I think HIV is a very interesting virus. It has important historical, epidemiological, and societal implications and has had an impact in health care policy. The diagnosis of HIV now is actually relatively straight forward in most centers, but I wanted to take a step back and figure out the reasoning behind the algorithms that exist out there, as well as to review the mechanism of some of the tools used to diagnose it. Though if you want the quick run down, here it is
- We use what is called a “fourth generation antibody/antigen immunoassay.” In short terms, this uses recombinant HIV products to pick up either IgM or IgG OR antibodies against p24. Because it picks up IgM, you are able to pick up acute infection (i.e. when there is a bunch of virus running around but the immune system is barely making antibodies)
- Confirmatory testing is the differentiation immunoassay. It is similar to above, but uses different proteins for either HIV-1 or HIV-2
- We use 2 different tests to improve our positive predictive value (i.e. minimize the chances we get a false positive aka we know the positive test we got is a true positive)
- Western blots are a thing of the past
- Sensitivity and specificity in chronic disease is around 99% each. For acute infection ranges, but around 80% is what I have found.
- All that to say, if you see on EPIC that the HIV Ab/Ag is positive, wait for the differentiation assay before telling someone they’re positive. If its negative and you have a high suspicion, you may want to run an RNA (aka a viral load)
Basic HIV Structure:
I think it is important to go over the basic structure of the virus itself. At the end of the day, all the diagnostic tools we have for HIV attempts to figure out
- The presence of an HIV proteins
- The presence of antibodies against HIV
The simplified version of the HIV virus structure is as follows:
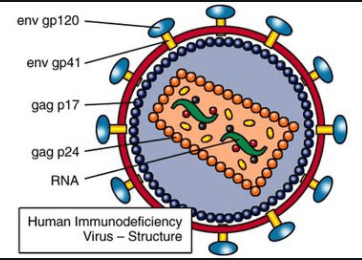
The p corresponds to the size of proteins. A more detailed structure is as follows:
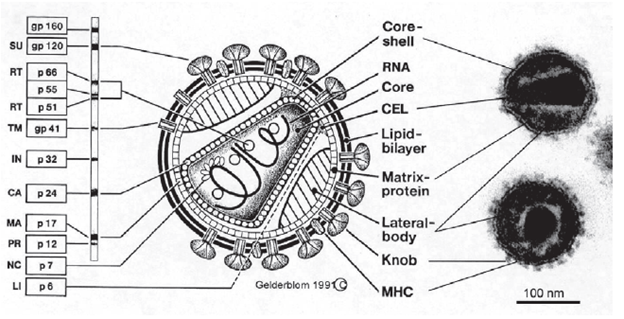
As you can see, we have several targets to pick from. There are 2 ways general testing modalities that are broadly used, immunoassays and western blotting.
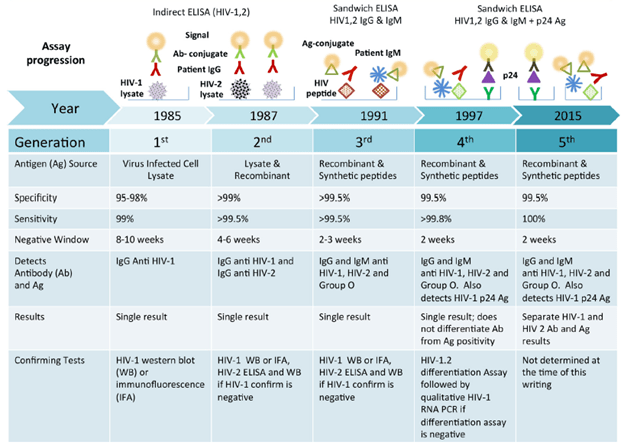
- Immunoassays – these come in 2 flavors, indirect and sandwich ELISA
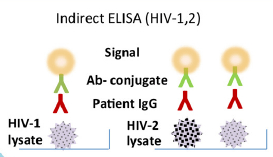
Here, HIV-lysate material is used as an antigen (i.e you culture HIV, grab it, lyse it and use that as an antigen for your test). A patient’s antibodies are added, which interact with the lysate. Afterwards, and antibody-conjugate interacts with the patient’s antibodies with a signal.
