Pneumococcus, or Streptococcal pneumoniae, is an encapsulated gram positive organism. SO that is not a surprise to most people reading this. Here is a diagram of the capsule:
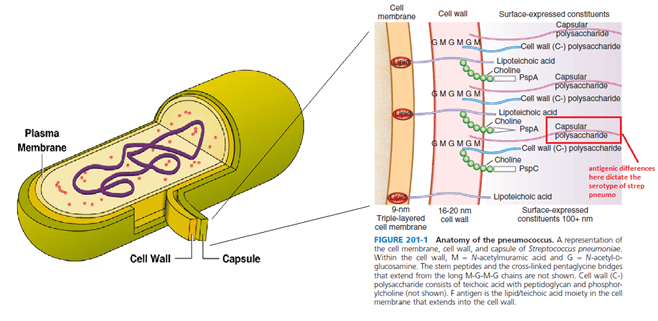
Strep pneumo joins other encapsulated organisms such as Haemophilus influenza and Nisseria meningiditis in causing havoc in certain immunodeficiency states. These include anything that affect the opsonization process:
- Complement deficiency
- B cell deficiency OR B cell line is screwy (read a type of lymphocytic leukemia/lymphoma)
- Lack of spleen (which has lots of macrophages and other lymphoid tissue)
- Any situation where you make too much of an antibody to the point you disregard all the other ones (read: myeloma)
- HIV – because you also screw around with your immunoglobulin process
In other words, if you can follow these images, you will figure out what diseases lead to increased to an incidence in encapsulated organisms.
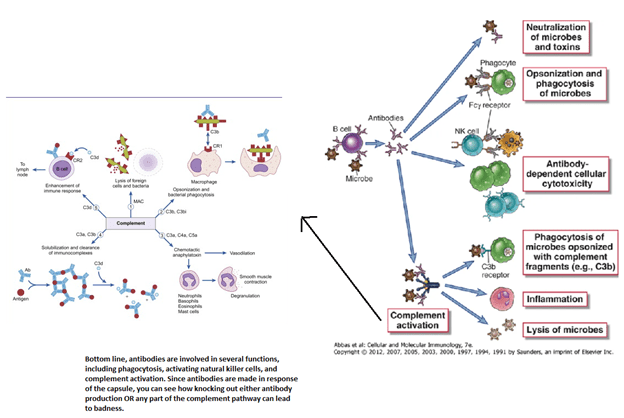
So what does this mean clinically? Of course, patients with CVID (the “teaching” immunodeficiency) tend to have higher incidence of invasive streptococcal pneumonia (1), however many times we will not know about any underlying immunodeficiency when they present. Most of the data tends to involve paediatric patients (2). In one retrospective study of 163 pediatric cases of invasive pneumococcal infections, immunological deficiency evaluation was done in 127 of these cases and an immune deficiency was found in 16% of cases. Most of these involved a sort of antibody deficiency. The point here is to look for some sort of immunodeficiency in cases of invasive Pneumococcus.
The data on adults is a bit more spares. An early retrospective study (3) evaluated 15 patients with recurrent pneumococcal bacteremia and matched them with 30 controls:
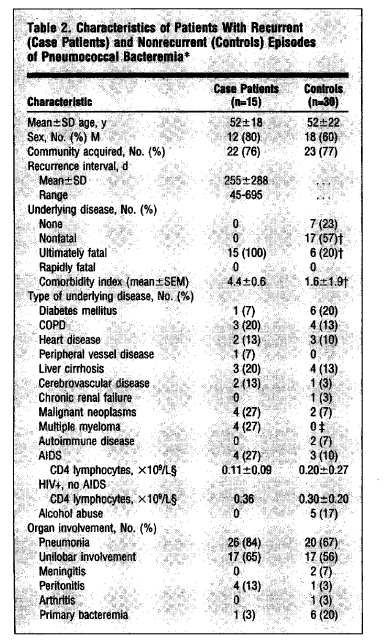
As seen, one of the notable findings was that those with invasive pneumococcus were more likely to have an underlying malignancy and MM. In both univariate and multivariate analysis, multiple myeloma was a risk factor for recurrent invasive pneumococcal disease (p value <0.02). Indeed, even in patients who had not being previously diagnosed with immunodeficiency, invasive infections can be the first clue to an underlying disorder. In a retrospective study (4) of patients under 40 with invasive infections, 7 were found to have an immunodeficiency after the first episode (2 had CVID, 1 had SPAD, one had IgG deficiency, one had IPH, and 2 had complement deficiency, while 2 more were diagnosed after the second episode (both had complement deficiency).
