Lyme disease is a difficult beast to diagnose. I think most people know some of the characteristics, including the fact it is a vector-borne illness that causes a characteristic rash (EM) that can lead to CNS disease, cardiac disease, and large joint arthritis. While the clinical characteristics are known, there is some difficulty in diagnosing this beast. As some know, it is a spirochete and these guys tend to be difficult to culture.
People have employed culture methods, however the conditions for Borrelia burgdorferi to grow tends to be specific and it can take up to 12 weeks of incubation in order to have any significant growth. This is partly due to its prolonged generation time (7h up to 20h or longer) during the log-phase growth. Periodically, someone must look for spirochetes under dark-field microscopy which is hard enough. When was the last time anyone used dark field microscopy? Exactly.
Having said that, people have been able to culture B. burgdorferi off skin lesions early during the primary phase of the illness:

But if you try to culture it off blood, the yield is not great with some studies citing anywhere from a 5% to up to a 15% yield, with more blood drawn = higher sensitivity. So, no one uses culture because
- Takes too much time (12 weeks?? Giving TB a run for its money)
- Need experience (again, I don’t know of anyone with dark field microscopes)
- Need lots of blood to make it count
- Not very sensitive overall, especially if someone gets Doxycycline, then cultures are useless
What about PCR? This has been used in studies but these were abandoned due to the fact that it lacks any meaningful sensitivity in blood and CSF (where it would be useful). PCR does show relatively decent sensitivity in skin biopsy, however it these scenarios, the clinical diagnosis is as good as the PCR.
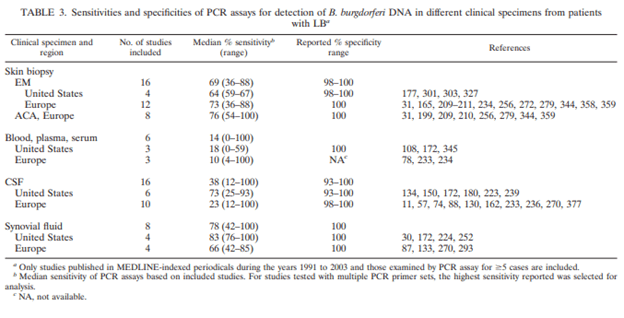
Given the difficulty of culture and low sensitivity of PCR, other non-direct methods were employed. Enter antibodies, where the body’s response to the organism is measured as opposed to the organism itself. Of course, the problem is cross reactivity of antibodies between diseases (like the lupus anticoagulant and syphilis, to name an example), but better than nothing. Prior to going further, lets review the structure of this spirochete:
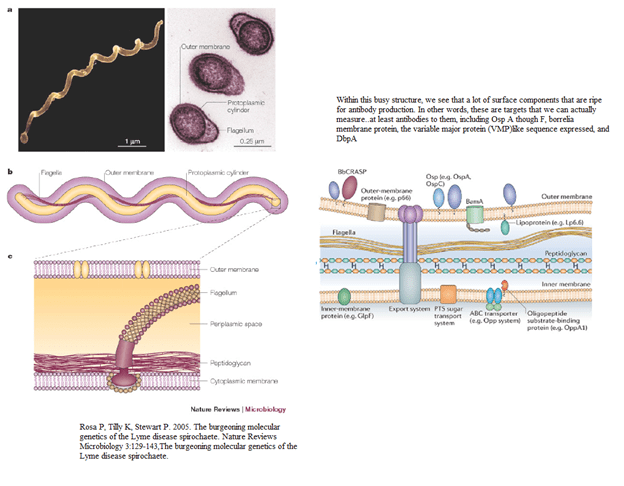
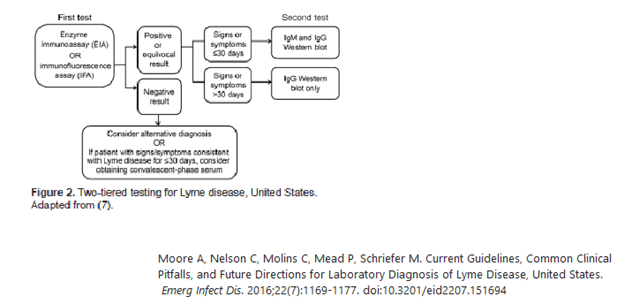