Usually, first and second generation use HIV lysates. This leads to false-positive results due to contamination of the antigen with proteins from cells used to culture the virus (1). As such, the sandwich ELISA was used in one of 2 ways: Antigen – patient antibody – conjugated antigen OR; antibody – patient antigen (p24) – conjugated antibody (2):

By using recombinant antigen in the assay, it reduces the number of false positives. It also allows for the use of patient’s IgM (which precedes IgG) that is also recognized by the recombinant antigen, meaning the sensitivity improves and you are able to diagnose the infection much earlier (1).
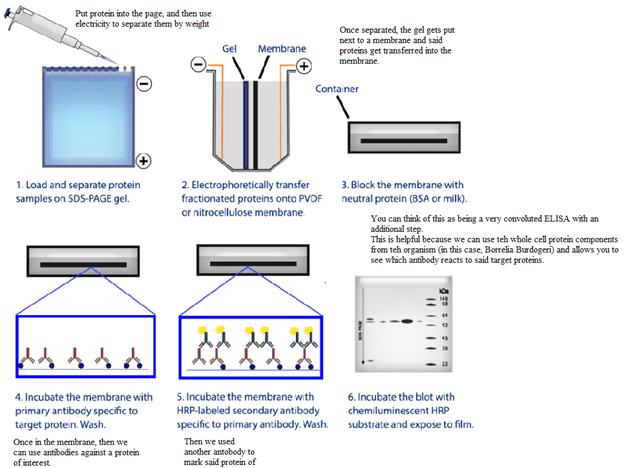
- Western Blot – this is used as a confirmatory assay if the initial immunoassay was positive (in the early days of HIV testing, this was employed to the concern for false positivity, as highlighted above). The virus particles are obtained from the patient, it is lysed, and put through electrophoresis to separate the proteins. Afterwards, the proteins are transferred into a nitrocellulose membrane and then antibodies are used to mark these proteins (increasing specificity). Then you have a number of bands (1)
- Nucleic acid amplification: this is not commonly used as part of the initial algorithm (see later)
- Viral culture – not really used either as it can take a long time (up to 6 weeks) for growth
Viral Kinetics
Why is this important? First of all, the antigens used for the assay plays a role in terms of when you will have a positive test. If you are attempting to detect IgG, remember that it takes a few weeks for IgG to be produced in the body. This has important implications.
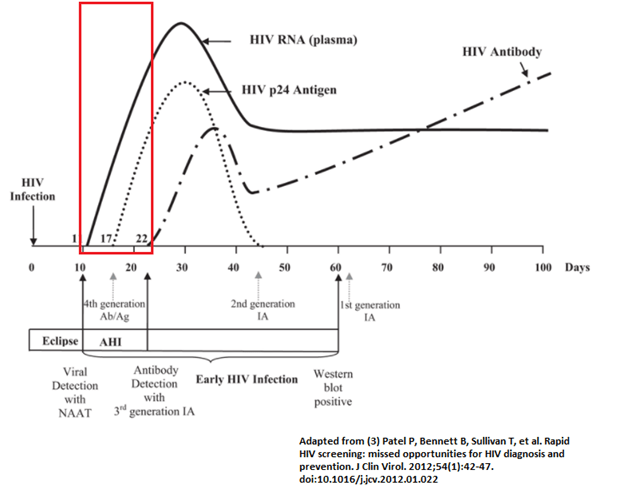
Because the initial available testing relied on antibodies, it could take up to 8 weeks for you to have a positive test (2). This is significant, because the highest viral load is usually seen within 2 weeks (4), and it can take up to 3-6 weeks before the humoral immune system kicks in to produce antibodies. This is important, because at this stage, HIV can be transmitted without the knowledge of an infection, and early testing with older assays can lead to a false positive. One analysis found the rate of transmission of HIV in primary infections is 26 times greater than during the asymptomatic period (5).
