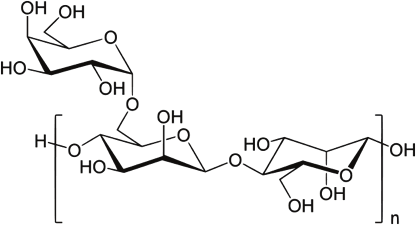
Galactomannan is a component of the cell wall of both Aspergillus spp and Penicillium spp. This component is secreted during the growth phase of the fungus, and higher galactomannan levels are correlated with fungal load in tissues.
This is the general structure of the galactomannan:
The test of choice is a sandwich ELISA. The double-sandwich ELISA incorporate a rat monoclonal antibody called EBA2, which is specific for the B 1-5 galactofuranose. For all I know, that is part of galactomannan polysaccharide. Here is the schematic of how this works.
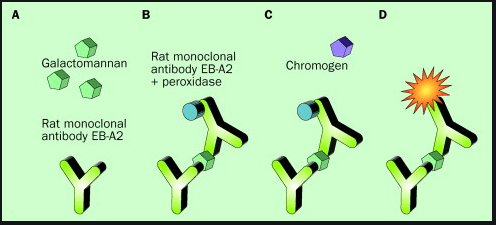
Once the antigen binds to the plate antibody, it gets sandwiched by another antibody, which in turn induces a reaction that leads to the production of a pigment. This can be read by spectrophotometer that is read out as an optical density (similar to how the BD glucan is read). OD in this case means the degree to which a refractive medium retards transmitted rays of light. Whatever that means.
The utility of this test comes down to it being a non-invasive test that we can use to attempt to diagnose invasive mold infections faster. How does it perform?
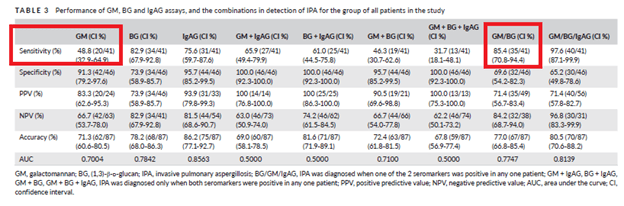
Most of the data tends to deal with neutropenic patients. One of the largest meta-analysis (1) evaluated 27 studies, mostly in the BMT/liquid malignancy population. The overall sensitivity was 71 (95% CI 0.68-0.74) and specificity was 89% (95% CI 0.88-0.9), with a positive likelihood ratio of 6.45 and negative likelihood ratio of 0.33. Most of the data is driven by the BMT population, where it performs the best when it comes to the adult population, as well as the hematology population. SOT population did not perform well, but this is likely due to the low numbers. Further, the OD cutoff of 0.5 paradoxically had the lower sensitivity but again this was likely due to the low numbers.

A Cochrane review from 2017 (2) found that using a cutoff of 0.5 had a higher sensitivity than reported in the prior review:
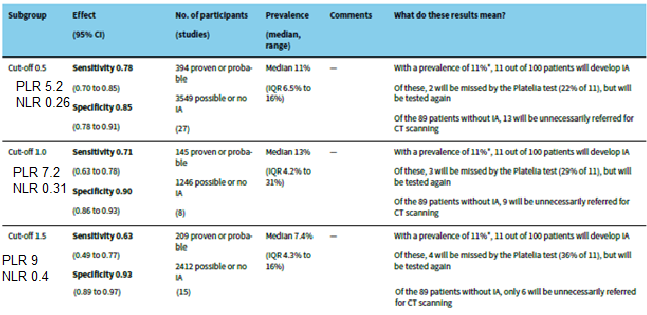
The pooled sensitivity was 73% (95% CI 65-80) and specificity was 89% (95% CI 84-92%) with hematology patients having the highest sensitivity overall (77%).
