So MRSA is really hard to kill. If you have done any sort of ID, you will not be surprised by that statement. MRSA bacteremia has a high relapse rate. Because of this, people have tried multiple types of antibiotics, including Daptomycin and Ceftaroline, have been used with minimal change in the outcomes. In the past few years, there has been a movement towards using dual therapy for MRSA bacteremia that includes a beta-lactam antibiotic. Ceftaroline, a beta-lactam antibiotic with MRSA activity, has been in the forefront for dual therapy in MRSA bacteremia. The punchline here, however, is that dual therapy may not be beneficial and may lead to harm. While there is in-vitro data suggesting a synergistic effect between anti-staph beta lactams and MRSA antibiotics such as vancomycin and daptomycin, I will focus mostly on the clinical data.
Davis JS, Sud A, O’Sullivan MVN, et al. Combination of Vancomycin and β-Lactam Therapy for Methicillin-Resistant Staphylococcus aureus Bacteremia: A Pilot Multicenter Randomized Controlled Trial. Clin Infect Dis. 2016;62(2):173-180. doi:10.1093/cid/civ808
This is a pilot, multicenter, open-label RCT from Australia evaluated the use of anti-staphylococcal B-lactam (in this case flucoxacillin) in combination with vancomycin for MRSA. Patients were randomized in a 1:1 fashion to receive vancomycin as standard of care or vancomycin + flucoxacillin 2g qid for 7d. Primary end point was duration of MRSA bacteremia; secondary endpoints included hepatotoxicity, rate of nephrotoxicity, all cause 28d and 90d mortality, relapsed bacteremia, metastatic complications within the first 10d and development of septic shock after randomization.
Nothing surprising in the characteristics of the patients, but seems some of the combination therapy group had nosocomial bacteremia. Everything else seems to balance out:
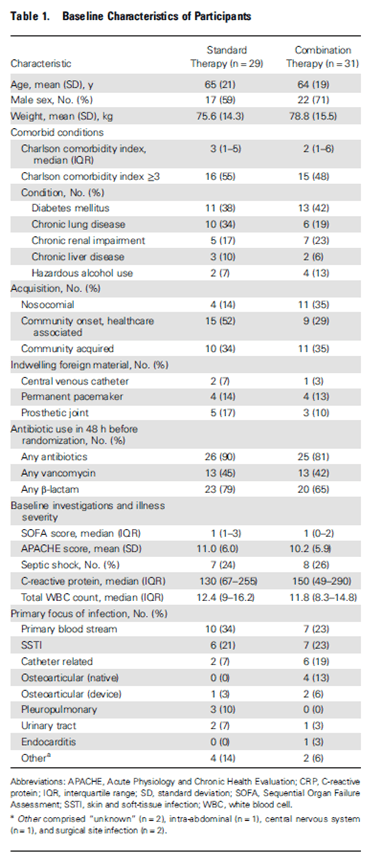
Lucky for us, they also gave us the MIC and the genotypes (PVL being the most important):
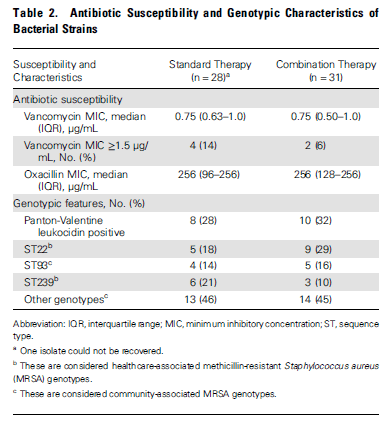
Overall there was a trend towards shorter duration of bacteremia in both the ITT group and the per protocol group (3 vs 1.94, p-value 0.06 in ITT, 2.92 vs 1.82 p-value 0.055 in Per protocol):

In terms of more patient centered outcomes, there was no significant difference in 28d or 90d mortality between groups:
Having said that, these were secondary outcomes and not primary outcomes, so this is more of a hypothesis generating study for the mortality question. Plus, the study was underpowered for this question anyways. While not significant, there was again a small trend towards nephrotoxicity in the combination group (foreshadowing).