In the first few weeks, low titers of IgM and IgG are targeted against p24, gp120 and gp41. After about 9-12 weeks, one should have antibodies against many HIV proteins (4). One notable thing is there is a brief period of time (usually ~1-2 weeks) where p24 is circulating without antibodies being present. Because of this, the 4th generation immunoassay detects both HIV antibody and p24, decreasing the serologic window. A summary:
Why use multiple tests?
The simple answer is to improve the positive predictive value. HIV tests are highly specific, which means that a negative test correctly classifies an person as not having a disease (in this case, a negative HIV test means the patient does not have HIV). They are also highly sensitive, meaning those who have a positive test truly have the disease (6). However, this does not eliminate the possibility of false positives. Further, if you test a population that has low rates of HIV, you run the risk of more false positives than true positives (i.e. if you have a population where only 1 out of 1000 has HIV, and you have a test that is 99% sensitive, 9 patients will be labeled as false positive, while you end up catching the one patient). In other words, how you interpret testing is related to how prevalent the disease is in your population.
TL;DR – we use multiple test to make sure the initial positive was a true positive. While having a sensitive test is good, if you test this is a population without much disease to go around, you run the risk of getting more false positives than you anticipate:
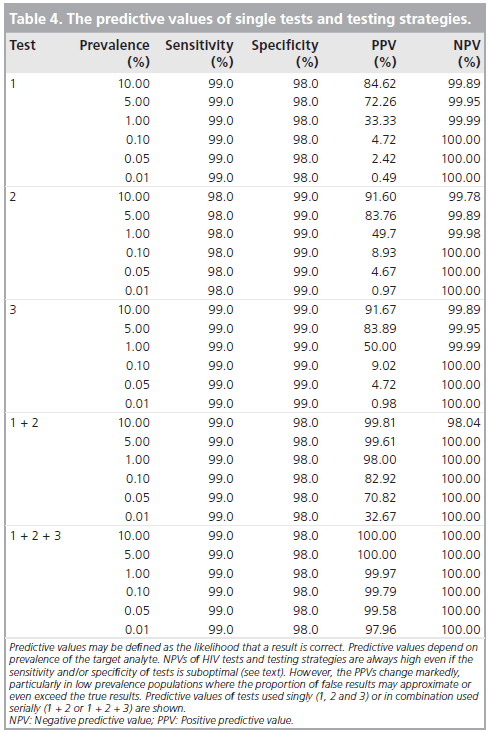
As you can see, despite the high sensitivity, if the prevalence is low, then the PPV. By adding additional test, we improve the PPV and thus, we can say that all patients who tested positive actually have the disease (i.e no false positivity).
Performance of HIV Testing – 3rd Generation
By in large, the sensitivity of 3rd generation immunoassays (we’ll get to Western Blot as confirmatory testing later) is actually quite good. This is, in part, due to the assays using recombinant or synthetic peptide antigens from HIV-1 and HIV-2 (generally p24, gp160, and gp36), as well as the ability to detect both IgM and IgG. An analysis of an HIV 1/2 Plus Assay (7) compared its sensitivity with 109 samples against a second generation standard. The 3rd generation (LG) assay was comparable in sensitivity and specificity to that of the prior generation (Behring):
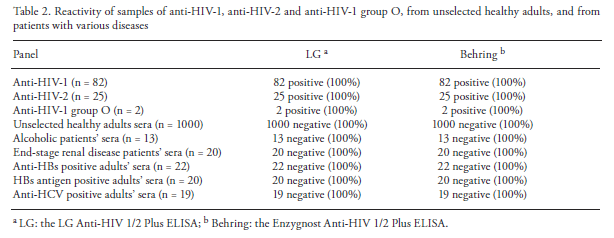
Though there was no difference between the 2 in the window period between infection and the first antibody response:

Another retrospective study (8) evaluated the third generation assay HIV-1/HIV-2 PLUS O EIA vs a first generation one and western blotting (amongst other assays). The third generation assay had a sensitivity of 100% and specificity of 100%. The authors also evaluated the performance of the third generation assay within the window period (positive RNA for HIV, negative antibodies) in 19 samples and found that the assay detected HIV antibody in 7/19 samples (37%), suggesting it has a higher sensitivity during the window period of infection.
