I think I have talked quite a bit about tuberculosis these past few posts. This is because it is difficult to diagnose in certain cases, it can infect virtually any organ, and it can become deadly once it infects any other organ outside of the lungs. Namely the CNS. Tuberculous meningitis is a deadly disease, and if it is not diagnosed early, it can be game over.
There are several issues with CNS tuberculosis. One of these is diagnosis. As mentioned previously, extrapulmonary tuberculosis tends to be a pauci-bacillary disease so staining can be low yield. Further, cultures take forever to grow. As such other modalities have been used. For instance, in a prospective, observational study from Kenya (1) evaluated 58 patients and looked at average glucose and protein value, as well as the utility of stains, cultures, and nucleic acid amplification. Patients with definitive tuberculosis were more likely to have higher CSF protein levels (to convert protein from g/L to mg/dL, multiply by 100) as well as lower glucose levels:
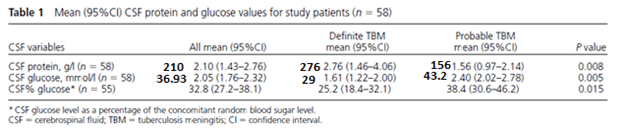
Roughly 12% of patients with definitive TB had 5g/L and about 50% had CSF glucose <25mmol:
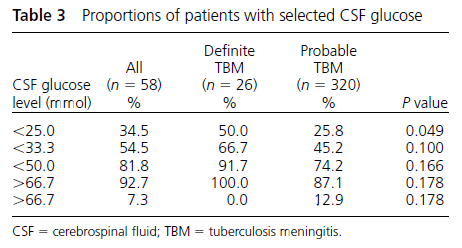
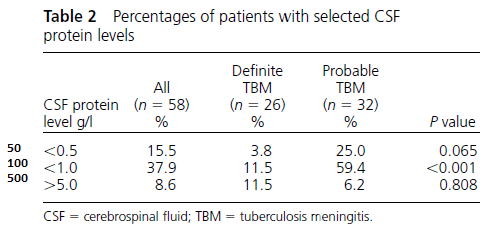
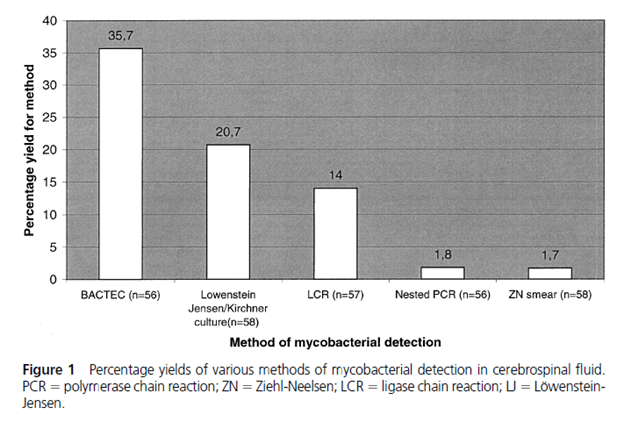
Cultures were able to detect around roughly 35% of cases, with the lowest yield being that of a Zeihl-Neelsen smear:
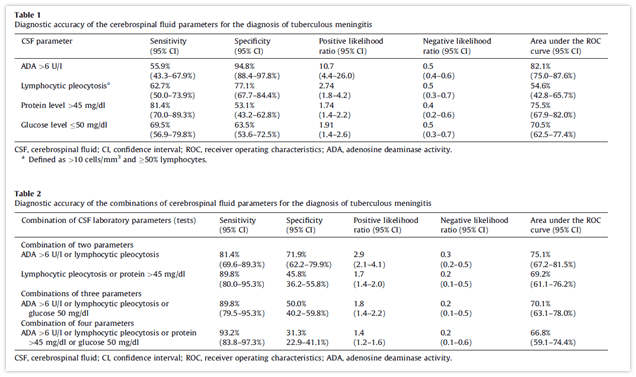
Two retrospective studies evaluated different cut-offs for different CSF parameters. In a retrospective study from Lima, Peru (2) evaluated 157 patients, of which 59 had tuberculous meningitis. The most sensitive was a CSF protein >45mg/dL. Notably, the combination of 2 parameters yielded a minimum sensitivity of 81.4% and combining all 4 parameters yielded a sensitivity of 93.2%, though this decreased specificity significantly:
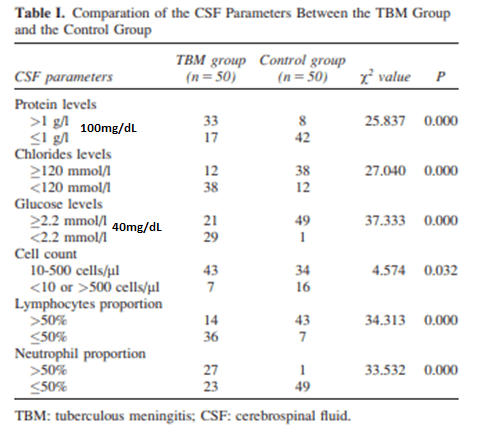
