Survival in Patients With Coronavirus Disease 2019. J Infect Dis. 2020;222(1):38-43. doi:10.1093/infdis/jiaa228
- Deftereos SG, Giannopoulos G, Vrachatis DA, et al. Effect of Colchicine vs Standard Care on Cardiac and Inflammatory Biomarkers and Clinical Outcomes in Patients Hospitalized With Coronavirus Disease 2019: The GRECCO-19 Randomized Clinical Trial. JAMA Netw Open. 2020;3(6):e2013136. Published 2020 Jun 1. doi:10.1001/jamanetworkopen.2020.13136
- Schiffer JT, Johnston C, Wald A, Corey L. An Early Test-and-Treat Strategy for Severe Acute Respiratory Syndrome Coronavirus 2. Open Forum Infect Dis. 2020;7(7):ofaa232. Published 2020 Jun 12. doi:10.1093/ofid/ofaa232
- Lee S, Kim T, Lee E, et al. Clinical Course and Molecular Viral Shedding Among Asymptomatic and Symptomatic Patients With SARS-CoV-2 Infection in a Community Treatment Center in the Republic of Korea [published online ahead of print, 2020 Aug 6]. JAMA Intern Med. 2020;e203862. doi:10.1001/jamainternmed.2020.3862
- Merzon E, Tworowski D, Gorohovski A, et al. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population-based study [published online ahead of print, 2020 Jul 23]. FEBS J. 2020;10.1111/febs.15495. doi:10.1111/febs.15495
- Loeb M, Dang AD, Thiem VD, et al. Effect of Vitamin D supplementation to reduce respiratory infections in children and adolescents in Vietnam: A randomized controlled trial. Influenza Other Respir Viruses. 2019;13(2):176-183. doi:10.1111/irv.12615
- Camargo CA, Sluyter J, Stewart AW, et al. Effect of Monthly High-Dose Vitamin D Supplementation on Acute Respiratory Infections in Older Adults: A Randomized Controlled Trial. Clin Infect Dis. 2020;71(2):311-317. doi:10.1093/cid/ciz801
- Rees JR, Hendricks K, Barry EL, et al. Vitamin D3 supplementation and upper respiratory tract infections in a randomized, controlled trial. Clin Infect Dis. 2013;57(10):1384-1392. doi:10.1093/cid/cit549
- Yonker LM, Neilan AM, Bartsch Y, et al. Pediatric SARS-CoV-2: Clinical Presentation, Infectivity, and Immune Responses [published online ahead of print, 2020 Aug 18]. J Pediatr. 2020;S0022-3476(20)31023-4. doi:10.1016/j.jpeds.2020.08.037
- Spinner CD, Gottlieb RL, Criner GJ, et al. Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial [published online ahead of print, 2020 Aug 21]. JAMA. 2020;10.1001/jama.2020.16349. doi:10.1001/jama.2020.16349
- Kox M, Waalders NJB, Kooistra EJ, Gerretsen J, Pickkers P. Cytokine Levels in Critically Ill Patients With COVID-19 and Other Conditions. JAMA. 2020 Sep 3:e2017052. doi: 10.1001/jama.2020.17052. Epub ahead of print. PMID: 32880615; PMCID: PMC7489366.
- Putman M, Chock YPE, Tam H, Kim AHJ, Sattui SE, Berenbaum F, Danila MI, Korsten P, Sanchez-Alvarez C, Sparks JA, Coates LC, Palmerlee C, Peirce A, Jayatilleke A, Johnson SR, Kilian A, Liew J, Prokop LJ, Murad MH, Grainger R, Wallace ZS, Duarte-García A; COVID-19 Global Rheumatology Alliance. Antirheumatic Disease Therapies for the Treatment of COVID-19: A Systematic Review and Meta-analysis. Arthritis Rheumatol. 2020 Aug 2:10.1002/art.41469. doi: 10.1002/art.41469. Epub ahead of print. PMID: 32741139; PMCID: PMC7435536.
- Liu STH, Lin HM, Baine I, Wajnberg A, Gumprecht JP, Rahman F, Rodriguez D, Tandon P, Bassily-Marcus A, Bander J, Sanky C, Dupper A, Zheng A, Nguyen FT, Amanat F, Stadlbauer D, Altman DR, Chen BK, Krammer F, Mendu DR, Firpo-Betancourt A, Levin MA, Bagiella E, Casadevall A, Cordon-Cardo C, Jhang JS, Arinsburg SA, Reich DL, Aberg JA, Bouvier NM. Convalescent plasma treatment of severe COVID-19: a propensity score-matched control study. Nat Med. 2020 Sep 15. doi: 10.1038/s41591-020-1088-9. Epub ahead of print. PMID: 32934372.
- Avendaññ-Sola et al. Convalescent Plasma for COVID-19: A multicenter, randomized clinical trial. medRxiv preprint doi: https://doi.org/10.1101/2020.08.26.20182444.this version posted September 29, 2020.
- Joyner et al. Effect of Convalescent Plasma on Mortality among Hospitalized patients with COVID-19: Initial Three-Month Experience. medRxiv preprint doi: https://doi.org/10.1101/2020.08.26.20182444.this version posted September 29, 2020.
- Jamal AJ, Mozafarihashjin M, Coomes E, Powis J, Li AX, Paterson A, Anceva-Sami S, Barati S, Crowl G, Faheem A, Farooqi L, Khan S, Prost K, Poutanen S, Taylor M, Yip L, Zhong XZ, McGeer AJ, Mubareka S; Toronto Invasive Bacterial Diseases Network COVID-19 Investigators. Sensitivity of nasopharyngeal swabs and saliva for the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020 Jun 25:ciaa848. doi: 10.1093/cid/ciaa848. Epub ahead of print. PMID: 32584972; PMCID: PMC7337630.