In a large retrospective study (9), third generation immunoassay was used as part of an algorithm that also used a differentiation immunoassay (more on this later, but needless to say this resembles the current US guidelines for HIV testing), as its confirmatory test:
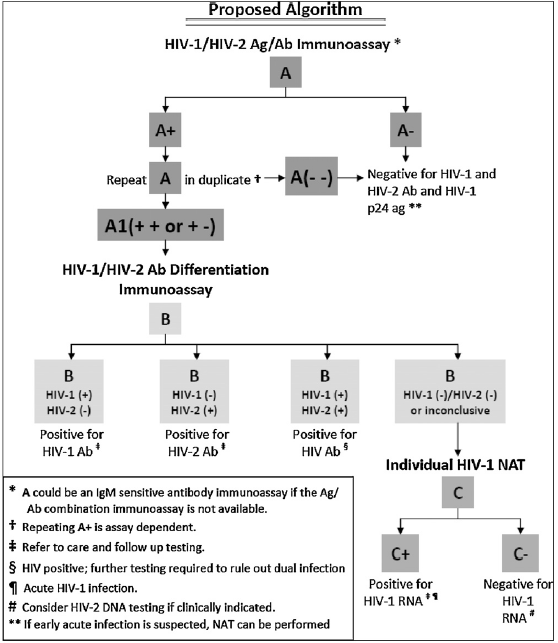
Notably, this algorithm actually used all FDA-approved third generation antibody/antigen test available at the time of the study. Using 2202 HIV-1 specimens and 1517 negative specimens, the sensitivity and specificity of the available third generation immunoassays are as follows:
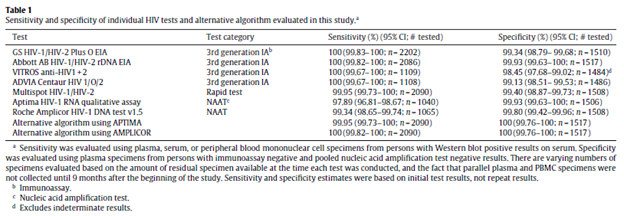
Notably, the multisport HIV-/HIV-2 assay also had a high sensitivity and specificity on its own. Notably, only a small proportion of patients needed a NAAT run due to indeterminate results (2/2059 or 0.1% for the Abbott IA, 11/2100 or 0.5% for the GS IA, 14/1097 or 1.3% for the ADVIA and 27/1110 or 2.4% for the VITROS), suggesting this algorithm performs well without the use of confirmatory western blot.
