The timely diagnosis of invasive pulmonary aspergillosis is incredibly important. Non-invasive modalities of diagnosis such as beta-D glucan and galactomannan can take a few days to come back and much of the time, we rely on chest imaging to guide treatment decision. The classic finding that has been attributed to invasive pulmonary aspergillosis has been the halo sign, which is an area of consolidation surrounded by ground glass:
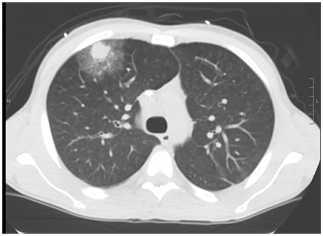
But how reliable is this sign? Is this enough to pull the trigger on empiric voriconazole or amphotericin B? Apart from signs and symptoms, if you were to see a halo-sign in a patient with hemoptysis, would that warrant empiric therapy? Here are the guidelines for invasive mould diagnosis. Keep in mind that definitive diagnosis is based on pathology showing angioinvasion to clinch the diagnosis:
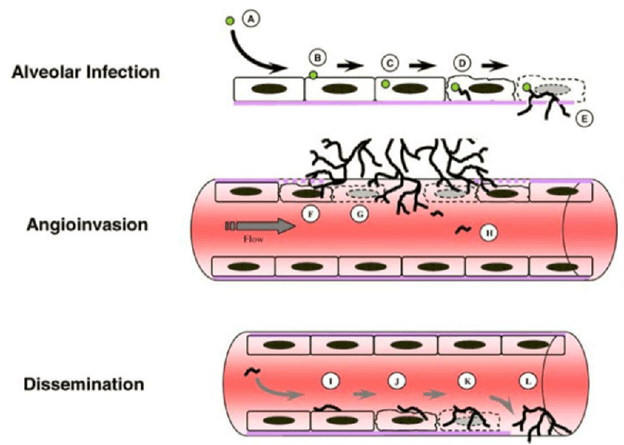
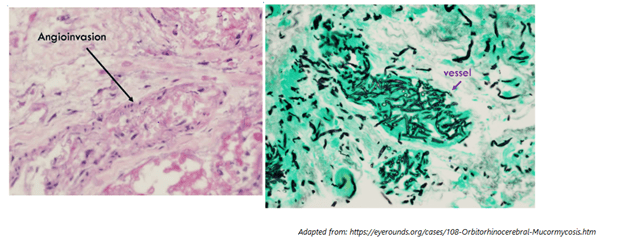
This is where we find ourselves most of the time, in the probable diagnosis. This means we will not get a biopsy but rather other methods to diagnose IFI, such as BD glucan, galactomannan, bronch, etc. Keep in mind that in order to apply this criteria, you need to have some sort of host factors:

Looking at the Host Factors, one thing warrants attention: most of the risk centers around the lack of neutrophils in relationship to malignancy. Even the data on steroid was based on its usage in leukemia patients (1). Having said that, it does not mean that other immunosuppressive states (ie the use of steroids for rheumatological disorders, AIDS, other types of immunomodulators) do not confer risk for invasive aspergillosis, but rather if you encounter a neutropenic patients on steroids with something going on in the lungs, that is an invasive mold until proven otherwise. Now on to the halo sign:
One of the first papers on the topic was on the description of CT findings in patients with invasive pulmonary aspergillosis (11). They looked at the CT chest findings of 9 patients with acute leukemia. Of these, only 2 had the classic “halo” sign:

They note the halo sign preceded the development of typical cavitation or air crescent by at least 2-3 weeks and typically the progression went from “halo” to cavitation. A small number of patients, but the takeaway was that invasive pulmonary aspergillosis could look like anything.