- Cheuk S, Wong Y, Tse H, Siu HK, Kwong TS, Chu MY, Yau FYS, Cheung IYY, Tse CWS, Poon KC, Cheung KC, Wu TC, Chan JWM, Cheuk W, Lung DC. Posterior oropharyngeal saliva for the detection of SARS-CoV-2. Clin Infect Dis. 2020 Jun 21:ciaa797. doi: 10.1093/cid/ciaa797. Epub ahead of print. PMID: 32562544; PMCID: PMC7337706.
- Gudbjartsson DF, Norddahl GL, Melsted P, Gunnarsdottir K, Holm H, Eythorsson E, Arnthorsson AO, Helgason D, Bjarnadottir K, Ingvarsson RF, Thorsteinsdottir B, Kristjansdottir S, Birgisdottir K, Kristinsdottir AM, Sigurdsson MI, Arnadottir GA, Ivarsdottir EV, Andresdottir M, Jonsson F, Agustsdottir AB, Berglund J, Eiriksdottir B, Fridriksdottir R, Gardarsdottir EE, Gottfredsson M, Gretarsdottir OS, Gudmundsdottir S, Gudmundsson KR, Gunnarsdottir TR, Gylfason A, Helgason A, Jensson BO, Jonasdottir A, Jonsson H, Kristjansson T, Kristinsson KG, Magnusdottir DN, Magnusson OT, Olafsdottir LB, Rognvaldsson S, le Roux L, Sigmundsdottir G, Sigurdsson A, Sveinbjornsson G, Sveinsdottir KE, Sveinsdottir M, Thorarensen EA, Thorbjornsson B, Thordardottir M, Saemundsdottir J, Kristjansson SH, Josefsdottir KS, Masson G, Georgsson G, Kristjansson M, Moller A, Palsson R, Gudnason T, Thorsteinsdottir U, Jonsdottir I, Sulem P, Stefansson K. Humoral Immune Response to SARS-CoV-2 in Iceland. N Engl J Med. 2020 Sep 1:NEJMoa2026116. doi: 10.1056/NEJMoa2026116. Epub ahead of print. PMID: 32871063; PMCID: PMC7494247.
- EMERGENCY USE AUTHORIZATION (EUA) SUMMARY COVID-19 RT-PCR TEST. (n.d.). Retrieved October 6, 2020, from https://www.fda.gov/media/136151/download
- World Health Organization. Antigen-detection in the diagnosis of SARS-CoV-2 infection using rapid immunoassays. Interim guidance. https://www.who.int/publications/i/item/antigen-detection-in-the-diagnosis-of-sars-cov-2infection-using-rapid-immunoassays (Accessed October 6, 2020)
- Dinnes J, Deeks JJ, Adriano A, Berhane S, Davenport C, Dittrich S, Emperador D, Takwoingi Y, Cunningham J, Beese S, Dretzke J, Ferrante di Ruffano L, Harris IM, Price MJ, Taylor-Phillips S, Hooft L, Leeflang MM, Spijker R, Van den Bruel A; Cochrane COVID-19 Diagnostic Test Accuracy Group. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst Rev. 2020 Aug 26;8:CD013705. doi: 10.1002/14651858.CD013705. PMID: 32845525.
- Entrenas Castillo M, Entrenas Costa LM, Vaquero Barrios JM, Alcalá Díaz JF, López Miranda J, Bouillon R, Quesada Gomez JM. “Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study”. J Steroid Biochem Mol Biol. 2020 Oct;203:105751. doi: 10.1016/j.jsbmb.2020.105751. Epub 2020 Aug 29. PMID: 32871238; PMCID: PMC7456194
- Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW, Li VW, Mentzer SJ, Jonigk D. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N Engl J Med. 2020 Jul 9;383(2):120-128. doi: 10.1056/NEJMoa2015432. Epub 2020 May 21. PMID: 32437596; PMCID: PMC7412750.
- Bundgaard H, Bundgaard JS, Raaschou-Pedersen DET, von Buchwald C, Todsen T, Norsk JB, Pries-Heje MM, Vissing CR, Nielsen PB, Winsløw UC, Fogh K, Hasselbalch R, Kristensen JH, Ringgaard A, Porsborg Andersen M, Goecke NB, Trebbien R, Skovgaard K, Benfield T, Ullum H, Torp-Pedersen C, Iversen K. Effectiveness of Adding a Mask Recommendation to Other Public Health Measures to Prevent SARS-CoV-2 Infection in Danish Mask Wearers : A Randomized Controlled Trial. Ann Intern Med. 2020 Nov 18:M20-6817. doi: 10.7326/M20-6817. Epub ahead of print. PMID: 33205991; PMCID: PMC7707213.
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED, Zerbini C, Bailey R, Swanson KA, Roychoudhury S, Koury K, Li P, Kalina WV, Cooper D, Frenck RW Jr, Hammitt LL, Türeci Ö, Nell H, Schaefer A, Ünal S, Tresnan DB, Mather S, Dormitzer PR, Şahin U, Jansen KU, Gruber WC; C4591001 Clinical Trial Group. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020 Dec 10. doi: 10.1056/NEJMoa2034577. Epub ahead of print. PMID: 33301246.
- Zhu FC, Li YH, Guan XH, Hou LH, Wang WJ, Li JX, Wu SP, Wang BS, Wang Z, Wang L, Jia SY, Jiang HD, Wang L, Jiang T, Hu Y, Gou JB, Xu SB, Xu JJ, Wang XW, Wang W, Chen W. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. Lancet. 2020 Jun 13;395(10240):1845-1854. doi: 10.1016/S0140-6736(20)31208-3. Epub 2020 May 22. PMID: 32450106; PMCID: PMC7255193.
- Buchbinder SP, McElrath MJ, Dieffenbach C, Corey L. Use of adenovirus type-5 vectored vaccines: a cautionary tale. Lancet. 2020 Oct 31;396(10260):e68-e69. doi: 10.1016/S0140-6736(20)32156-5. Epub 2020 Oct 19. PMID: 33091364; PMCID: PMC7571904.