And a retrospective study (3) evaluated 6 different CSF parameters and compared 50 TB patients with 50 controls who had Japanese encephalitis as control. Overall, patients with tuberculous meningitis were more likely to have a protein >100mg/dL and glucose <40mg/dL when compared to the control group:
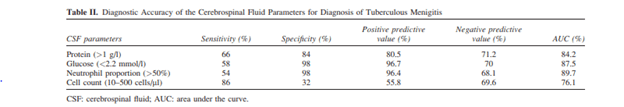
The most sensitive parameter was actually a cell count between 10-500, while the most specific were both neutrophils >50% and glucose <40mg/dL
Notably, using all 4 parameters resulted in no patients in the control group being identified as TBM:
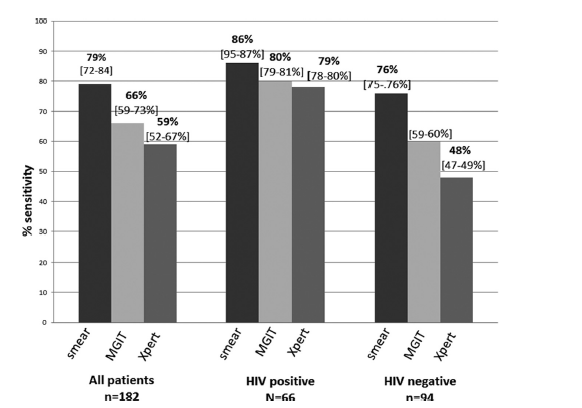
So the parameters in and of themselves are not terribly specific, but a combination of each, in context of the clinical picture, can yield a rapid diagnosis. This is important; the sensitivity of stain is not great and cultures will take a long time to return, so a rapid diagnosis with CSF parameters is paramount. What about NAAT and GeneXpert? As mentioned previously, the sensitivity varies depending on stain positivity. 2 retrospective studies evaluated the sensitivity of GeneXpert for this particular scenario. One study from Lima, Peru (4) evaluated 37 patients with presumed tuberculous meningitis against culture confirmation or clinical evidence. Of these 37 patients, 23/37 were HIV co-infected. For definite TBM, Gene Xpert had a sensitivity of 88% (95% CI 47-100%) compared to 75% for MGIT and 50% for LJ culture. When taken into consideration define, probable and possible TBM, the sensitivity dropped 23% (95% CI 9.9-42%). In a larger study from Vietnam (5) evaluated 379 patients, of whom 151 were classified as having definite tuberculous meningitis and 79/379 patients were co-infected with HIV. The overall sensitivity of GeneXpert was 59.3% (95% CI 51.8-66.5). Specificity was 99.5%. Smear, surprisingly, was high at 78.6%. Though one thing to consider was the fact that a small amount of patients were diagnosed based on clinical grounds (13 having possible TBM, and 18 having probable TBM). In other words, most of these patients had positive culture, so it is not surprising this is the case.