Casapao AM, Jacobs DM, Bowers DR, Beyda ND, Dilworth TJ; REACH-ID Study Group. Early Administration of Adjuvant β-Lactam Therapy in Combination with Vancomycin among Patients with Methicillin-Resistant Staphylococcus aureus Bloodstream Infection: A Retrospective, Multicenter Analysis. Pharmacotherapy. 2017;37(11):1347-1356. doi:10.1002/phar.2034
This retrospective multicenter study evaluated the use of vancomycin in MRSA bacteremia with vancomycin and a beta lactam (ampicillin, nafcillin, oxacillin, pip-tazo, amp-sulbactam, CTX, cefoxitin, CTX, cefepime, imipenem, ertapenem, doripenem meropenem, anypenem) within 24hrs after starting vancomycin for at least 48hrs. Primary outcome was a composite of the following: 30d mortality, persistent bacteremia for >7d, bacteremia relapse (i.e. repeat blood culture positive after 48hrs of clearance), change of antibiotic due to lack of infection resolution. 97 patients were evaluated, 41 in the monotherapy group, 57 in the combination group. Overall, there were no significant differences between groups, though the combination group was younger (62yo vs 68 p-value 0.031). The most common beta lactam antibiotic given were pip-tazo (54.4%), a cephalosporin (33.3%), and carbapenem (12.3%). Most patients in the combination group received 5d of dual therapy.
Looking at the outcomes, there was no statistical difference in terms of composite outcome, though there was a trend towards rapidly clearance of bacteremia in the combination group.
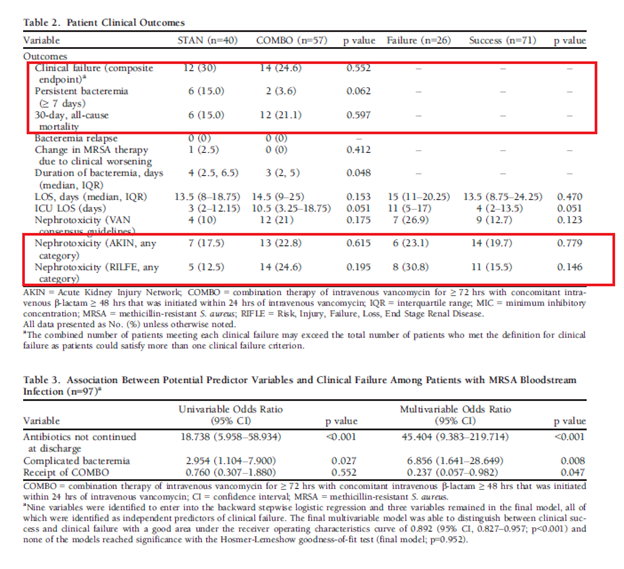
Nephrotoxicity was not terribly different either, but again, there was a trend towards more renal injury in the combination group. The higher mortality (at least the trend) in the combination group is a bit surprising, but again, this is a retrospective study where multiple things cannot be controlled. The other issue is they counted all patients with any beta-lactam antibiotic rather than those with anti-staphylococcal antibiotics (which tend to be better for staph anyways) such as nafcillin, flucloxacillin, or oxacillin. Either way this is some early noise about combination therapy not being terribly helpful in terms of patient centered outcomes.