- WHO Solidarity Trial Consortium, Pan H, Peto R, Henao-Restrepo AM, Preziosi MP, Sathiyamoorthy V, Abdool Karim Q, Alejandria MM, Hernández García C, Kieny MP, Malekzadeh R, Murthy S, Reddy KS, Roses Periago M, Abi Hanna P, Ader F, Al-Bader AM, Alhasawi A, Allum E, Alotaibi A, Alvarez-Moreno CA, Appadoo S, Asiri A, Aukrust P, Barratt-Due A, Bellani S, Branca M, Cappel-Porter HBC, Cerrato N, Chow TS, Como N, Eustace J, García PJ, Godbole S, Gotuzzo E, Griskevicius L, Hamra R, Hassan M, Hassany M, Hutton D, Irmansyah I, Jancoriene L, Kirwan J, Kumar S, Lennon P, Lopardo G, Lydon P, Magrini N, Maguire T, Manevska S, Manuel O, McGinty S, Medina MT, Mesa Rubio ML, Miranda-Montoya MC, Nel J, Nunes EP, Perola M, Portolés A, Rasmin MR, Raza A, Rees H, Reges PPS, Rogers CA, Salami K, Salvadori MI, Sinani N, Sterne JAC, Stevanovikj M, Tacconelli E, Tikkinen KAO, Trelle S, Zaid H, Røttingen JA, Swaminathan S. Repurposed Antiviral Drugs for Covid-19 – Interim WHO Solidarity Trial Results. N Engl J Med. 2020 Dec 2:NEJMoa2023184. doi: 10.1056/NEJMoa2023184. Epub ahead of print. PMID: 33264556; PMCID: PMC7727327.
- Tomazini BM, Maia IS, Cavalcanti AB, et al. Effect of Dexamethasone on Days Alive and Ventilator-Free in Patients With Moderate or Severe Acute Respiratory Distress Syndrome and COVID-19: The CoDEX Randomized Clinical Trial. JAMA. 2020;324(13):1307–1316. doi:10.1001/jama.2020.17021
- The Writing Committee for the REMAP-CAP Investigators. Effect of Hydrocortisone on Mortality and Organ Support in Patients With Severe COVID-19: The REMAP-CAP COVID-19 Corticosteroid Domain Randomized Clinical Trial. JAMA. 2020;324(13):1317–1329. doi:10.1001/jama.2020.17022
- Dequin P, Heming N, Meziani F, et al. Effect of Hydrocortisone on 21-Day Mortality or Respiratory Support Among Critically Ill Patients With COVID-19: A Randomized Clinical Trial. JAMA. 2020;324(13):1298–1306. doi:10.1001/jama.2020.16761
- The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group. Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis. JAMA. 2020;324(13):1330–1341. doi:10.1001/jama.2020.17023
- Jones B, Brown-Augsburger P, Corbett K, et al. LY-CoV555, a rapidly isolated potent neutralizing antibody, provides protection in a non-human primate model of SARS-CoV-2 infection. bioRxiv. Published online October 1, 2020. doi:10.1101/2020.09.30.318972
- Chen P, Nirula A, Heller B, Gottlieb RL, Boscia J, Morris J, Huhn G, Cardona J, Mocherla B, Stosor V, Shawa I, Adams AC, Van Naarden J, Custer KL, Shen L, Durante M, Oakley G, Schade AE, Sabo J, Patel DR, Klekotka P, Skovronsky DM; BLAZE-1 Investigators. SARS-CoV-2 Neutralizing Antibody LY-CoV555 in Outpatients with Covid-19. N Engl J Med. 2020 Oct 28:NEJMoa2029849. doi: 10.1056/NEJMoa2029849. Epub ahead of print. PMID: 33113295; PMCID: PMC7646625.
- Weinreich DM, Sivapalasingam S, Norton T, Ali S, Gao H, Bhore R, Musser BJ, Soo Y, Rofail D, Im J, Perry C, Pan C, Hosain R, Mahmood A, Davis JD, Turner KC, Hooper AT, Hamilton JD, Baum A, Kyratsous CA, Kim Y, Cook A, Kampman W, Kohli A, Sachdeva Y, Graber X, Kowal B, DiCioccio T, Stahl N, Lipsich L, Braunstein N, Herman G, Yancopoulos GD; Trial Investigators. REGN-COV2, a Neutralizing Antibody Cocktail, in Outpatients with Covid-19. N Engl J Med. 2020 Dec 17. doi: 10.1056/NEJMoa2035002. Epub ahead of print. PMID: 33332778.
- Kalil AC, Patterson TF, Mehta AK, Tomashek KM, Wolfe CR, Ghazaryan V, Marconi VC, Ruiz-Palacios GM, Hsieh L, Kline S, Tapson V, Iovine NM, Jain MK, Sweeney DA, El Sahly HM, Branche AR, Regalado Pineda J, Lye DC, Sandkovsky U, Luetkemeyer AF, Cohen SH, Finberg RW, Jackson PEH, Taiwo B, Paules CI, Arguinchona H, Goepfert P, Ahuja N, Frank M, Oh MD, Kim ES, Tan SY, Mularski RA, Nielsen H, Ponce PO, Taylor BS, Larson L, Rouphael NG, Saklawi Y, Cantos VD, Ko ER, Engemann JJ, Amin AN, Watanabe M, Billings J, Elie MC, Davey RT, Burgess TH, Ferreira J, Green M, Makowski M, Cardoso A, de Bono S, Bonnett T, Proschan M, Deye GA, Dempsey W, Nayak SU, Dodd LE, Beigel JH. Baricitinib plus Remdesivir for Hospitalized Adults with Covid-19. N Engl J Med. 2020 Dec 11. doi: 10.1056/NEJMoa2031994. Epub ahead of print. PMID: 33306283.