Western Blot
I’ll speak about Western Blot for a bit, however will not spend much time on it as in major hospitals this is not a confirmatory modality anymore, but nevertheless it is interesting. There are 12 antibodies against proteins that can be obtained in a Western Blot, and they are classified according to environmental (ENV) proteins, group specific antigen (GAG), and polymerase (POL):
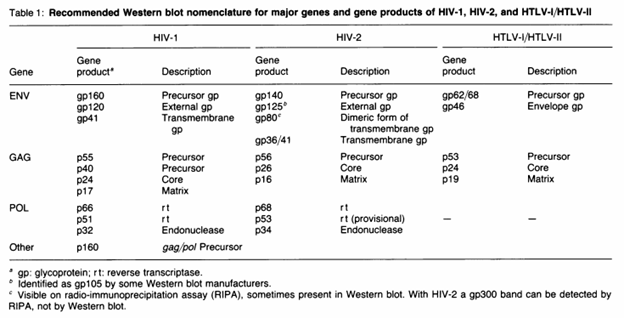
In general, gp160 (the precursor protein) and its subsequent final proteins, gp41 and gp120 are present in virtually all HIV-infected patients (10). Hence, positivity of 2 of these confirms the diagnosis. In contrast, GAG proteins tend to decrease or become undetectable as the disease progresses (10). As such, WHO guidelines require at least 2 of the ENV for positivity and plus/minus GAG/POL (11):

This was also confirmed in a CDC bulleting (11). Any other band positivity represents an “indeterminate” result, with all negative bands being a negative result. What is the issue here? Remember, Western Blot detect IgG against proteins, so it can take a while before it can turn positive (usually around 45-60 days, sometimes longer, 12). As such, in the first few days or weeks of infection, the confirmatory test will be either negative or indeterminate. At this point, the viral load is high and there is a high risk of transmission. In other words, we need to close the window of acute infection. Enter, fourth-generation immunoassays:

Fourth Generation Assay
I’ve already alluded to this algorithm (well, more like flat out put it out there), but here it is in full color:

Again, this assay uses both IgG and IgM, as well as attempting to identify p24 in order to catch early infection as soon as possible. Using 9150 samples (14) to compare GS HIV combo Ag/Ab (a 4th generation assay) with a 3rd generation assay, the overall sensitivity and specificity was comparable for both:
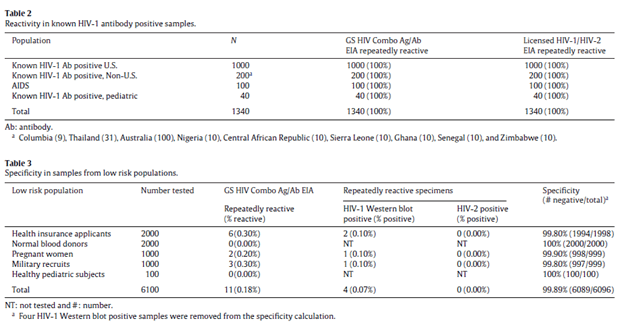
Notably, thirty patients with acute HIV infection (positive NAAT) were tested with both assays. The third generation confirmed 10% of those with acute HIV, compared to 33.33% for the 4th generation assay.
Another assay, the ARCHITECT HIV/Ag/Ab (15) was also compared using 4 different sources of sample sets for a total of 10,995 specimens (3384 of which were HIV positive). The overall sensitivity and specificity was ~99% for both, with this specific assay having a sensitivity of 82.7% for acute HIV infections.
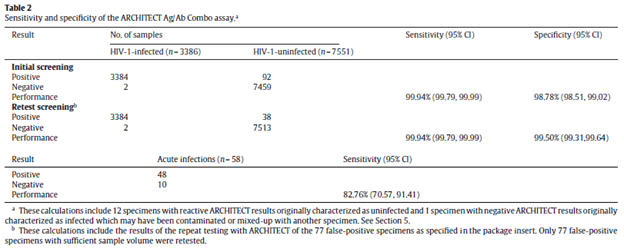
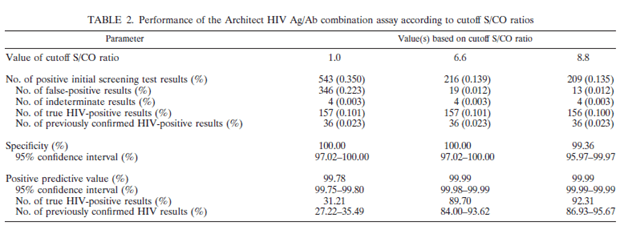
These tests also perform well in low incidence areas. For instance, in one retrospective study (16), 155,339 samples were tested with the ARCHITECT HIV Ag/Ab combo in South Korea. The overall sensitivity was roughly 99.36 and specificity was 100%. Using higher cutoff signal to cut-off ratio, the specificity was maintained, but the PPV increased significantly:
These sensitivities and specificities relate to chronic infections, which is not terribly surprising. In a large prospective study (17) evaluating the Architect HIV test, 86,836 patients were evaluated. They were first screened with a point of care test, and those who were negative were then screened with the fourth-generation test and confirmed with a pooled RNA test. Of 168 acute HIV infections diagnosed, the HIV Ag/Ab combination detected 134 acute infections, yielding sensitivity of 79.8% (95% CI 72.9-85.6), specificity of 99.9%, PPV of 59% and NPV of 96.5%. In other words, sensitivity decreases during the acute presentation, which as noted before is not surprising. The Determine Ab/Ag test also is able to detect HIV roughly 15.5 days before a western blot (20) with a fairly good sensitivity of about 92.5%.