Geriak M, Haddad F, Rizvi K, et al. Clinical Data on Daptomycin plus Ceftaroline versus Standard of Care Monotherapy in the Treatment of Methicillin-Resistant Staphylococcus aureus Bacteremia. Antimicrob Agents Chemother. 2019;63(5):e02483-18. Published 2019 Apr 25. doi:10.1128/AAC.02483-18
This is a prospective study evaluating vancomycin or daptomycin alone with daptomycin and ceftaroline in combination. Primary outcome was duration of bacteremia and in-hospital mortality. Of note, enrollment was stopped due to a mortality benefit seen in the combination therapy. We’ll get to that later. One things to note about this trial is the use of ceftaroline, a 5th gen cephalosporine (aka beta lactam) that has activity against MRSA. 40 patients were enrolled, 17 in the dual therapy group and 23 in the monotherapy group. Looking at the co-morbidities, a significant number of patients in the monotherapy group had malignancy or chronic lung disease. Reassuringly, the Pitt bacteremia score was fairly well balanced, though seems the monotherapy group had more co-morbidities:
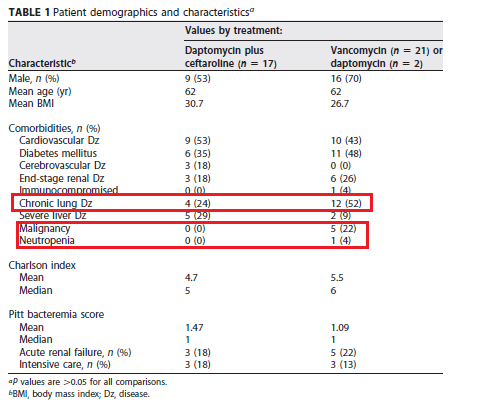
Overall, there was a trend towards survival in the dual therapy group:
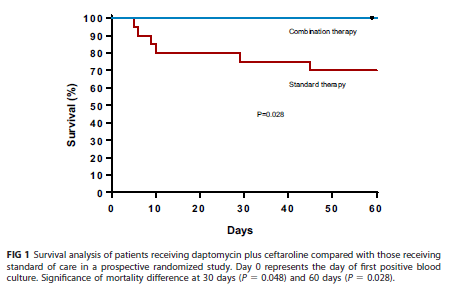
There were no renal injury reported, and only one treatment failure that occurred in the combination therapy. Given the prospective nature of this study, the lack of randomization, and the imbalance of malignancy in the groups (as well as the overall small size of the trial), it is difficult to make any meaningful assessment here. While the “signal” towards mortality benefit may justify the early stop in enrollment, further data may have suggested the lack of said benefit.
Jorgensen SCJ, Zasowski EJ, Trinh TD, et al. Daptomycin Plus β-Lactam Combination Therapy for Methicillin-resistant Staphylococcus aureus Bloodstream Infections: A Retrospective, Comparative Cohort Study. Clin Infect Dis. 2020;71(1):1-10. doi:10.1093/cid/ciz746
This retrospective observational cohort study compared Daptomycin monotherapy vs Daptomycin and beta lactam therapy (this was done at Detroit Medical Center and Henry Ford Hospital, where cefazolin was recommended to be added to vancomycin or daptomycin and to be continued for 48-72 hours after blood culture sterilization). Primary outcome was 60d all cause mortality and MRSA recurrence. The two groups did have some differences. For instance the dapto monotherapy group were more likely to have chronic kidney disease or heart failure, and the dual therapy group was more likely to have SSTI or bone/joint as the source of bacteremia. Further, more patients in the dapto monotherapy group had MICs to vanc >2 (75.8% vs 50% in the dual therapy, p-value <0.001). Cefepime and cefazolin were the most commonly used beta lactams.