- National SARS-CoV-2 Serology Assay Evaluation Group. Performance characteristics of five immunoassays for SARS-CoV-2: a head-to-head benchmark comparison. Lancet Infect Dis. 2020 Dec;20(12):1390-1400. doi: 10.1016/S1473-3099(20)30634-4. Epub 2020 Sep 23. Erratum in: Lancet Infect Dis. 2020 Dec;20(12):e298. PMID: 32979318; PMCID: PMC7511171.
- Simonovich VA, Burgos Pratx LD, Scibona P, Beruto MV, Vallone MG, Vázquez C, Savoy N, Giunta DH, Pérez LG, Sánchez MDL, Gamarnik AV, Ojeda DS, Santoro DM, Camino PJ, Antelo S, Rainero K, Vidiella GP, Miyazaki EA, Cornistein W, Trabadelo OA, Ross FM, Spotti M, Funtowicz G, Scordo WE, Losso MH, Ferniot I, Pardo PE, Rodriguez E, Rucci P, Pasquali J, Fuentes NA, Esperatti M, Speroni GA, Nannini EC, Matteaccio A, Michelangelo HG, Follmann D, Lane HC, Belloso WH; PlasmAr Study Group. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. N Engl J Med. 2020 Nov 24:NEJMoa2031304. doi: 10.1056/NEJMoa2031304. Epub ahead of print. PMID: 33232588; PMCID: PMC7722692.
- Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial). BMJ. 2020 Nov 3;371:m4232. doi: 10.1136/bmj.m4232. Erratum for: BMJ. 2020 Oct 22;371:m3939. PMID: 33144278; PMCID: PMC7607867.
- White D, MacDonald S, Bull T, Hayman M, de Monteverde-Robb R, Sapsford D, Lavinio A, Varley J, Johnston A, Besser M, Thomas W. Heparin resistance in COVID-19 patients in the intensive care unit. J Thromb Thrombolysis. 2020 Aug;50(2):287-291. doi: 10.1007/s11239-020-02145-0. Erratum in: J Thromb Thrombolysis. 2020 Jun 22;: PMID: 32445064; PMCID: PMC7242778.
- Beun R, Kusadasi N, Sikma M, Westerink J, Huisman A. Thromboembolic events and apparent heparin resistance in patients infected with SARS-CoV-2. Int J Lab Hematol. 2020;42 Suppl 1:19-20. doi:10.1111/ijlh.13230
- Dutt T, Simcox D, Downey C, McLenaghan D, King C, Gautam M, Lane S, Burhan H. Thromboprophylaxis in COVID-19: Anti-FXa-the Missing Factor? Am J Respir Crit Care Med. 2020 Aug 1;202(3):455-457. doi: 10.1164/rccm.202005-1654LE. PMID: 32510975; PMCID: PMC7397804.
- Trunfio M, Salvador E, Cabodi D, Marinaro L, Alcantarini C, Gaviraghi A, Trentalange A, Lipani F, Sciascia S, Roccatello D, Bonora S, Di Perri G, Calcagno A; e-COVID Study group. Anti-Xa monitoring improves low-molecular-weight heparin effectiveness in patients with SARS-CoV-2 infection. Thromb Res. 2020 Dec;196:432-434. doi: 10.1016/j.thromres.2020.09.039. Epub 2020 Oct 8. PMID: 33049598; PMCID: PMC7543686.
- Stattin K, Lipcsey M, Andersson H, Pontén E, Bülow Anderberg S, Gradin A, Larsson A, Lubenow N, von Seth M, Rubertsson S, Hultström M, Frithiof R. Inadequate prophylactic effect of low-molecular weight heparin in critically ill COVID-19 patients. J Crit Care. 2020 Dec;60:249-252. doi: 10.1016/j.jcrc.2020.08.026. Epub 2020 Sep 3. PMID: 32920503; PMCID: PMC7470731.
- Lemos ACB, do Espírito Santo DA, Salvetti MC, Gilio RN, Agra LB, Pazin-Filho A, Miranda CH. Therapeutic versus prophylactic anticoagulation for severe COVID-19: A randomized phase II clinical trial (HESACOVID). Thromb Res. 2020 Dec;196:359-366. doi: 10.1016/j.thromres.2020.09.026. Epub 2020 Sep 21. PMID: 32977137; PMCID: PMC7503069.
- Flaczyk A, Rosovsky RP, Reed CT, Bankhead-Kendall BK, Bittner EA, Chang MG. Comparison of published guidelines for management of coagulopathy and thrombosis in critically ill patients with COVID 19: implications for clinical practice and future investigations. Crit Care. 2020 Sep 16;24(1):559. doi: 10.1186/s13054-020-03273-y. PMID: 32938471; PMCID: PMC7492793.
- Bikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, Nigoghossian C, Ageno W, Madjid M, Guo Y, Tang LV, Hu Y, Giri J, Cushman M, Quéré I, Dimakakos EP, Gibson CM, Lippi G, Favaloro EJ, Fareed J, Caprini JA, Tafur AJ, Burton JR, Francese DP, Wang EY, Falanga A, McLintock C, Hunt BJ, Spyropoulos AC, Barnes GD, Eikelboom JW, Weinberg I, Schulman S, Carrier M, Piazza G, Beckman JA, Steg PG, Stone GW, Rosenkranz S, Goldhaber SZ, Parikh SA, Monreal M, Krumholz HM, Konstantinides SV, Weitz JI, Lip GYH; Global COVID-19 Thrombosis Collaborative Group, Endorsed by the ISTH, NATF, ESVM, and the IUA, Supported by the ESC Working Group on Pulmonary Circulation and Right Ventricular Function. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020 Jun 16;75(23):2950-2973. doi: 10.1016/j.jacc.2020.04.031. Epub 2020 Apr 17. PMID: 32311448; PMCID: PMC7164881.