Point of Care Testing:
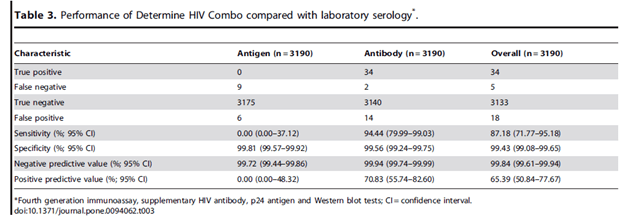
In general, these are able to provide rapid results, usually within 30min. Of these, 2 fourth generation tests are available, the ARCHITECT which is a lab-based test and the Determine HIV 1/2 Ag/Ab combo test, which is a lateral flow assay (essentially a pregnancy test). One prospective study (18) evaluated the Determine test in a high incidence setting (outpatient MSM and sexual health clinic) using the Architect immunoassay as a standard. 3195 test were sent out of 2468 men, of which 39 tested positive by reference test (12 early infection). This yielded a sensitivity for the Determine POC test of 87.2% (95% CI 71.8-95.2%) and sensitivity of 99.4%. Notably, the antibody component was the most sensitive and had the highest PPV:
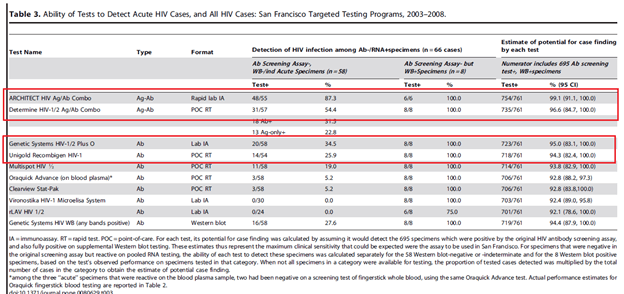
In a retrospective study (19) from San Francisco, patients were screened with a 1st generation immunoassay or a POC immunoassay prior to undergoing a follow up IFA. 21,234 patients were screened with several POC modalities, of which 58 had acute and 703 had established HIV infections. Overall, 1st generation and 3rd generation immunoassays had a 92% and 96% sensitivity, respectively. The Oraquick rapid test had a sensitivity of 86% on oral fluid samples, but 92% on fingerstick samples.

In terms of the acute case setting, the ARCHITECT Ag/Ab IA had a sensitivity of 87.4% while the Determine Ag/Ag rapid test had a sensitivity of 54.4%.