With this, you can see the outer membrane structures are prime real-estate for antibodies to attack and do their thing. Let’s review a bit of ELISA/EIA and the western blot, which are the modalities that are used to diagnose this beast:
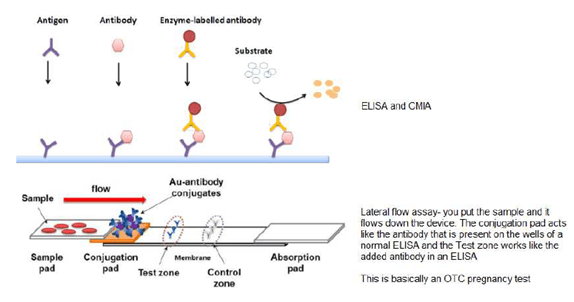
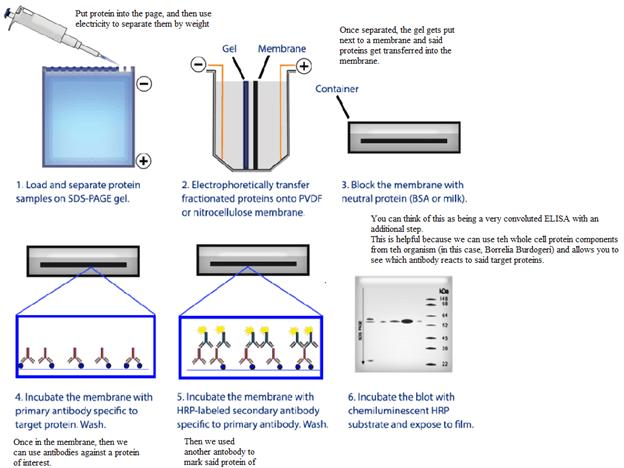
In other words, using EIA you detect antibodies against an antigen. Then, you confirm those with a western blot. Why you use two tests instead of one will be explained shortly.
So what are these antigens? They are as follows:
- Flagellin (FlaB) – illicits a strong IgG and IgM response, but it cross reacts with other bacteria, and antigens in neural tissue, synovium,, and myocardial tissue.
- Flagellar outer sheath protein (FlaA)
- OspC – expressed during tick feeding, may have a role in initial pathogenesis
- Decorin binding protein A – binds to host-collagen-associated proteoglycan decorin
- OspA and OspB – expressed later on during infection
- Vmp-like sequence expressed (VisE) protein
One of the problems of EIA is that it lacks specificity. Many of its antigens cross-react with bacterial antigens or other flagellar antigens. Why does this happen? Because the EIA assays use Whole cell sonicates: essentially lyse the bacteria and use all the proteins from said bacteria as antigens that the test will pick up. But if someone has an antibody for another infection and it cross-reacts with the Borrelia antigen, then you get a false positive.

One of the things to note is the low sensitivity of the EIA upfront. As noted above, in the acute phase (within 4 weeks), the overall sensitivity for the EIA is 40% and goes up to 61% beyond 4 weeks (aka the convalescent phase). This is because of the delayed antibody response i.e. the longer the infection has gone/it was, the more time the body has to make antibodies.
The point is proven, when looking at the above sensitivities for stage II and III lyme disease, but the other thing to note is the degree to which patients with other infections test positive for Lyme. This point is proven by another study evaluating EIA and Western Blot:
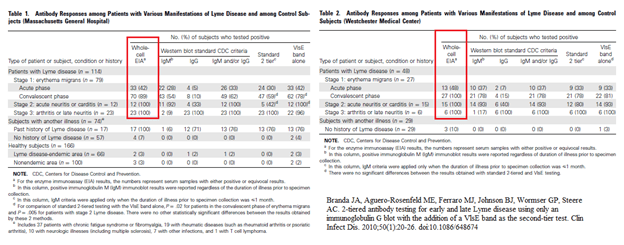
In other words, in late stages of Lyme, EIA becomes more sensitive. The caveat here is that this does not tell you past or current infection. So if you pick up the antibodies by EIA, and the patient was already treated, all it tells you they were exposed to Borrelia Burdgoferi but nothing else. The other caveat is that EIA is not very specific; again, these antigens cross-react with other antibodies so we need to confirm any positive EIA with Western Blot.