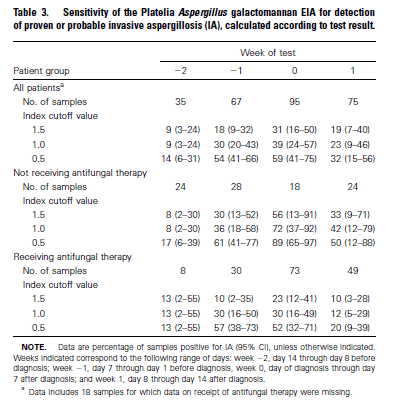
While not perfect, the serum galactomannan seems to be a good non-invasive modality for the BMT/hematology population. The performance of GM tends to drop with anti-mold therapy. This was evaluated in a cohort of patients receiving chemotherapy for heme malignancies or HSCT (3). These patients were included in a trial comparing itraconazole or fluconazole for anti-mold PPx according to standard practices. They also received empiric amphotericin B for therapy in cases where it was clinically indicated. Using day 0 as the day of diagnosis, they saw that anti-fungal therapy decreased the overall sensitivity when compared to those who were not receiving therapy in all cut off values:
In another study evaluated the use of posaconazole as antifungal prophylaxis in high risk patients undergoing AML chemo or allo HCT (4). More specifically, the investigators had data on AML with prolonged neutropenia following chemo, HSCT with prolonged neutropenia until engraftment, and GVHD requiring steroids. Primary endpoint was the diagnostic performance of serum GM, which was drawn twice per week. The vast majority of galactomannan drawn were negative, 71.7%. GM that were true positive accounted for 1.9% of all episodes, while 11.4% of positive galactomannan were false positive.
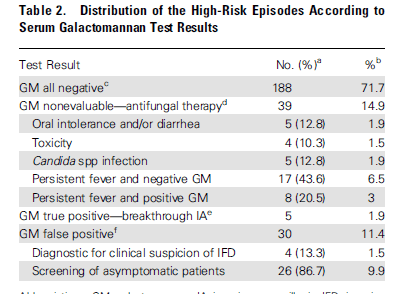
They evaluated the PPV and NPV value of using galactomannan in this population and noted that using galactomannan as a diagnostic modality rather than a screening modality resulted in a higher PPV, suggesting the lack of utility of “screening” high risk patients with GM.
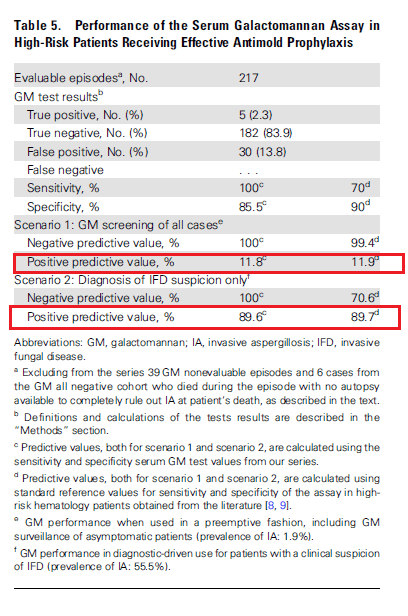
In other words, the use of galactomannan as a screening tool in setting of effective prophylaxis may not be useful, but using it as a diagnostic tool in conjunction with both CT and other markers may be of help.
Further data comes from a prospective study evaluating patients receiving conditioning chemotherapy and treatment for GVHD (5). Patients were given fluconazole for prophylaxis and given amphotericin B if there was a suspicion for invasive mold infection. In 67 patients, the sensitivity of galactomannan was as follows using a cut off of >1.0:
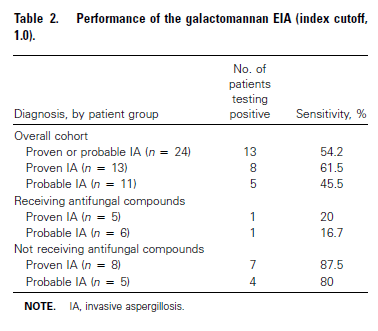
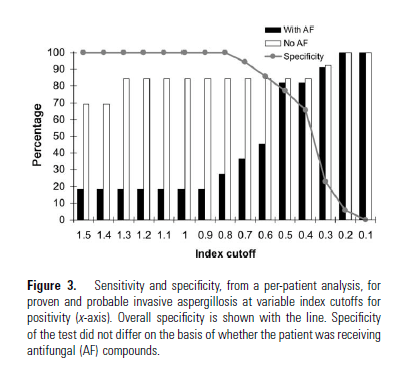
Dropping the cutoff to 0.5 (the standard now), increased the overall sensitivity without dropping the specificity. This was more significant in the group getting antifungals for prophylaxis, where the sensitivity increased dramatically at a GM cut off for 0.5:
The investigators also evaluated the relationship between fungal load and galactomannan. In a rabbit model, 10 uninfected rabbits were compared to 20 infected models. There was a strong correlation between fungal load and galactomannan serum level, with those getting therapy with amphotericin day showing significantly lower levels of galactomannan:
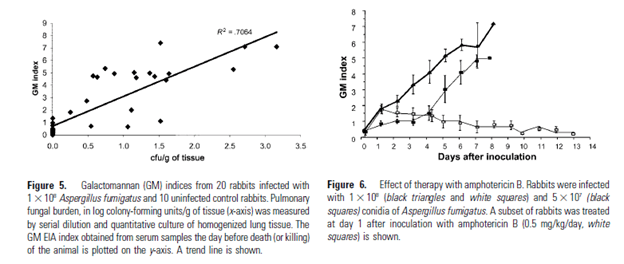
This suggest that antifungal therapy has an impact on the galactomannan level, with prophylaxis likely reducing the sensitivity of galactomannan in detecting invasive molds in the neutropenic population.