While not commonly used in certain parts of the US, rapid tests using saliva and fingerstick are also available. These are able to pick up IgG antibodies but the concentration tends to be about 800 times lower than in serum. In one retrospective study (21), 179 HIV positive patients and 60 HIV-negative patients were evaluated using 4 oral fluid based tests and one fingerstick one. The overall sensitivity of the oral tests ranged from 28% to up to 87.2%, when compared to the reported sensitivity of the fingerstick test:

Notably, the sensitivity seem to be impacted by both the CD4 and viral load:
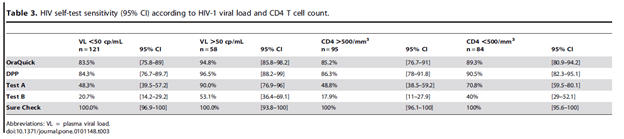
Another retrospective study of 287 newly infected HIV patients (22) evaluated factors associated with false negativity for the oral HIV tests and found that time to kit expiration, operator proficiency, randomization to PrEP, and in at least one site, low viral loads were associated with higher degree of false negativity:
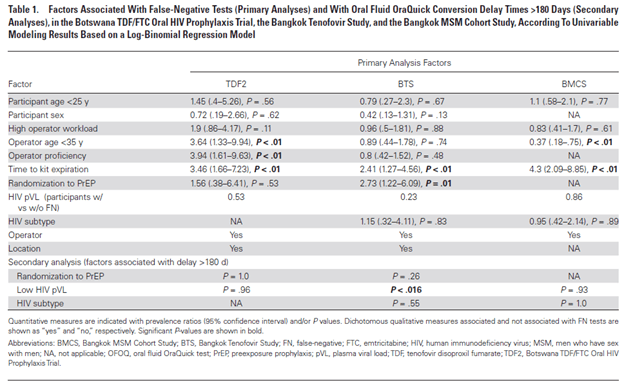
In relation to other tests, of the total 233 false negatives with the oral testing, 208 had a positive EIA. And of the 122 false negatives that had western blot data, 80% occurred after the appearance of the gp41 band while 95% came after the appearance of either a gp41 or gp160 band. So this may not be a terribly sensitive modality to screen someone in the acute setting, and would likely be better to use a fingerstick screen (though as mentioned previously, you would need to confirm with a follow up test).
Note: the title is a reference to a, well, not very good documentary. If you want a counter documentary, I recommend checking Myles Power’s YouTube channel: https://www.youtube.com/watch?v=0NnEijB1MHc&ab_channel=MylesPower
References:
- Chappel RJ, Wilson KM, Dax EM. Immunoassays for the diagnosis of HIV: meeting future needs by enhancing the quality of testing. Future Microbiol. 2009;4(8):963-982. doi:10.2217/fmb.09.77
- Alexander TS. Human Immunodeficiency Virus Diagnostic Testing: 30 Years of Evolution. Clin Vaccine Immunol. 2016;23(4):249-253. Published 2016 Apr 4. doi:10.1128/CVI.00053-16
- Patel P, Bennett B, Sullivan T, et al. Rapid HIV screening: missed opportunities for HIV diagnosis and prevention. J Clin Virol. 2012;54(1):42-47. doi:10.1016/j.jcv.2012.01.022
- German Advisory Committee Blood (Arbeitskreis Blut), Subgroup ‘Assessment of Pathogens Transmissible by Blood’. Human Immunodeficiency Virus (HIV). Transfus Med Hemother. 2016;43(3):203-222. doi:10.1159/000445852
- Hollingsworth TD, Anderson RM, Fraser C. HIV-1 transmission, by stage of infection. J Infect Dis. 2008;198(5):687-693. doi:10.1086/590501
- Parikh, Rajul et al. “Understanding and using sensitivity, specificity and predictive values.” Indian journal of ophthalmology vol. 56,1 (2008): 45-50. doi:10.4103/0301-4738.37595
- Yeom JS, Lee JB, Ryu SH, et al. Evaluation of a new third-generation ELISA for the detection of HIV infection. Ann Clin Lab Sci. 2006;36(1):73-78.
- Louie B, Pandori MW, Wong E, Klausner JD, Liska S. Use of an acute seroconversion panel to evaluate a third-generation enzyme-linked immunoassay for detection of human immunodeficiency virus-specific antibodies relative to multiple other assays. J Clin Microbiol. 2006;44(5):1856-1858. doi:10.1128/JCM.44.5.1856-1858.2006
- Wesolowski LG, Delaney KP, Hart C, et al. Performance of an alternative laboratory-based algorithm for diagnosis of HIV infection utilizing a third generation immunoassay, a rapid HIV-1/HIV-2 differentiation test and a DNA or RNA-based nucleic acid amplification test in persons with established HIV-1 infection and blood donors. J Clin Virol. 2011;52 Suppl 1:S45-S49. doi:10.1016/j.jcv.2011.09.026
- Centers for Disease Control (CDC). Interpretation and use of the western blot assay for serodiagnosis of human immunodeficiency virus type 1 infections. MMWR Suppl. 1989;38(7):1-7.