A review from 2007 (2) evaluated chest CT findings in a large series of patients with IPA from the Global Comparative Aspergillosis study. In this study, halo sign in allo-HSCT or heme patient with recent neutropenia was automatically considered probable IPA (see above). The goal of this study was to assess response to treatment and survival at 12 weeks. Of 235 patients, 86% of them had some sort of heme suppressive conditions, either leukemia or allogeneic HSCT. 14% had other conditions such as high dose steroids in 12, HIV in 11, SOT in 8, and solid tumor in 1. Most patients in this group had a micronodule, including those with the halo sign:
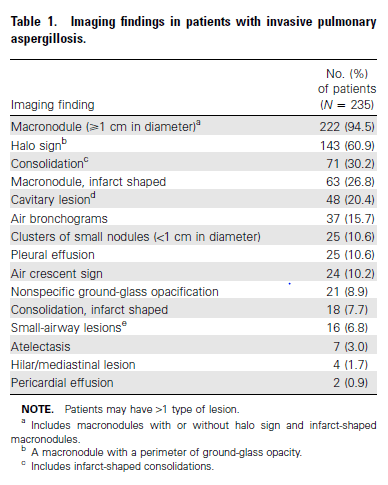
But a good percentage also had a consolidation, cavities, and air bronchograms. They assessed the response to treatment at 12 weeks in patients with halo sign and those without. At 12 weeks, 52% of patients with the halo sign had a satisfactory global response rate compared to 29% and this also translated to survival:
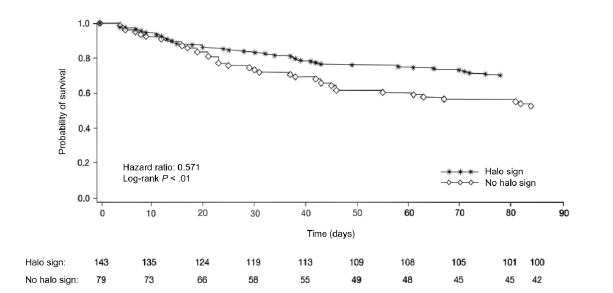
Indeed, seems that those patients with the halo sign had better outcomes and responded better to treatment overall:
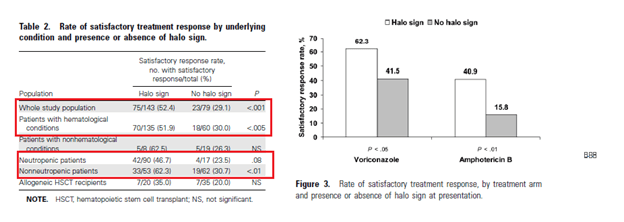
The authors do not mention the timing of therapy, which may play a role in terms of treatment response (i.e. those with halo sign were recognized as having IPA and thus started on therapy much sooner than other patients).
In a 2019 paper evaluating data from a major RCT comparing voriconazole with and without analdafungin for IPA treatment (3), a post-hoc analysis was done to evaluate the association between lesions and other findings on baseline CT and eventual diagnosis if IPA. While they go into details about the mortality differences between confirmed IPA and non-confirmed IPA, I want to focus on the CT findings. 395 patients were included and of these, 240 (60.8%) were classified as having confirmed IPA, the rest (155 or 39.2%) were non-confirmed. All patients had hematological malignancies.
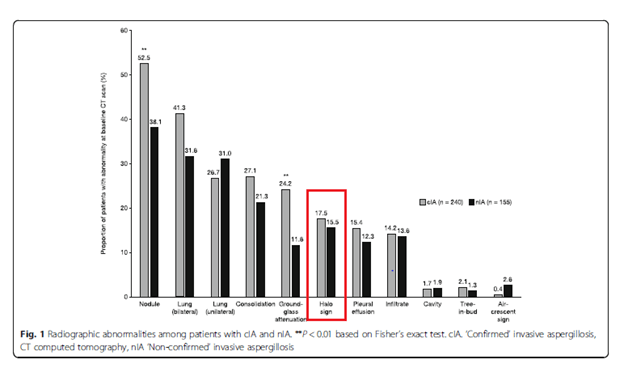
The percentage of patients with the halo sign in confirmed IPA was only 17.5% VS 52.5% for nodules.