- Thachil J. The versatile heparin in COVID-19. J Thromb Haemost. 2020 May;18(5):1020-1022. doi: 10.1111/jth.14821. Epub 2020 Apr 27. PMID: 32239799.
- Zantah M, Dominguez Castillo E, Townsend R, Dikengil F, Criner GJ. Pneumothorax in COVID-19 disease- incidence and clinical characteristics. Respir Res. 2020 Sep 16;21(1):236. doi: 10.1186/s12931-020-01504-y. PMID: 32938445; PMCID: PMC7492794.
- Hameed M, Jamal W, Yousaf M, Thomas M, Haq IU, Ahmed S, Ahmad M, Khatib M. Pneumothorax In Covid-19 Pneumonia: A case series. Respir Med Case Rep. 2020;31:101265. doi: 10.1016/j.rmcr.2020.101265. Epub 2020 Oct 21. PMID: 33101895; PMCID: PMC7576439.
- Martinelli AW, Ingle T, Newman J, Nadeem I, Jackson K, Lane ND, Melhorn J, Davies HE, Rostron AJ, Adeni A, Conroy K, Woznitza N, Matson M, Brill SE, Murray J, Shah A, Naran R, Hare SS, Collas O, Bigham S, Spiro M, Huang MM, Iqbal B, Trenfield S, Ledot S, Desai S, Standing L, Babar J, Mahroof R, Smith I, Lee K, Tchrakian N, Uys S, Ricketts W, Patel ARC, Aujayeb A, Kokosi M, Wilkinson AJK, Marciniak SJ. COVID-19 and pneumothorax: a multicentre retrospective case series. Eur Respir J. 2020 Nov 19;56(5):2002697. doi: 10.1183/13993003.02697-2020. PMID: 32907891; PMCID: PMC7487269.
- do Lago VC, Cezare TJ, Fortaleza CMCB, Okoshi MP, Baldi BG, Tanni SE. Does COVID-19 Increase the Risk for Spontaneous Pneumothorax? Am J Med Sci. 2020 Dec;360(6):735-737. doi: 10.1016/j.amjms.2020.07.024. Epub 2020 Jul 16. PMID: 32819707; PMCID: PMC7365097.
Appendix 1: HCQ and Kaletra
Chloroquine/Hydroxychloroquine:
I have rewritten this section because of the hoopla that is going on in the media. If you would like to see the earlier version of this section, look at the Appendix at the end of the document.
There is a lot of controversy (for some reason) about the use of hydroxychloroquine and I think this stems mostly from how to interpret medical literature. The early studies that suggested some sort of benefit were cohort studies (mostly retrospective). These types of studies are at best, hypothesis generating rather than conclusive. I think the difference between RCT and cohort studies is crucial; when it comes to treatment, it becomes incredibly difficult to control for several variables in retrospective studies despite statistical wizardy (i.e who do you decide who ends up getting the treatment. While propensity score matching can control to a certain extent the variables that decide who gets treatment, this only accounts for those variable you actually measure). When it comes to RCTs, HCQ has unfortunately failed in the major studies. Having said that, one reasonable argument for many of these is the fact most of the therapy was initiated later in the course of the illness, when most antivirals are kind of useless (read: Oseltamavir and its younger, sexier brother Baloxivir).
Most of the early excitement came from early case series from France (56, 57). Another study came form Henry Ford (172), which demonstrated mortality benefit. This was a retrospective study (again, at best hypothesis generating) and a significant percentage of patients were getting steroids (you can take that as a steroid trial, but again, it is at best hypothesis generating). Recently, an outpatient study (190) evaluated the “Zalenko Protocol” of HCQ, azithromycin, and zinc in healthy outpatients also garnered some media attention, however the lack of knowledge of the control group (there are no details), the way these patients were allocated, and the lack of propensity score matching really puts into question how interpretable these results are. From now on, I’ll focus on the major RCTs rather than retrospective data (if you look hard enough, you’ll find a website that tracks all the studies that have been done on HCQ. Most of them are cohort studies).
HCQ – Mild to moderate
The weakest study (62) is an open-label multicenter RCT from China evaluating HCQ (1200mg daily x3 days followed by 800mg daily for a total of two to three weeks) and standard of care. Primary end point was negative conversion by day 28. The original target number of patients was 200 but given the decline of patients admitted with COVID, only 150 patients were enrolled. Patients were randomized a median of 16.6 days from symptom onset (SD 10.5, 3-41 days). There was no difference in primary outcome:
Secondary outcomes included alleviation of symptoms by day 28 (59.9% in HCQ group, 95% CI 45-75.3, and 66.6% without HCQ, 95% CI 39.5-90.9%) with a difference between groups being -6.6% (95% CI -41.3% to 28%). Issues stem from possible underpowering and lack of meaningful patient centered outcome, however HCQ failed to demonstrate any benefit in something I would have expected, namely viral clearance.
A recent multi-center, open label, RCT (191) from Spain enrolled 293 patients with mild to moderate COVID-19 (minimal O2 at best, usually <4L NC) to either standard of care (no placebo) or HCQ (800mg on day one and 400mg daily for 6 days). Notably, they tried to sneak in DRV/c but they stopped after they realized it had no in-vitro activity. Primary endpoint was reduction of viral RNA in NP swab at days 3 and 7; secondary outcome was clinical progression using the WHO progression scale. Notably, patients were enrolled within 3d of symptom onset (likely because the vast majority were healthcare workers). Overall, there were no differences in the primary outcome at days 3 or 7:
There was also no difference in secondary endpoints (RR 0.75, 95% CI 0.32 to 1.77), which risk of hospitalization. Further, there was no difference in the median time to resolution of COVID symptoms (12 days vs 10 days, p-value 0.38):
This was a better performed study (mostly able to get the med in a short time possible), but primary outcome again was not patient centered. The secondary outcome is intriguing, but at best this is hypothesis generating in the sense that it wasn’t powered for clinical improvement. Notably, most patients with HCQ had GI side effects (88% vs 3.8%) and no deaths related to adverse events were reported.