Steroids for TB meningitis
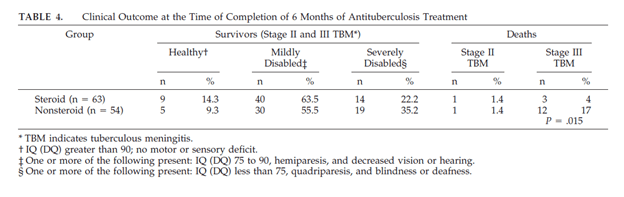
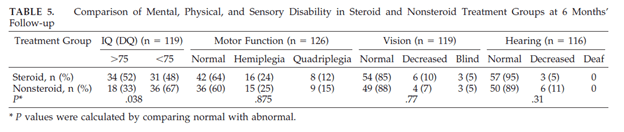
Early therapy is crucial for mortality. Besides RIPE, corticosteroids are a cornerstone of therapy in this disease with data suggesting mortality benefit. A 1997 RCT (6) in children used prednisone and measure ICP changes, brain parenchymal changes, mortality, and intelligence/morbidity. 70 patients were randomized to the steroid group (2mg/kg in the first 16 patients, which was changed to 4mg/kg later on due to rifampin interaction) and 71 in the non-steroid group. There were no differences in ICP change or degree of hydrocephalus, there was a difference in mortality as well as a higher IQ in the steroid group:
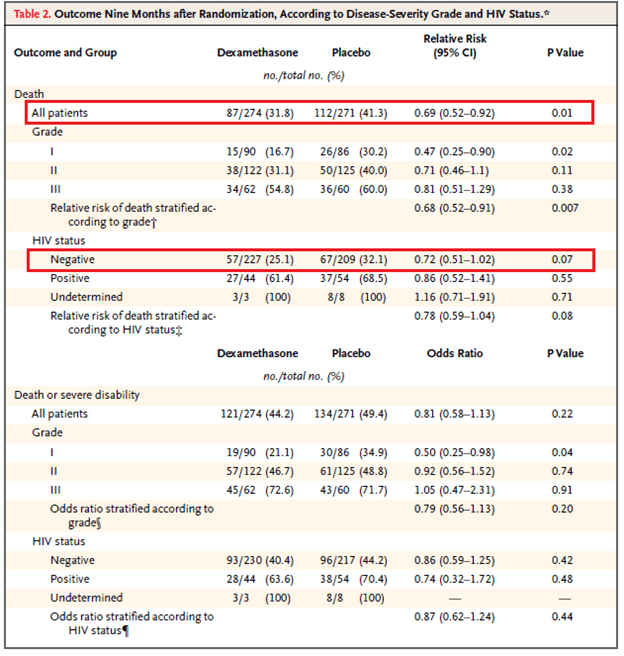
The largest and most well known RCT comes from Vietnam (7), which evaluated dexamethasone in 271 patients compared to 274 patients with a placebo. Patients were classified based on GCS (grade I = 15, grade II = 11 to 14, grade III = <10). Those with grade II and III disease received IV dexamethasone for four weeks (0.4mg/kg for week 1, 0.3mg/kg per day for week 2, 0.2mg/kg per day for week 3, and 0.1mg/kg per day for week 4) followed by a PO taper starting at 4mg per day, decreasing by 1mg each week. Those with grade I were given 2 weeks of IV dexamethasone followed by a 4 week oral taper. The primary outcome was death or severe disability at nine months after randomization. While there was no decrease in disability in the steroid group, dexamethasone was associated with reduced risk of death (RR 0.69, 95% CI 0.52 to 0.92, p-value 0.01), the effect of which was consistent among the different disabilities and HIV status.