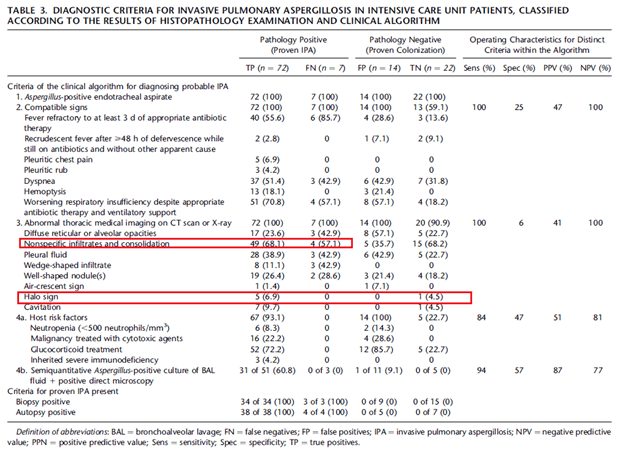
In those that are not immunosuppressed, the halo sign tends to fare worse in terms of sensitivity. In a multicenter observational study evaluating a clinical algorithm in the ICU setting for invasive pulmonary aspergillosis (4) patients with proven invasive pulmonary aspergillosis by biopsy were more likely to have non-specific infiltrates or consolidation than halo sign. Indeed, in this study, one patient with colonization actually had a halo sign on their CT:
Further data comes from an even older study from 1999 (5). This retrospective review evaluated chest CT of 305 patients with proven pulmonary disease. 16 of these patients were immunocompromised. 22 of these patients had a halo sign (7%), of which broncheoalveolar carcinoma made up 5 patients, tuberculoma 2, myxovirus, SCC, non-Hodgkin B cell lymphoma, and metastatic RCC made up one a piece. Those with multiple nodules were found to have KS and Wegners. Only one patient with a halo sign had invasive pulmonary aspergillosis, who also had leukemia. This suggest the lack of sensitivity overall for the halo sign in cases of IPA, and this drops for those without “classic” risk factors.
The most comprehensive study on the topic comes from Brazil (6). This was a multicenter retrospective study of CT chest images of 85 patients presenting to 7 tertiary centers with the goal of determining the diagnostic value of the CT halo sign in immunocompetent and immunocompromised patients (AIDS, congenital immunodeficiency, recent chemotherapy, radiation, neutropenia). In the immunocompetent group with the halo sign, the most common diagnosis was primary neoplasm, this contrasts with the immunocompromised, where aspergillosis was more common:
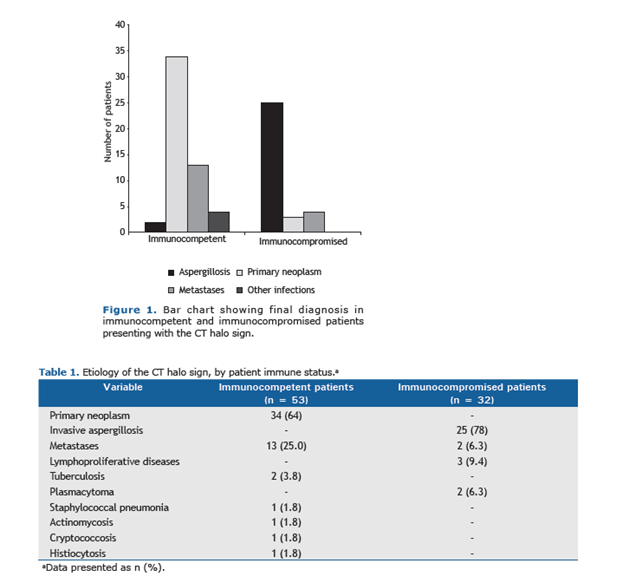
How much immunosuppression is enough?
This is a difficult question. Some immunosuppressed states, such as liquid malignancies and post-HSCT are easy enough. One of the key questions is regarding steroids. Most of the data re: steroids comes from the stem cell transplant and leukemia population (7). For instance, one study (8) in those patients with recent allogeneic HSCT patients found that higher doses of steroids was associated with higher incidence of invasive mold infections (with doses of 0.1-1.9mg/kg/d having a HR 2.2 (95% CI 1.3-3.8) and doses >3mg/kg/d having a HR 4.9 (95% CI 2.3-10.6). Another study found that use of steroid for GVHD prophylaxis had an OR 2.1 (95% CI 1.1-4.1) for invasive IFI (9). Further studies (10) have found a similar trend:
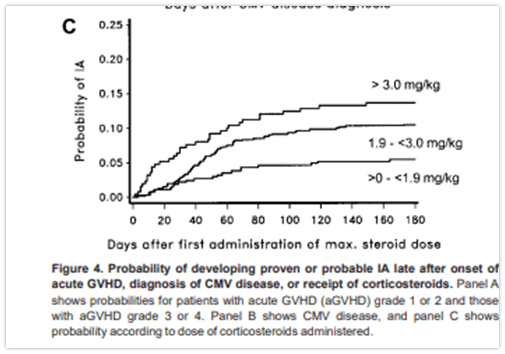
Most of the cases of steroids involved in IFI without underlying malignancy tend to be seen in case series. This does not mean they do not confer a risk, but probably not as much of a risk as some may think.