Why Western Blot? Remember that in Western Blot, we separate proteins based on weight (i.e. the proteins in the body). You know into which proteins your antibody should bind to. Let’s say, you know your protein of interest is 39 kDa, then when you finish running your Western Blot the antibodies should bind to this protein and you should see a band at 39 kDa, which we do here:

But what if instead of an expected binding, the antibody binds into a different antigen that is not one of the ones that we know? In other words, the antibody cross reacted with another antigen as the sample below and thus, we can ignore it (unlike an ELISA or EIA, which this binding may have flagged as positive):
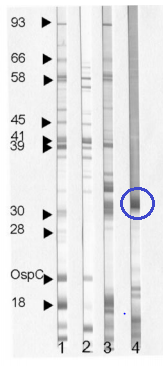
So to recap:
- If you have signs, symptoms, and epidemiological risk factors for Lyme disease, test with first an EIA or ELISA and then confirm with Western Blot
- Also, sensitivity tends to go up as you go through the stages
What about IgM vs IgG?
Official recommendations suggest that in the first 30d of illness, both IgM and IgG can be used. After 30d, IgG should be the only one used.
How to interpret Western Blot?
For positivity, two out of 3 bands need to be positive in the IgM immunoblot. These are bands OpsC, 41 or 39kDa. If using IgG immunoblot, then five out of 10 bands need to be positive (93, 66, 58, 45, 39, 30, 28, 21, OpsC, or 18kDa). This comes from a consensus guidelines on the topic. Further, immunoblot interpretation states that during the first 4 weeks of illness, either IgM or IgG can be used but after 4 weeks, only IgG immunoblot can be used. In this example, PC = positive control, T = test patient, 23= OpsC
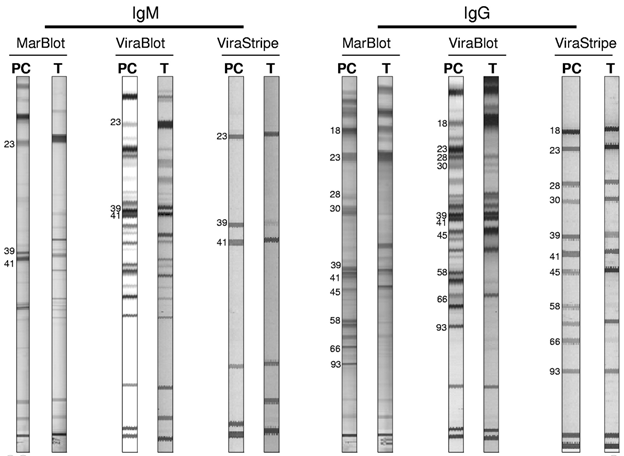
Notice the intensity of the bands; that represents antibody binding to the antigen. If you see a bend where you’re supposed to and meets the above criteria = Lyme. Despite this, sensitivity for the two-tiered testing tends to be fairly low still:
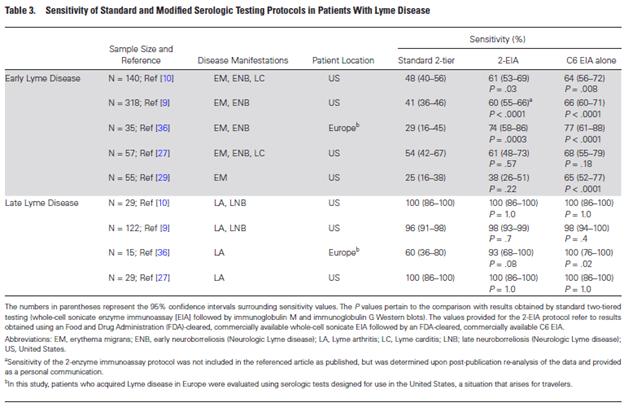
Again, this highlights the issue of diagnosing Lyme early on. Having said that, if someone has symptoms of Lyme disease and have a good exposure history, you probably should just give doxycycline anyways and don’t mess around with testing.