CSF Trends
One of the recent cases I worked in involved a young man with tuberculous meningitis that was missed a few weeks before ultimately presented. He had an elevated ICP and had a shunt placed to drain CSF. As such, there were several studies that were sent evaluating CSF protein, glucose, and cell counts, basically every other day. Here is the pattern that emerged in the CSF. Due to the uncertainty of the diagnosis at the time, we started him on rifampin, INH, linezolid (for possible nocardia), PZA, and dexamethasone on 1/10:
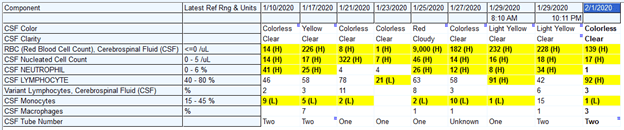


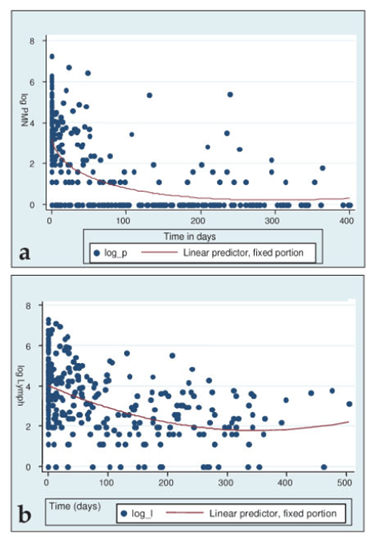
So why did his protein and cell count go back and forth after starting therapy? As it turns out, there is no clear pattern for the parameters after staring therapy. For instance, a retrospective study evaluated 99 patients with tuberculous meningitis (8) and 4 CSF parameters (PMN, glucose, lymphocytes, protein). While there was a statistically significant change overtime, the there was no clear decrease in the parameters, and in some cases patients had high protein even beyond 200 days:
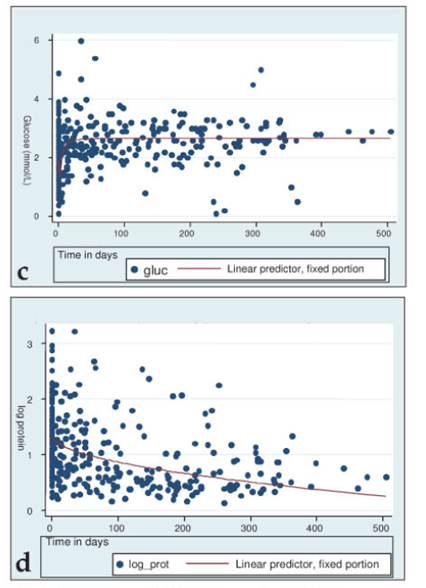
A larger retrospective study (9) evaluated CSF on admission, as well as week 1, 2, and 3 in pediatric patients under 13 years old. 318 patients were enrolled and of these, 91 had 4 CSF samples. Here, there was a significant decrease in parameters within week 2, especially for cell count, but CSF protein remained relatively constant:
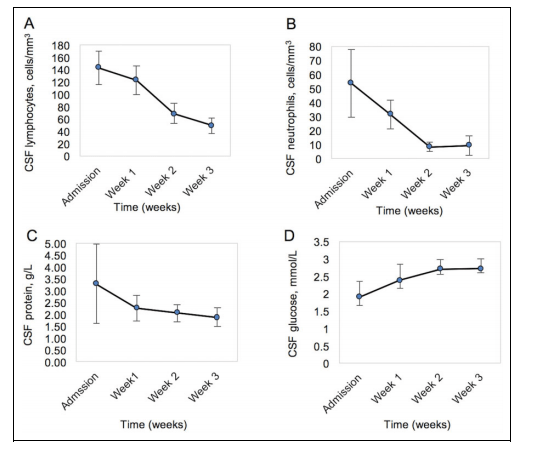
So while the trend is towards decrease protein and increase glucose, it is not surprising for the parameters to take a while to decrease even on therapy.