The largest multicenter, open label RCT (192) came from Brazil and it evaluated HCQ vs HCQ and azithro vs standard of care. HCQ was dosed 400mg BID for 7 days and Azithro was dosed 500mg daily for 7 days. Primary endpoint was clinical status at day 15 evaluated with a 7 point ordinal scale. They also analyzed the data as an ITT in those who had confirmed PCR diagnosis. 665 patients were enrolled and 504 were included in the mITT analysis. Patients were randomized within a median time of 7d from symptom onset. There was no difference between groups in outcomes on a 7 point ordinal scale:
There were also no differences between groups in secondary outcomes such as in-hospital mortality, duration of hospital stay, use of HFNC/non-invasive ventilation, and use of mechanical ventilation. There were also no significant adverse events in all groups, though the HCQ/Azithro group had more transaminitis.
Overall, data does not suggest the use of HCQ for mild to moderate COVID-19. There are some reasonable criticisms such as the difference in dosing (I can see someone using the Malaria dosing which involves loading 2g in the first 48hrs then 400mg daily), possible small effect that wasn’t picked up with these small numbers, or the time from symptom onset to therapy being to long for HCQ to do anything. Having said that, there is no evidence for the use of HCQ in this population at the moment.
HCQ – Severe Cases
While the HCQ arm of the RECOVERY trial had non-severe patients as well (193), this is the RCT that has the highest number of more severe cases (17% with invasive mechanical ventilation or ECMO and 60% with oxygen). This was an open label, multicenter RCT from the UK, evaluating several therapies. Patients were randomized in a 2:1 fashion to either standard of care or HCQ. Primary outcome was 28 day all cause mortality. 1561 patients were randomized to HCQ and 3155 were randomized to usual care. There was no difference in 28 day mortality between either group:
When limited to the patients with confirmed SARS-CoV2, the result was similar (RR 1.09, 95% CI 0.96 to 1.24). Those who were assigned to the HCQ group had a lower probability of being discharged alive within 28d (0.92, 95% CI 0.85 to 0.99), likely an anomaly.
So no real benefit to HCQ here either. We weren’t given the median time from symptoms to therapy, but this larger trial didn’t show any benefit either way. There was no subgroup analysis on severity, so I would expect that once the full trial comes out (this is a prelim result).
HCQ – Outpatient Data
Only one study I was able to find here. This was a multisite, international, double blind, placebo-controlled RCT (194) in non-hospitalized patients with confirmed diagnosis or high-risk exposure. People were randomized in a 1:1 fashion (HCQ dosed 800mg then 600mg 6-8hrs later, then 600mg daily for 4 additional days). Primary end point was change in symptom severity at day 14 (different than initial end point of hospital admissions, ICU admissions, and death). 491 patients were randomized, and at day 14 the HCQ had a mean reduction from baseline of 2.6 points on the 10-point visual scale compared to 2.33-point reduction in placebo group (absolute difference -0.27 points, 95% CI -0.61 to 0.07 p-value 0.117).
Notably in a post hoc analysis, those in HCQ group that had >75% adherence had significant improvement in their symptoms when compared to the placebo group (19.5% relative benefit) but this was not seen within the HCQ group when comparing it to those who had <75% adherence. While there is a trend towards HCQ being beneficial, this unfortunately did not reach statistical significance.
HCQ – Post exposure prophylaxis
A multicenter, double-blind, placebo controlled RCT (195) evaluated HCQ within 3 days after exposure (defined as high risk if there was no eye shield or mask and spending more than 10min with an infected person and moderate exposure if wearing a facemask). HCQ was dosed as 800mg loading dose followed by 600mg 6-8 hours later and 600mg daily for 4 days for a total of 5 days after exposure. Primary outcome was symptomatic illness confirmed by PCR or COVID-related symptoms. Notably, while they had calculated a sample size of 956 patients, the trial was halted on the third interim analysis. At day 14, there was no difference in the incidence of new COVID cases between groups (11.8% in HCQ group vs 14.3% in the placebo group, p-value 0.35, absolute difference -2.4 95% CI -7 to 2.2). Again a trend that did not reach statistical significance:
Overall, seems the current RCTs have not shown benefit for HCQ. It may be the effect is small, however the preprint for RECOVERY trial suggests that this may not be the case.
TL;DR – don’t count on hydroxychloroquine and wouldn’t recommend it outside an RCT at this point.
Lopinavir/Ritonavir aka Kaletra:
This is what I would consider a legacy drug for HIV. It is a combination of 2 protease inhibitors (which come with a bunch of baggage, given its interactions with everything under the sun), so I do not encounter this particular combination unless someone has been on it prior to the advent of integrase inhibitor combinations (aka Biktarvy).
– Early data: Kaletra, in combination with Ribavirin, was used for the original SARS
o 2004 open-label before/after study between historical controls and treatment group demonstrated a significant improvement in 21 day mortality, development of ARDS (2.4% vs 28% composite outcome), and viral load, however there were some inbalances between both groups (69).
o 2004 Retrospective study demonstrated significant mortality benefit to the Kaletra + Ribavirin combination (2.3 vs 16%) if given up front, but if given as salvage it had no improvement in outcomes (i.e. when patients were already intubated; 70).
– COVID-19 data
o The first study to come out was an open label study (71) that compared Kaletra vs standard of care. There was no difference in terms of time to clinical improvement, viral load, or other secondary outcomes. There may have been some improvement if the drug was started within 12 days, if you squint hard enough.
Of course, the average day before initiation of treatment was 13 days. Plus, there was no ribavirin, which is the combination that was tried in the original SARS as well as MERS.
o The latest study (72) actually tested Kaletra in combination with Ribavirin and Interferon beta vs control (IFN-beta was dropped if patients presented after 7d of illness). Primary outcome was time to negative PCR, which favored triple therapy. There was improvement in time to NEWS2 of 0 and SOFA score of 0, but these were secondary outcomes. These outcomes seem to have been driven by the early therapy group (i.e. those who were treated within 7d of illness and triple therapy).