False Positives:
There are several situations in which the GM can yield a false positive result. Some of these are quite comical, for instance, contaminated ice pops (6) that were contaminated with galactomannan. Further false positives have come from IVIG (7). For instance, in a prospective study, galactomannan levels in 6 IVIG products were obtained and GM levels were drawn in patients who received those products before and after. In 4/6, there was a rise in median GM levels.
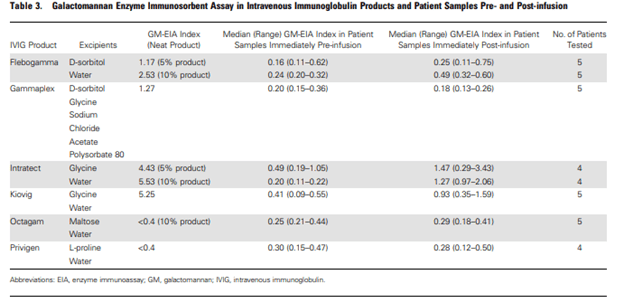
In a retrospective (8) study of hematology and SCT patients, 157 patients were evaluated for IFI with GM. 58 levels resulted positive, of which 29 were considered false positives. In this population, chronic GI GVHD was associated with false positive GM positivity when compared to other factors, including steroid use and other forms of GVHD. Further, the PPV of GM is low in the first 100 days following SCT, 37.5% versus 58.8%, however whether this was affected by the use of antifungal PPx is not addressed in the study. Other types of fungal infections, such as histoplasma, also has yielded false positive results.
The biggest culprit here is the ubiquitous piperacillin-tazobactam aka Zosyn aka half of the brain dead combination known as Parkla-myacin (the name of which can be modified depending on where you work, it may be referred to as Guerilla-cillin). Remember that Piperacillin is made from fermentation products of Penicillium, which also makes a galactomannan. This may be why cefepime tends to be used in neutropenic fevers, but I digress. One of the earliest studies came in 2004 (9). This prospective study evaluated 26 patients receiving a variety of antibiotics, including 13 getting pip-tazo and the other 13 getting a variety of antibiotics, including imipenem, ampicillin, nafcillin, ciprofloxacin and ceftriaxone. These patients were not high risk for invasive molds. They also evaluated the galactomannan level in vitro in several antibiotics. The overall in-vitro GM level was significantly higher in pip-tazo (at least this formulation) when compared to all others:
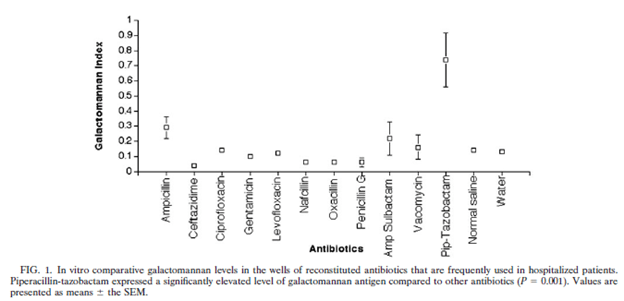
Further, the patients treated with pip-tazo were more likely to have a positive galactomannan.