Bottom Line:
- Halo sign may not be as sensitive for invasive fungal infection, in general
- In immunosuppressed patients, literally anything else can look like an invasive fungal infection (consolidations, nodules, etc). If the patient has the right risk factors (i.e. prolonged neutropenia in setting of recent chemo), and you find ANYTHING on CT = treat for invasive mold until proven otherwise
- In those who do not have the classic risk factors and have a halo sign on CT, further diagnostics are needed. In this case, do not treat for a mold, even if you isolate it from a trach aspirate of sputum culture. Chances are, in that clinical scenario, the mold is a colonizer of the airway.
- Steroids are a tricky thing. Most of the data tends to be on hematology patients. But if there is other risk factors (i.e. prolonged steroid usage on a flu patient in the ICU), then that would be a situation to think hard about invasive mould.
References:
- Kuhlman JE, Fishman EK, Siegelman SS. Invasive pulmonary aspergillosis in acute leukemia: characteristic findings on CT, the CT halo sign, and the role of CT in early diagnosis. Radiology. 1985;157(3):611-614. doi:10.1148/radiology.157.3.3864189
- Greene RE, Schlamm HT, Oestmann JW, et al. Imaging findings in acute invasive pulmonary aspergillosis: clinical significance of the halo sign. Clin Infect Dis. 2007;44(3):373-379. doi:10.1086/509917
- Jin J, Wu D, Liu Y, et al. Utility of CT assessment in hematology patients with invasive aspergillosis: a post-hoc analysis of phase 3 data. BMC Infect Dis. 2019;19(1):471. Published 2019 May 28. doi:10.1186/s12879-019-4039-7
- Blot SI, Taccone FS, Van den Abeele AM, et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients [published correction appears in Am J Respir Crit Care Med. 2012 Oct 15;186(8):808]. Am J Respir Crit Care Med. 2012;186(1):56-64. doi:10.1164/rccm.201111-1978OC
- Gaeta M, Blandino A, Scribano E, Minutoli F, Volta S, Pandolfo I. Computed tomography halo sign in pulmonary nodules: frequency and diagnostic value. J Thorac Imaging. 1999;14(2):109-113. doi:10.1097/00005382-199904000-00008
- Alves GR, Marchiori E, Irion K, et al. The halo sign: HRCT findings in 85 patients. J Bras Pneumol. 2016;42(6):435-439. doi:10.1590/S1806-37562015000000029
- Lewis RE, Kontoyiannis DP. Invasive aspergillosis in glucocorticoid-treated patients. Med Mycol. 2009;47 Suppl 1:S271-S281. doi:10.1080/13693780802227159
- Garcia-Vidal, Carol et al. “Epidemiology of invasive mold infections in allogeneic stem cell transplant recipients: biological risk factors for infection according to time after transplantation.” Clinical infectious diseases : an official publication of the Infectious Diseases Society of America vol. 47,8 (2008): 1041-50. doi:10.1086/591969
- Martino R, Subirá M, Rovira M, et al. Invasive fungal infections after allogeneic peripheral blood stem cell transplantation: incidence and risk factors in 395 patients. Br J Haematol. 2002;116(2):475-482. doi:10.1046/j.1365-2141.2002.03259.x
- Grow WB, Moreb JS, Roque D, et al. Late onset of invasive aspergillus infection in bone marrow transplant patients at a university hospital. Bone Marrow Transplant. 2002;29(1):15-19. doi:10.1038/sj.bmt.1703332
- De Pauw, Ben et al. “Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group.” Clinical infectious diseases : an official publication of the Infectious Diseases Society of America vol. 46,12 (2008): 1813-21. doi:10.1086/588660