Unadjusted results are below. There was a trend towards the dual therapy group having less persistent bacteremia. Further, the composite outcome seems to favor the dual therapy group, and this seems to be driven mostly by the recurrence rates:

The adjusted OR for clinical failure overall seems to favor the dual therapy group, though how an vanc MIC <2 does not reach any statistical significance is beyond me:
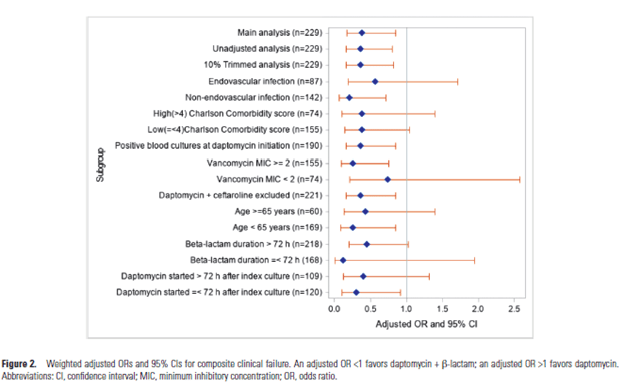
McCreary EK, Kullar R, Geriak M, et al. Multicenter Cohort of Patients With Methicillin-Resistant Staphylococcus aureus Bacteremia Receiving Daptomycin Plus Ceftaroline Compared With Other MRSA Treatments. Open Forum Infect Dis. 2019;7(1):ofz538. Published 2019 Dec 31. doi:10.1093/ofid/ofz538
This multicenter retrospective matched cohort study evaluated the use of daptomycin and ceftaroline for >72hrs in MRSA bacteremia. Patients were match with those using standard of care based on the source, age + or -10yrs and renal function. End point was all cause 30 day mortality, duration of bacteremia, and relapse/recurrence. Roughly 96% of patients in SOC received vancomycin while 6% got daptomycin. Groups were well matched for the most part, with the SOC having source control more often than dual therapy group:
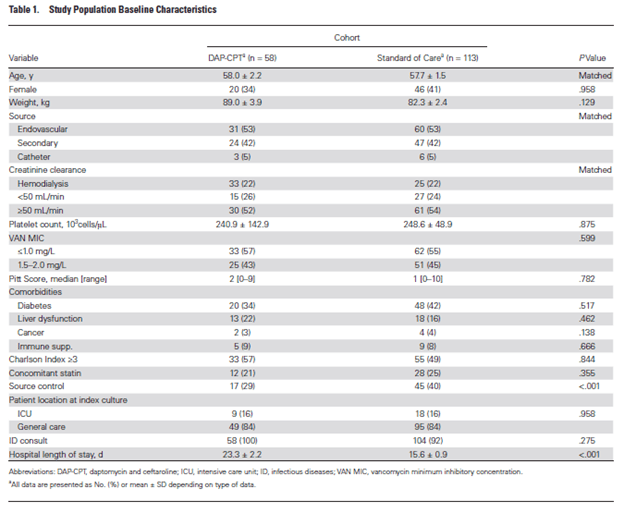
The overall summary of outcomes is highlighted in the figure below:

One thing to note here is the fact that patients in the dual therapy group did not receive DAP-CPT upfront. Rather, 29 (51%) patients received it as second line therapy, 26 (46%) as third line, and 2 as fourth line therapy. In other words, these were salvage regimens. Indeed, the authors note that there was no difference in 30d mortality in the SOC group and those who got Dual therapy within 72hrs. Despite how nice the numbers look, there are inherent biases in retrospective studies. Plus, given the fact these were salvage regimens, there may be further unaccounted for biases.
Alosaimy S, Sabagha NL, Lagnf AM, et al. Monotherapy with Vancomycin or Daptomycin versus Combination Therapy with β-Lactams in the Treatment of Methicillin-Resistant Staphylococcus Aureus Bloodstream Infections: A Retrospective Cohort Analysis. Infect Dis Ther. 2020;9(2):325-339. doi:10.1007/s40121-020-00292-8
This is a retrospective, single center study from Detroit evaluating monotherapy to Vanco/dapto with beta-lactam (at least >24hrs within 72hrs of vanco or dapto initiation). Primary outcome was a composite of 30d mortality, 60d recurrence, or persistent bacteremia (>5 days). 597 patients were evaluated (153 in monotherapy and 444 in the combination group). Given the large table for the patient characteristics, I’ll highlight some of the interesting differences. Patients in the combination group were more likely to be older (44.8% were >60 vs 32%, p-value 0.006), and were more likely to have been admitted from a nursing facility (23% vs 16.4% p-value 0.084). Further, patients in the combination therapy were more likely to have pneumonia as the source (22.1 vs 5.2% p-value <0.001), and monotherapy group was more likely to have bone and joint infection (26.1% vs 14% p-value 0.001). Further, the dual therapy group was more sick upfront (APACHE II 19.1 in combo group vs 7.2 in monotherapy group) and were more likely to be in the IC upfront (17.6% vs 9.8%, p-value 0.022). Beta lactam use as follows:

Outcomes are as follows:
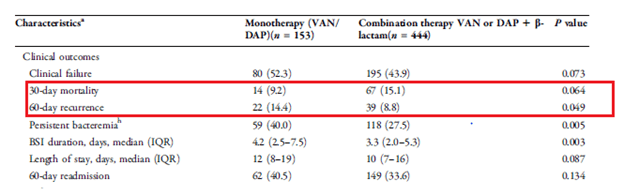
I guess I shouldn’t be surprised there was higher mortality in the combination group given how sick they were compared to the monotherapy. The results did not achieve statistical significance but approached it and it favored combination therapy. This was mostly driven by recurrence and persistence of bacteremia. There was no difference in terms of nephrotoxicity in both groups
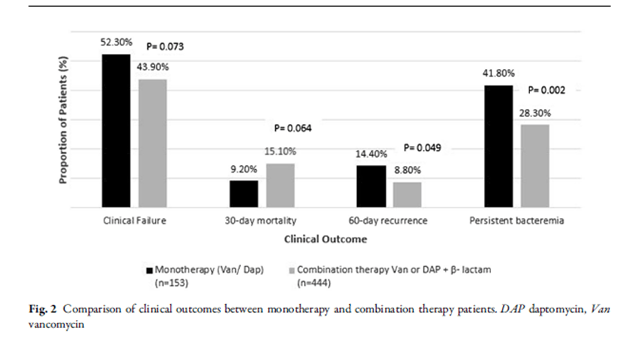
In multivariate analysis, after controlling for different variables, combination therapy was associated with lower odds for clinical failure (0.545 95% CI 0.364-0.817). There were no differences in renal failure in either group:
Tong SYC, Lye DC, Yahav D, et al. Effect of Vancomycin or Daptomycin With vs Without an Antistaphylococcal β-Lactam on Mortality, Bacteremia, Relapse, or Treatment Failure in Patients With MRSA Bacteremia: A Randomized Clinical Trial. JAMA. 2020;323(6):527-537. doi:10.1001/jama.2020.0103
So, this is the major trial that may have put a damper on the whole dual therapy for MRSA bit for at least now. This is a multicenter, open-0label, RCT powered for superiority in 27 hospitals throughout Australia, Singapore, New Zealand, and Israel. Patients were randomized 1:1 to standard of care (vanco or dapto) or addition of beta-lactam (cloxacillin, flucoxacillin, cefazolin) for the first 7 days of randomization. Primary outcome was a composite of 90d all cause mortality, persistent bacteremia at day 5, relapse, treatment failure 14d after randomization. 99% of patients received vancomycin during days 1-3 and overall troughs looked OK:
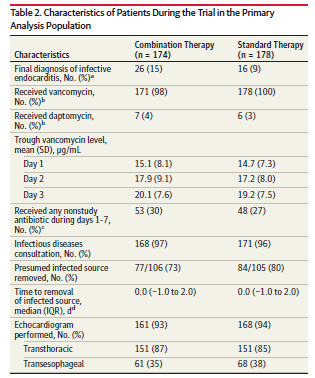
One of the things to note is the fact that enrollment stopped early due to higher than expected AKI in the combination group. They had originally planed to enroll 440 patients but this was stopped at 356 patients due to signal of harm. Ultimately, 352 patients were randomized, 174 to the combination arm, and 178 to the standard of care arm. Both groups were fairly well balanced. The primary outcome is as follows:
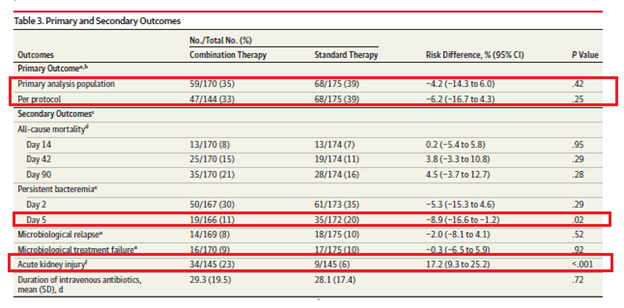
So, no difference between groups (the study was meant to be powered for superiority, but at least one can say that it was non-inferior?) in the composite outcome. The all cause mortality tended to favor the standard therapy group, while the combination group had less persistent bacteremia at day 5. Of course, the big issue here is the degree of renal injury that occurred here. In fact, the rise in creatinine held steady from day 5 though day 30. Of the 34 patients with renal failure, 6 required CRRT and 7 died.