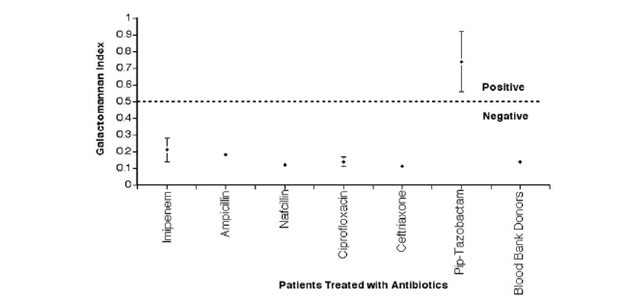
In another brief correspondence (10), 4 batches were found to be positive for galactomannan during a period in 2003. In another retrospective study during 2003, 218 patients had positive GMs of which, 38 were deemed to be false positives. In a case control analysis of 24 case patients and 24 controls, pip-tazo was associated with GM positivity (11):
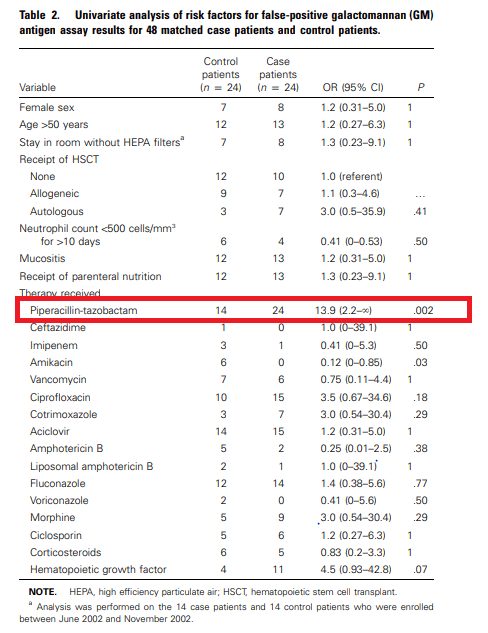
Further, 3 out of 4 batches of pip-tazo tested were found to be positive for GM.
The type of pip-tazo formulation seems to correlate with galactomannan positivity. For instance, another study evaluated the “original” pip-tazo with the generic formulation (12). In 85 patients getting pip-tazo for febrile neutropenia, 35 received the brand name while 50 received the generic formulation. The degree of positivity in the generic pip-tazo was an impressive 92% when compared to the 2.9% of the brand name one:
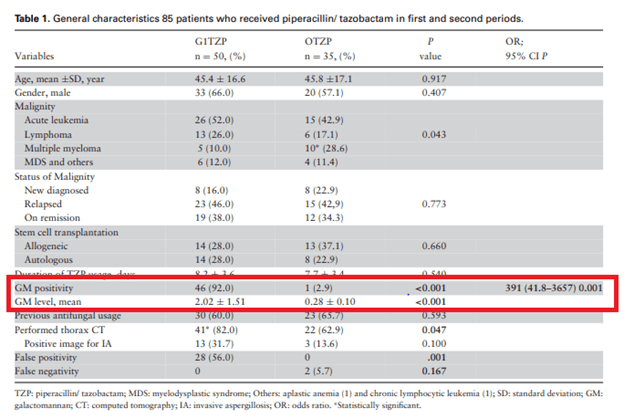
More recent data suggest this phenomenon may no longer be the case. For instance, in a study (13) evaluating the non-generic, Tazosin, there was no statistical difference between false positives in patients getting pip-tazo and those who did not:
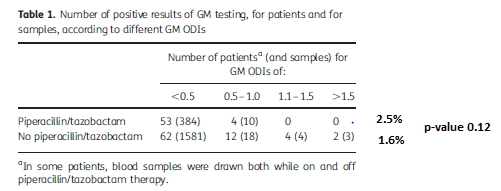
Further, in another small retrospective analysis (14), 27 patients who got pip-tazo were tested for galactomannan and only one tested positive, which was deemed a false positive. They also evaluated 32 lots of pip-tazo for the presence of GM and they did not find anyone that was positive. This suggests that perhaps newer formulations may not be responsible for false-positivity of GM as when compared to the 2002-2003 batches.
Utility of GM in Non-neutropenic Patients
So, if a risk factor for invasive molds is lack of neutrophils (aka the first line of defense against inhaling these things) and the GM positivity goes down with therapy, it would stand to reason that more neutrophils = less GM to go around = lower sensitivity of GM assay.