Appendix II: Ivermectin and Colchicine
Ivermectin:
This one is a bit interesting, actually. IT has been found that, in vitro, Ivermectin has broad spectrum anti-viral activity against Dengue, West Nile, Eastern equine, Venezuelan Equine, HIV, yellow fever, tick borne encephalitis viruses as well as influenza (112), however in vivo activity has been reported only on WNV and Newcastle virus (113).
A recent study (112) showed that Ivermectin given to Vero-hSLAM cells 2 hours after infection resulted in a ~5000 fold reduction in SARS-CoV2 viral load. It is believed that the effect comes from inhibition of integrase protein of viruses and the importin (IMP) α/β1 heterodimer (114). Recently, the ICON trial (173) was put on pre-print, which is a retrospective cohort study from Florida evaluating the impact of Ivermectin (at least one dose) on in-hospital mortality in 280 patients (173 Ivermectin, 107 usual care group). They found mortality was lower in the Ivermectin group (15 vs 25.2%, p-value 0.03), as well as in the subgroup for those with severe disease (38.8% vs 80.7% p-value 0.001). Of note, only 13 patients were re-dosed on day 7.
Further, Ivermectin was found on multivariate analysis to be associated with lower mortality:
Having said that, there is no randomization and we are not told how the decision to administer Ivermectin was done. Plus, little matching between groups was done. It is unclear if it is ready for prime time; the subgroup analysis seem too good to be true.
TL;DR – be on the look out for more data on Ivermectin.
Colchicine
The idea here is to tone down the immune system in another pathway that is different from corticosteroids (this goes over my head, but essentially inhibits inflammasome signaling, reduces production of interleukin-1 beta, and reduces neutrophil-platelet interaction aka NETosis). There is some data to suggest its benefit. An open label RCT (197) evaluated colchicine (1.5mg loading then 0.5mg an hour later, dosed 0.5mg BID until discharge or 21d). Primary outcome was time from baseline to clinical deterioration, defined as a 2-grade increase in the WHO ordinal clinical scale. This occurred in 14% of patients in the control group vs 1.8% in the colchicine group (P-value 0.02). OR 0.11, 95% CI 0.01-0.96, p-value 0.046:
This is the only RCT to come out thus far and it was underpowered given the flattening of the curve in Greece. Physiologically, it make sense, but until a larger study comes out to replicate these results, I wouldn’t put too much weight on it. Not a lot of severe side effect seen, however.
Appendix III: Immunomodulators
Immunomodulators – Anakinra, Toculizumab
Please see the end of this document for a previous version of this section.
Immunomodulators were used in the beginning of the pandemic with the idea these drugs would be able to tone down the subsequent cytokine storm and improve mortality. A recent letter to the editor (206) suggest that cytokine levels in COVID-19 were lower when compared to those with septic shock or cardiac arrest. A recent review and meta-analysis (207) evaluated the use of anti-rheumatic drugs and their role in COVID-19. IL-6 inhibitors (tocilizumab, siltuximab) did not find significant benefits to their use in cohort studies (one study finding no difference, one finding lower mortality rate, and another one finding higher mortality rate).
In the meta-analysis, Anakinra was found to be associated with lower risk of mortality (pooled HR 0.2, CI 0.1-0.4) when compared to the standard of care.
IVIG was also evaluated, however while one study found evidence of survival, 2 other cohorts did not find difference in mortality. Overall, seems the data suggests some benefit to the use of Anakinra, but the data is sparse and not great.
Ver 1.
Immunomodulators
The idea here is to tone down the “cytokine storm” that seems to be responsible for the deterioration of patients at roughly the second week. There is evidence that this is playing a role in mortality when looking at the lab values of those who died:
Unfortunately, there is very little high quality data on the use of Tocilizumab.
In this prospective observational study (83), 21 patients were given Tocilizumab 4-8mg/kg up to 2 doses if persistently febrile. The primary endpoint was inflammatory markers, including reduction in CRP. There was a significant drop in CRP (from 75 to 2.72 in 5 days) as well as a drop in oxygen requirement in 75% of patients. All 21 patients were discharged after ~15 days. IL6 actually increased from 153 to 274.9.
In another retrospective observational study (84), 15 patients with moderately to critical COVID were given Tocilizumab and they saw a mean drop in CRP from 126.9 to 11.2. 3 patients who were given only one dose of Toci actually died, but those who got more than one dose did not die at the end of the study. Of note, the IL-6 levels here were all over the place, but one thing that did stand out is that those who did worse had a significant spike in IL-6
The highest quality data comes from a retrospective, observational, multicohort study that actually was done to study the effect of HCQ, but also examined the effect of tocilizumab in ICU patients (85). Doses were either 400mg or 800mg and only one dose was given in 96%.
Hopefully we will be able to gain some insight form the upcoming RECOVERY trial.
Sarilumab: monoclonal antibody against IL-6. This was used in New York, however this was as part of a trial. As of now, there is no data yet for this, though some news outlets have reported less than exciting results.
TL;DR exciting therapy for the cytokine storm, but no high quality data is out there yet. May be a good salvage therapy with doses of 400mg every other day (max 3 doses) until defervescence being a reasonable option. Need to monitor CRP and IL-6 (the latter if you can get it within a day), since these can be good prognosticators.
Anakinra: an IL-1 receptor antagonist that is used primarily for rheumatoid arthritis, but has also been used for septic shock with multiorgan dysfunction and HLH. There is some early data for its used in severe COVID.
– A retrospective cohort study compared anakinra with a historical control group (separated by about a week; 86). The dose used was 100mg bid for 3 days followed by 100mg daily for 7 additional days. Primary outcome was composite of death and ICU admission.
The CRP was also significantly lower in the anakinra group (~140 vs 20) when compared to the historical control.