Next gen EIAs: as opposed of using the entire cell as an antigen, next gen EIA uses the C6 epitope within VlsE. One problem is these are not expressed early on when infection occurs, but later on, allow for good sensitivity. One of the ideas that has been floated around has been using a two-tiered tests with the “old” EIA and the “new” EIA. The idea being the targets are different for each EIA. This allows for fairly good sensitivity overall:

Other methods have included using a different type of IgG banding that uses the VIsE and eliminating IgM banding all together. A study from Mass Gen evaluated the use of such a method, comparing standard Western Blotting to a modified IgG where only 3 bands had to be positive, a VIsE tier where only the Vlse band was positive, and a hybrid of the 2. Sensitivities were comparable early on but were better for stage II and III lyme for the VlsE band:
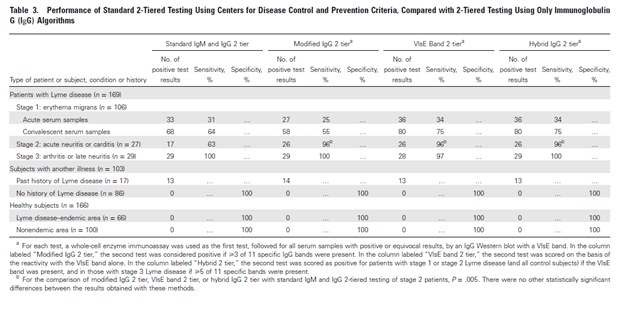
So what are the general caveats?
- If someone has had symptoms for <2 weeks and antibodies are negative, then repeat them at 2-4 weeks
- If the diagnosis is suspected, and initial testing is negative, then you can run another set after a month to increase sensitivity
- One can be antibody positive years down the line without disease
TL;DR –
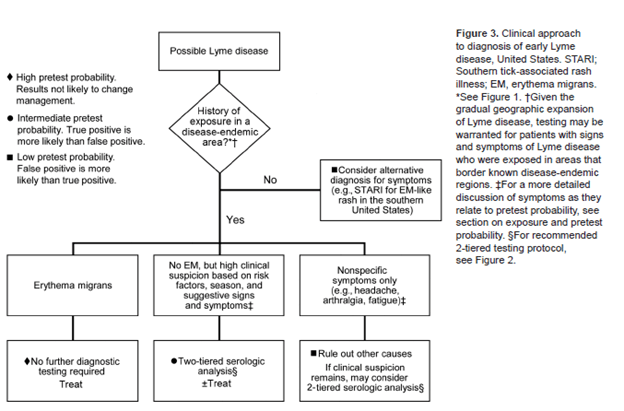
References:
Branda JA, Aguero-Rosenfeld ME, Ferraro MJ, Johnson BJ, Wormser GP, Steere AC. 2-tiered antibody testing for early and late Lyme disease using only an immunoglobulin G blot with the addition of a VlsE band as the second-tier test. Clin Infect Dis. 2010;50(1):20-26. doi:10.1086/648674
Aguero-Rosenfeld ME, Wang G, Schwartz I, Wormser GP. Diagnosis of lyme borreliosis. Clin Microbiol Rev. 2005;18(3):484-509. doi:10.1128/CMR.18.3.484-509.2005
Binnicker MJ, Jespersen DJ, Harring JA, Rollins LO, Bryant SC, Beito EM. Evaluation of two commercial systems for automated processing, reading, and interpretation of Lyme borreliosis Western blots. J Clin Microbiol. 2008;46(7):2216-2221. doi:10.1128/JCM.00200-08
Moore A, Nelson C, Molins C, Mead P, Schriefer M. Current Guidelines, Common Clinical Pitfalls, and Future Directions for Laboratory Diagnosis of Lyme Disease, United States. Emerg Infect Dis. 2016;22(7):1169-1177. doi:10.3201/eid2207.151694
Branda JA, Body BA, Boyle J, et al. Advances in Serodiagnostic Testing for Lyme Disease Are at Hand. Clin Infect Dis. 2018;66(7):1133-1139. doi:10.1093/cid/cix943
Waddell LA, Greig J, Mascarenhas M, Harding S, Lindsay R, Ogden N. The Accuracy of Diagnostic Tests for Lyme Disease in Humans, A Systematic Review and Meta-Analysis of North American Research. PLoS One. 2016;11(12):e0168613. Published 2016 Dec 21. doi:10.1371/journal.pone.0168613