In a 2017 study (15), 37 invasive pulmonary aspergillosis patients and 83 controls (all of which were not neutropenic) were evaluated in terms of risk factors and galactomannan measurements in both serum and BAL samples. In generally, those with aspergillosis were more likely to have received steroids:
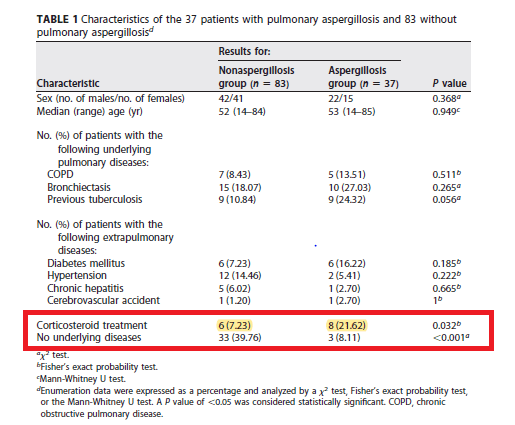
The overall sensitivity for GM detection off the serum was 37.8% (p-value 0.001), which is significantly lower than in the previously mentioned studies:
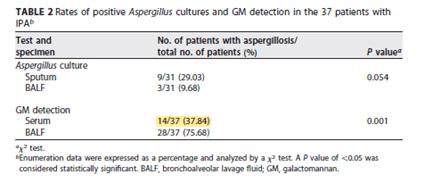
Indeed, looking at the previously mentioned meta-analysis of serum galactomannan (1), the sensitivity of serum galactomannan drop significantly when comparing the hematological population to the solid organ transplant population. At the risk of repeating images:
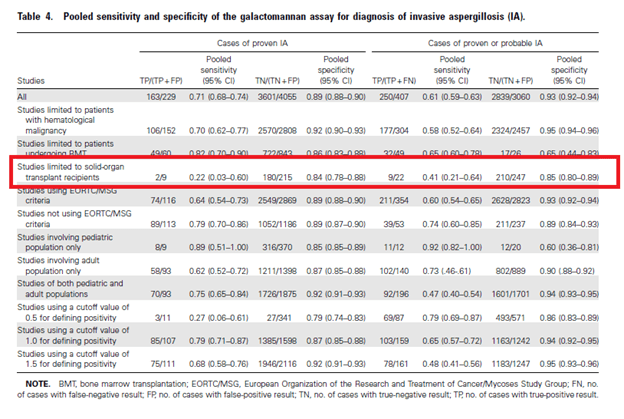
A multicenter retrospective study from the Czech Republic (16) evaluated the sensitivity and specificity of several fungal markers (BD glucan, GM, aspergillus IgA and IgG by themselves and in combination for the diagnosis of putative IPA (based on the AspICU criteria). Of 87 patients, 10 were diagnosed with proven IPA and 31 were diagnosed with putative IPA. Rest were found to be colonized. None of the patients were neutropenic. The overall sensitivity for galactomannan was 48.8% and combining the result with BD glucan yielded the highest sensitivity overall, yielding a PLR of 5.61 and NLR of 0.56:
The sensitivity of GM was highest in the ICU population when compared to those who were on other wards:

A single center prospective study (17) found the sensitivity and specificity of serum galactomannan in non-neutropenic, critically ill patients to be 66.6%, and specificity at 97.6% (PLR and NLR of 27.75 and 0.34 respectively). PPV and NPV were 80 and 95.4% respectively, using a cut-off value of >0.5. This is a tad bit higher than previously reported, but still lower than in the neutropenic population. Overall, seems that while helpful, it is not as useful in ruling out disease in this population if the suspicion is high enough.
TL;DR –
- Serum galactomannan is a good non-invasive tool in those who are at high risk of invasive molds
- The level (measured as an optical density) is correlated with fungal load
- Note that GM is produced by aspergilus and penicillium spp, not any of the molds of the Mucorales (i.e Rhizopus, Murcor, etc)
- Sensitivity drops if someone is on anti-mold prophylaxis, not neutropenic
- On that vein, if neutropenic patients are on anti-mold prophylaxis (usually voriconazole, posaconazole, or isavuconazole depending on your shop), then screening with weekly GM is useless. GM for diagnostic purposes seems to be where it is at
- False positives are seen with old formulations of piperacillin-tazobactam (because it is a byproduct of penicillium) and some fungal diseases. The other big one to watch out for is chronic GI graft vs host disease, likely due to leakage of some fungi/bacteria that cross-reacts with GM assay.