– Another similarly designed retrospective cohort study with historical controls (87) actually studied 3 groups; standard of care (16 patients) , anakinra standard dose (100mg bid x7d; 7 patients), and high dose (5mg/kg daily until >75% reduction in CRP and >2d with P/F ratio >200); 29 patients. Here standard dose anakinra really didn’t do much for CRP, so the analysis was limited to high dose and standard of care. Here, the goal was to look at the change of clinical outcome at day 21:
TL;DR – unclear of what this means. May be similarly used to tocilizumab, as salvage therapy once someone is crashing form cytokine storm. Can likely use it if your prognostic labs (ie. CRP, ferritin) are going out of control before someone ends up on the vent.
Ruxolitinib:
This -nib (I cannot pronounce it this all the time) is a Janus kinase inhibitor:
Essentially, it tones down the inflammatory response. Unlike sarilumab or anakinra, which targets IL-6 and IL-1 respectively, this one targets more parts of the cytokine cascade. So far, only one study has been conducted in COVID-19. Ruxolitinib is approved for the treatment of polycythemia vera and myelofibrosis, and in the fashion of 2020 it is been refurbished to fight COVID-19. This (155) is a multicenter, RCT, phase II trial from China involving severe COVID-19 cases (defined as hypoxemia at room air and RR >30). Patients were randomized in a 1:1 fashion. In the end, 41 patients were included (21 in control and 20 in experimental group). Time to randomization was 20 days on average. In general, groups were well matched.
In general, there were no significant differences in both groups, though recovery trended towards the experimental group:
For cytokines, in general, the exeperimental group tended to fare better:
There were no changes in the viral load of either group, but the Ruxolitinib group tended to have higher levels of IgM antibodies.
Appendix IV: Lupus anticoagulant and Platelet activation:
There has been evidence arising on the role of the lupus anticoagulant to COVID-19; given the degree to which COVID causes coagulation issues, it shouldn’t be surprising. To elaborate further:
- Lupus anticoagulant is an antibody that makes elongates aPTT (aka makes you look like you’re about to bleed)
- While in the body, it actually causes you to clot
- “The” Lupus anticoagulant is actually a bunch of antibodies against “phospholipids” (since they’re everywhere), so they’re a bunch of them going around. Since the assay for measuring aPTT requires phospholipids, you can see how this can “erroneously” elongate the aPTT.
- The more common antibodies are those against cardiolipin, beta-2 glycoprotein.
- Having said that, the finding of lupus anticoagulants are not specific to COVID and can actually be seen in other viral infections (150).
For instance, one study out of France (151), evaluated 56 patients with dilute Russell Viper Venom and aPTT and found that 45% of those patients were positive for lupus anticoagulant, with 10% positive for both aCL or anti-beta-2 antibody. Another case series of six patients with large-vessel occlusive stroke who were also diagnosed with COVID found that 5/6 were positive for lupus anticoagulant, thought they did not measure aCL or beta-2 glycoprotein antibodies (152). In another series of cases involving cerebral infarcts (153), 3 patients were found to have prolonged aPTT as well as anticardiolipin antibodies. Finally, in a London study, 44 patients who had prolonged aPTT were evaluated and in 31 of 34 of those, they were found to have lupus anticoagulant:
Looking at the supplemental appendix (because they do not have the decency to put it in the correspondence letter..), the factor assays for those patients were as follows:
So Factor VIII is elevated. To flashback to the bad days of hematology:
So while low XII may not play a role, the elevated VIII may reflect the hypercoagulability of these patients. Having said that, what does this mean? IT may be that the coagulation issue is more complicated than once thought (i.e. ACE-2 receptor binding in endothelial tissue). While I think that, if there is no contraindication, full anticoagulation should be done if D-dimer is significantly elevated, keep an eye on the aPTT. If it is prolonged, it may be an indication of the presence of the lupus anticoagulant and if this is the case, I would advocate for full on anticoagulation. IF heparin is used, I would track anti-Xa activity level.
Platelet Activation:
Recent basic science data has come out, suggesting that SARS-CoV2 may have an effect on platelet activation. For instance, in a prospective study (174), 41 acutely COVID patients were compared to healthy donors. They found that platelets of COVID patients were hyper-reactive, had greater formation of platelet-leukocyte aggregates, and exhibited increased aggregation, adhesion, and spreading. This was found to be driven by increased release of thromboxane A1. Further data (175) suggests that platelets contribute to the overwhelming inflammation, with elevated cytokines being found as a result of platelet activation, including IL-1, IL-6, and TNF-alpha.
How does this play a role in coagulation? One savvy guy (176) went deep into the pathophysiology of clot progression and came up with a pretty nifty model:
- Binding of SARS-CoV2 to ACE-2 receptors leads to damaged endothelium, which in turns increases permeability into the alveoli
- The alveoli fills with platelets, which end up becoming active, and releasing granules with several things including serotonin
- Because serotonin is mostly stored in platelets and lungs clear the vast majority of serotonin, now theat lung s are taken out, there is more serotonin circling around
- Serotonin leads to more platelet aggregation and activation
– Platelets begin the process of “immune-thrombosis” to “wall off” a viral pathogen:
- Because of immune response to the organism (in this case, a virus), there is increased arrival of neutrophils and thus, NETosis:
- Essentially, neutrophils release their own matrix of “stuff” that is littered with pathogen killing goodies. This matrix (made up of DNA, histone, MPO, and oh, look at that, TNF, IFN, and IL-6). They literally are a trap!
- Indeed, one study (178) suggested there is elevated levels of MPO-DNA complexes, suggesting the presence of NETs
- Because of the lack of clearance of serotonin due to lung damage, there is more serotonin release from platelets and more aggregation and… you get the picture. This is a feed-forward mechanism for more thrombosis.
This means there is probably more going on than just “APLS” or “sepsis induced hypecoagulation.”