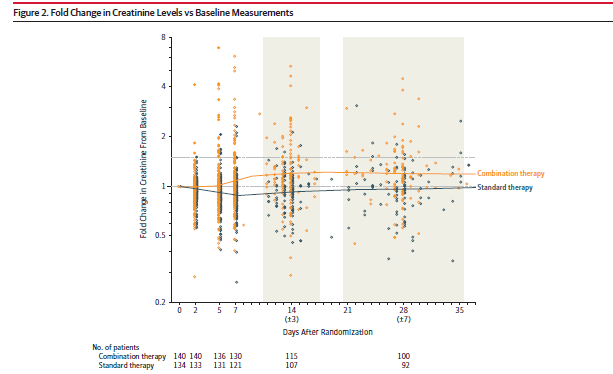
Why was there such excitement about dual therapy?
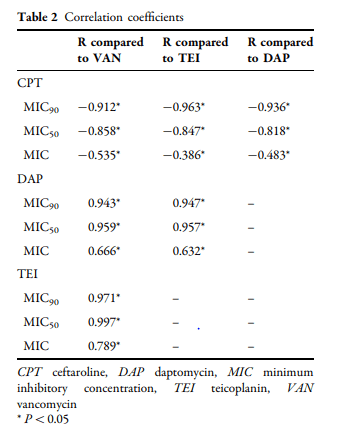
It has been proposed that as Staph aureus isolates get more resistant to Vancomycin or Daptomycin, it allows anti-staphylococcal beta lactams to become more susceptible. In other words, as the MIC to daptomycin or vancomycin increase, the MIC to beta lactam goes down. This has been seen in a few in-vitro studies. For instance, there has been an inverse correlation between ceftaroline MIC and daptomycin/vancomycin MIC (9):
Further, the addition of an anti-staphylococcal beta-lactam to MRSA allow significantly enhanced killing in a few scenarios (10):
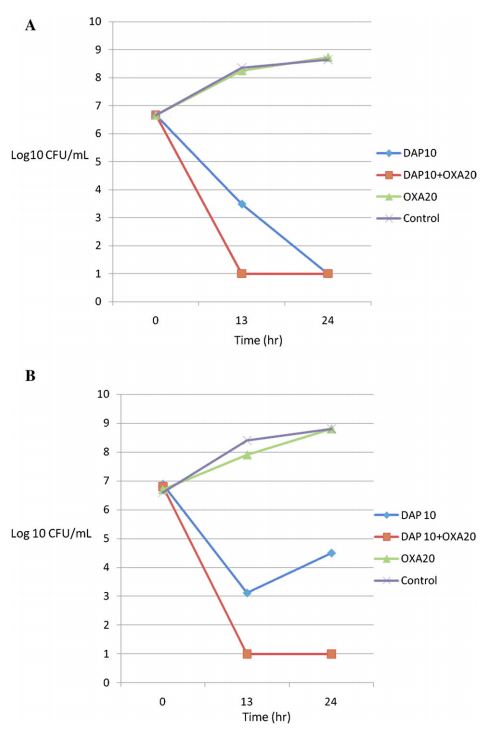
Why this happens is proposed to be due to a change in penicillin-binding protein expression. This is way beyond my paygrade, but suffice to say, selective pressure of vancomycin/daptomycin may allow an alteration in expression of specific PBP that allows beta-lactams to work better.
What conclusions can we draw from this?
It is difficult. On the one hand, it is not the first time that something that works in-vitro has no effect in real life. The highest quality data suggests the lack of benefit from dual antibiotic therapy, and worse of all, shows harm. Having said that, most of the patients in the CAMERA-2 study were given IV vancomycin in conjunction of beta lactams that do not have anti-MRSA activity (i.e not Ceftaroline). Read my prior post on vancomycin to see why this is an issue, but I am not surprised that the combination of vancomycin and beta-lactam resulted in higher incidence of renal failure. The other retrospective studies using Daptomycin and Ceftaroline did not show this level of harm. While wary of retrospective data, it may be that a less nephrotoxic combination (daptomycin-ceftaroline) may have some clinical utility in the right scenario. But as it stands, I am less than excited to pull the trigger on adding a beta-lactam for MRSA therapy. Maybe soon.