- AIDS: proposed WHO criteria for interpreting western blot assays for HIV-1, HIV-2, and HTLV-I/HTLV-II. (1991). Bulletin of the World Health Organization, 69(1), 127–133.
- Cohen MS, Gay CL, Busch MP, Hecht FM. The detection of acute HIV infection. J Infect Dis. 2010;202 Suppl 2:S270-S277. doi:10.1086/655651
- Branson BM, Stekler JD. Detection of acute HIV infection: we can’t close the window. J Infect Dis. 2012;205(4):521-524. doi:10.1093/infdis/jir793
- Bentsen C, McLaughlin L, Mitchell E, et al. Performance evaluation of the Bio-Rad Laboratories GS HIV Combo Ag/Ab EIA, a 4th generation HIV assay for the simultaneous detection of HIV p24 antigen and antibodies to HIV-1 (groups M and O) and HIV-2 in human serum or plasma. J Clin Virol. 2011;52 Suppl 1:S57-S61. doi:10.1016/j.jcv.2011.09.023
- Chavez P, Wesolowski L, Patel P, Delaney K, Owen SM. Evaluation of the performance of the Abbott ARCHITECT HIV Ag/Ab Combo Assay. J Clin Virol. 2011;52 Suppl 1:S51-S55. doi:10.1016/j.jcv.2011.09.010
- Kim S, Lee JH, Choi JY, Kim JM, Kim HS. False-positive rate of a “fourth-generation” HIV antigen/antibody combination assay in an area of low HIV prevalence. Clin Vaccine Immunol. 2010;17(10):1642-1644. doi:10.1128/CVI.00258-10
- Peters PJ, Westheimer E, Cohen S, et al. Screening Yield of HIV Antigen/Antibody Combination and Pooled HIV RNA Testing for Acute HIV Infection in a High-Prevalence Population. JAMA. 2016;315(7):682-690. doi:10.1001/jama.2016.0286
- Conway DP, Holt M, McNulty A, et al. Multi-centre evaluation of the Determine HIV Combo assay when used for point of care testing in a high risk clinic-based population [published correction appears in PLoS One. 2014;9(7):e103399]. PLoS One. 2014;9(4):e94062. Published 2014 Apr 8. doi:10.1371/journal.pone.0094062
- Pilcher CD, Louie B, Facente S, et al. Performance of rapid point-of-care and laboratory tests for acute and established HIV infection in San Francisco. PLoS One. 2013;8(12):e80629. Published 2013 Dec 12. doi:10.1371/journal.pone.0080629
- Masciotra S, Luo W, Youngpairoj AS, et al. Performance of the Alere Determine™ HIV-1/2 Ag/Ab Combo Rapid Test with specimens from HIV-1 seroconverters from the US and HIV-2 infected individuals from Ivory Coast. J Clin Virol. 2013;58 Suppl 1:e54-e58. doi:10.1016/j.jcv.2013.07.002
- Jaspard M, Le Moal G, Saberan-Roncato M, et al. Finger-stick whole blood HIV-1/-2 home-use tests are more sensitive than oral fluid-based in-home HIV tests. PLoS One. 2014;9(6):e101148. Published 2014 Jun 27. doi:10.1371/journal.pone.0101148
- Curlin ME, Gvetadze R, Leelawiwat W, et al. Analysis of False-Negative Human Immunodeficiency Virus Rapid Tests Performed on Oral Fluid in 3 International Clinical Research Studies. Clin Infect Dis. 2017;64(12):1663-1669. doi:10.1093/cid/cix228