One of the biggest prospective studies came from France (5) and evaluated 119 patients who presented with invasive pneumococcal disease or H.flu and evaluated Ig deficiencies. In 37.8% of those, an immunoglobulin abnormality was found:
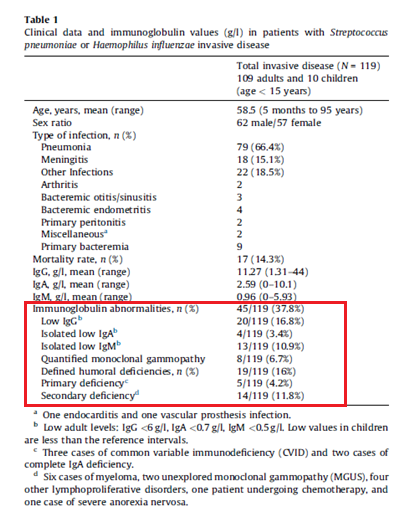
In these, 3 had CVID, 2 had complete IgA deficiency, and 14 had secondary immunodeficiency, including 6 MM, 2 MGUS, and 4 lymphoproliferative disorders. In another retrospective study (6) from Canada, 133 patients with invasive pneumococcal infection were evaluated. Of these, 57 had Ig levels measured and 18 of these (31.6%) were found to have hypogammaglobulinemia (defined as IgG < 7g/L or gamma globulin <6g/L by SPEP). In 13 of these cases, an underlying hematological malignancy was at play (CLL, MM, or NHL).
An interesting study (7) described 11 patients with specific polysaccharide antibody deficiency (SPAD). Here, the authors evaluated antibody titers against 7 serotypes before and after 4 to 8 weeks after administration of PPV23. A positive titer was a post-immunization titer <1.3ug/mL and/or less than a 4-fold rise (2-fold if already above >1.3). In this population, 6 patients with isolated SPAD presented with severe symptoms of meningiococal infections. Another patient with isolated SPAD presented with recurrent bronchitis, and 4 more had at least 2 hospitalizations and up to 8 in one case.
The bottom line?
- In patients who present with invasive pneumococcal or H. flu infections (i.e. bacteremia, meningitis), be on the lookout for an immunoglobulin deficiency
- Run at least an SPEP, UPEP, HIV to start with (HIV is also associated with invasive pneumococcus) and in some cases IgG, IgA and IgM levels
- If there is recurrent invasive pneumococcus, just call an immunologist. That topic makes my head hurt..
References:
- Oksenhendler E, Gérard L, Fieschi C, et al. Infections in 252 patients with common variable immunodeficiency. Clin Infect Dis. 2008;46(10):1547-1554. doi:10.1086/587669
- Gaschignard J, Levy C, Chrabieh M, et al. Invasive pneumococcal disease in children can reveal a primary immunodeficiency. Clin Infect Dis. 2014;59(2):244-251. doi:10.1093/cid/ciu274
- Rodríguez-Créixems M, Muñoz P, Miranda E, Peláez T, Alonso R, Bouza E. Recurrent pneumococcal bacteremia. A warning of immunodeficiency. Arch Intern Med. 1996;156(13):1429-1434. doi:10.1001/archinte.1996.00440120087008
- Sanges S, Wallet F, Blondiaux N, et al. Diagnosis of primary antibody and complement deficiencies in young adults after a first invasive bacterial infection. Clin Microbiol Infect. 2017;23(8):576.e1-576.e5. doi:10.1016/j.cmi.2017.02.005
- Martinot M, Oswald L, Parisi E, et al. Immunoglobulin deficiency in patients with Streptococcus pneumoniae or Haemophilus influenzae invasive infections. Int J Infect Dis. 2014;19:79-84. doi:10.1016/j.ijid.2013.10.020
- Cowan J, Do TL, Desjardins S, Ramotar K, Corrales-Medina V, Cameron DW. Prevalence of Hypogammaglobulinemia in Adult Invasive Pneumococcal Disease. Clin Infect Dis. 2018;66(4):564-569. doi:10.1093/cid/cix836
- Huson MA, Stolp SM, van der Poll T, Grobusch MP. Community-acquired bacterial bloodstream infections in HIV-infected patients: a systematic review. Clin Infect Dis. 2014;58(1):79-92. doi:10.1093/cid/cit596