References:
- Rieg S, Joost I, Weiß V, et al. Combination antimicrobial therapy in patients with Staphylococcus aureus bacteraemia-a post hoc analysis in 964 prospectively evaluated patients. Clin Microbiol Infect. 2017;23(6):406.e1-406.e8. doi:10.1016/j.cmi.2016.08.026
- Davis JS, Sud A, O’Sullivan MVN, et al. Combination of Vancomycin and β-Lactam Therapy for Methicillin-Resistant Staphylococcus aureus Bacteremia: A Pilot Multicenter Randomized Controlled Trial. Clin Infect Dis. 2016;62(2):173-180. doi:10.1093/cid/civ808
- Casapao AM, Jacobs DM, Bowers DR, Beyda ND, Dilworth TJ; REACH-ID Study Group. Early Administration of Adjuvant β-Lactam Therapy in Combination with Vancomycin among Patients with Methicillin-Resistant Staphylococcus aureus Bloodstream Infection: A Retrospective, Multicenter Analysis. Pharmacotherapy. 2017;37(11):1347-1356. doi:10.1002/phar.2034
- Geriak M, Haddad F, Rizvi K, et al. Clinical Data on Daptomycin plus Ceftaroline versus Standard of Care Monotherapy in the Treatment of Methicillin-Resistant Staphylococcus aureus Bacteremia. Antimicrob Agents Chemother. 2019;63(5):e02483-18. Published 2019 Apr 25. doi:10.1128/AAC.02483-18
- Jorgensen SCJ, Zasowski EJ, Trinh TD, et al. Daptomycin Plus β-Lactam Combination Therapy for Methicillin-resistant Staphylococcus aureus Bloodstream Infections: A Retrospective, Comparative Cohort Study. Clin Infect Dis. 2020;71(1):1-10. doi:10.1093/cid/ciz746
- McCreary EK, Kullar R, Geriak M, et al. Multicenter Cohort of Patients With Methicillin-Resistant Staphylococcus aureus Bacteremia Receiving Daptomycin Plus Ceftaroline Compared With Other MRSA Treatments. Open Forum Infect Dis. 2019;7(1):ofz538. Published 2019 Dec 31. doi:10.1093/ofid/ofz538
- Alosaimy S, Sabagha NL, Lagnf AM, et al. Monotherapy with Vancomycin or Daptomycin versus Combination Therapy with β-Lactams in the Treatment of Methicillin-Resistant Staphylococcus Aureus Bloodstream Infections: A Retrospective Cohort Analysis. Infect Dis Ther. 2020;9(2):325-339. doi:10.1007/s40121-020-00292-8
- Tong SYC, Lye DC, Yahav D, et al. Effect of Vancomycin or Daptomycin With vs Without an Antistaphylococcal β-Lactam on Mortality, Bacteremia, Relapse, or Treatment Failure in Patients With MRSA Bacteremia: A Randomized Clinical Trial. JAMA. 2020;323(6):527-537. doi:10.1001/jama.2020.0103
- Barber KE, Ireland CE, Bukavyn N, Rybak MJ. Observation of “seesaw effect” with vancomycin, teicoplanin, daptomycin and ceftaroline in 150 unique MRSA strains. Infect Dis Ther. 2014;3(1):35-43. doi:10.1007/s40121-014-0023-0
- Dhand, Abhay et al. “Use of antistaphylococcal beta-lactams to increase daptomycin activity in eradicating persistent bacteremia due to methicillin-resistant Staphylococcus aureus: role of enhanced daptomycin binding.” Clinical infectious diseases : an official publication of the Infectious Diseases Society of America vol. 53,2 (2011): 158-63. doi:10.1093/cid/cir340