What about intrathecal therapy? Unfortunately, there is no good data to support the use here. Most of what I was able to find was from the 1950s and 1960s. There are some modern case reports of the use of intrathecal INH in refractory cases (10), including one in a pregnant patient who continued to have worsening neurological symptoms (aphasia, hemiplegia) despite high INH doses (11) as well as in those where patients did not tolerate INH due to liver dysfunction (12).
TL;DR
- Tuberculous meningitis is difficult to diagnose
- The utility of all CSF parameters (protein, glucose, cell count) increases the overall sensitivity
- Smear is not terribly helpful for diagnosis
- Since tuberculous meningitis is pauci-bacillary, the GeneXpert is not terribly useful (still get it, but if its negative, I wouldn’t put any weigh on it)
- Getting serial CSF studies may be all over the place even after starting therapy
- There is no data for intrathecal therapy
References:
- Hooker JA, Muhindi DW, Amayo EO, Mc’ligeyo SO, Bhatt KM, Odhiambo JA. Diagnostic utility of cerebrospinal fluid studies in patients with clinically suspected tuberculous meningitis. Int J Tuberc Lung Dis. 2003;7(8):787-796.
- Solari L, Soto A, Agapito JC, et al. The validity of cerebrospinal fluid parameters for the diagnosis of tuberculous meningitis. Int J Infect Dis. 2013;17(12):e1111-e1115. doi:10.1016/j.ijid.2013.06.003
- Zou Y, He J, Guo L, Bu H, Liu Y. Prediction of cerebrospinal fluid parameters for tuberculous meningitis. Diagn Cytopathol. 2015;43(9):701-704. doi:10.1002/dc.23284
- Metcalf T, Soria J, Montano SM, et al. Evaluation of the GeneXpert MTB/RIF in patients with presumptive tuberculous meningitis. PLoS One. 2018;13(6):e0198695. Published 2018 Jun 18. doi:10.1371/journal.pone.0198695
- Nhu NT, Heemskerk D, Thu do DA, et al. Evaluation of GeneXpert MTB/RIF for diagnosis of tuberculous meningitis. J Clin Microbiol. 2014;52(1):226-233. doi:10.1128/JCM.01834-13
- Schoeman JF, Van Zyl LE, Laubscher JA, Donald PR. Effect of corticosteroids on intracranial pressure, computed tomographic findings, and clinical outcome in young children with tuberculous meningitis. Pediatrics. 1997;99(2):226-231. doi:10.1542/peds.99.2.226
- Thwaites GE, Nguyen DB, Nguyen HD, et al. Dexamethasone for the treatment of tuberculous meningitis in adolescents and adults. N Engl J Med. 2004;351(17):1741-1751. doi:10.1056/NEJMoa040573
- Patel VB, Burger I, Connolly C. Temporal evolution of cerebrospinal fluid following initiation of treatment for tuberculous meningitis. S Afr Med J. 2008;98(8):610-613.
- Grobbelaar M, van Toorn R, Solomons R. Lumbar Cerebrospinal Fluid Evolution in Childhood Tuberculous Meningitis. J Child Neurol. 2018;33(11):700-707. doi:10.1177/0883073818785553
- Nakatani Y, Suto Y, Fukuma K, et al. Intrathecal Isoniazid for Refractory Tuberculous Meningitis with Cerebral Infarction. Intern Med. 2017;56(8):953–957. doi:10.2169/internalmedicine.56.6945
- Takahashi T, Ogawa K, Sawada S, et al. A case of refractory tuberculous meningitis markedly improved by intrathecal administration of isoniazid (INH). Rinsho Shinkeigaku 43: 20-25, 2003(in Japanese, Abstract in English).
- Danielides IC, Constantoulakis M, Daikos GK. Hepatitis on high dose isoniazid: reintroduction of the drug in severe tuberculous meningitis. Am J Gastroenterol 148: 650-655, 1983