References:
- Pfeiffer CD, Fine JP, Safdar N. Diagnosis of invasive aspergillosis using a galactomannan assay: a meta-analysis. Clin Infect Dis. 2006;42(10):1417-1427. doi:10.1086/503427
- Leeflang MM, Debets-Ossenkopp YJ, Wang J, et al. Galactomannan detection for invasive aspergillosis in immunocompromised patients. Cochrane Database Syst Rev. 2015;2015(12):CD007394. Published 2015 Dec 30. doi:10.1002/14651858.CD007394.pub2
- Marr KA, Laverdiere M, Gugel A, Leisenring W. Antifungal therapy decreases sensitivity of the Aspergillus galactomannan enzyme immunoassay. Clin Infect Dis. 2005;40(12):1762-1769. doi:10.1086/429921
- Duarte RF, Sánchez-Ortega I, Cuesta I, et al. Serum galactomannan-based early detection of invasive aspergillosis in hematology patients receiving effective antimold prophylaxis. Clin Infect Dis. 2014;59(12):1696-1702. doi:10.1093/cid/ciu673
- Marr KA, Balajee SA, McLaughlin L, Tabouret M, Bentsen C, Walsh TJ. Detection of galactomannan antigenemia by enzyme immunoassay for the diagnosis of invasive aspergillosis: variables that affect performance. J Infect Dis. 2004;190(3):641-649. doi:10.1086/422009
- Guigue N, Menotti J, Ribaud P. False positive galactomannan test after ice-pop ingestion. N Engl J Med. 2013;369(1):97-98. doi:10.1056/NEJMc1210430
- Ramsay I, Gorton RL, Patel M, et al. Transmission of Hepatitis B Core Antibody and Galactomannan Enzyme Immunoassay Positivity via Immunoglobulin Products: A Comprehensive Analysis. Clin Infect Dis. 2016;63(1):57-63. doi:10.1093/cid/ciw222
- Asano-Mori Y, Kanda Y, Oshima K, et al. False-positive Aspergillus galactomannan antigenaemia after haematopoietic stem cell transplantation. J Antimicrob Chemother. 2008;61(2):411-416. doi:10.1093/jac/dkm463
- Walsh TJ, Shoham S, Petraitiene R, et al. Detection of galactomannan antigenemia in patients receiving piperacillin-tazobactam and correlations between in vitro, in vivo, and clinical properties of the drug-antigen interaction. J Clin Microbiol. 2004;42(10):4744-4748. doi:10.1128/JCM.42.10.4744-4748.2004
- Sulahian A, Touratier S, Ribaud P. False positive test for aspergillus antigenemia related to concomitant administration of piperacillin and tazobactam. N Engl J Med. 2003;349(24):2366-2367. doi:10.1056/NEJM200312113492424
- Adam O, Aupérin A, Wilquin F, Bourhis JH, Gachot B, Chachaty E. Treatment with piperacillin-tazobactam and false-positive Aspergillus galactomannan antigen test results for patients with hematological malignancies. Clin Infect Dis. 2004;38(6):917-920. doi:10.1086/383148
- Demiraslan H, Atalay MA, Eren E, et al. Assessing the risk of false positive serum galactomannan among patients receiving piperacillin/tazobactam for febrile neutropenia. Med Mycol. 2017;55(5):535-540. doi:10.1093/mmy/myw129
- Mikulska M, Furfaro E, Del Bono V, et al. Piperacillin/tazobactam (Tazocin™) seems to be no longer responsible for false-positive results of the galactomannan assay. J Antimicrob Chemother. 2012;67(7):1746-1748. doi:10.1093/jac/dks111
- Vergidis P, Razonable RR, Wheat LJ, et al. Reduction in false-positive Aspergillus serum galactomannan enzyme immunoassay results associated with use of piperacillin-tazobactam in the United States. J Clin Microbiol. 2014;52(6):2199-2201. doi:10.1128/JCM.00285-14
- Zhou W, Li H, Zhang Y, et al. Diagnostic Value of Galactomannan Antigen Test in Serum and Bronchoalveolar Lavage Fluid Samples from Patients with Nonneutropenic Invasive Pulmonary Aspergillosis. J Clin Microbiol. 2017;55(7):2153-2161. doi:10.1128/JCM.00345-17
- Dobias R, Jaworska P, Tomaskova H, et al. Diagnostic value of serum galactomannan, (1,3)-β-d-glucan, and Aspergillus fumigatus-specific IgA and IgG assays for invasive pulmonary aspergillosis in non-neutropenic patients. Mycoses. 2018;61(8):576-586. doi:10.1111/myc.12765
- Acosta J, Catalan M, del Palacio-Pérez-Medel A, et al. Prospective study in critically ill non-neutropenic patients: diagnostic potential of (1,3)-β-D-glucan assay and circulating galactomannan for the diagnosis of invasive fungal disease. Eur J Clin Microbiol Infect Dis. 2012;31(5):721-731. doi:10.1007/s10096-011-1365-0
