Strongylodiasis is probably one of those disease you don’t think about. After all, it is generally thought to be a tropical infectious disease, and as a result, there is little Western familiarity with it. While the number of infected people worldwide ranges from 30-100 million (1), with a recent review noting the prevalence being 10-40% in subtropical countries reaching as high as 60% in some areas, the vast majority tend to be asymptomatic or chronically infected with little to no symptoms. Further, travel and global warming means the population carrying this helminth is increasing. The increasing proportion immunodeficient patients, ranging from rheumatological patients to hematological and solid organ transplant patients in the Western world means that we need to be more aware of the implications of this helminth infection.
Life Cycle and Pathophysiology of Hyperinfection
The lifecycle is a bit complicated for most parasites, but it is an interesting one for Strongylodiasis. Part of this is the fact that it can actually complete its life cycle within the host (3):
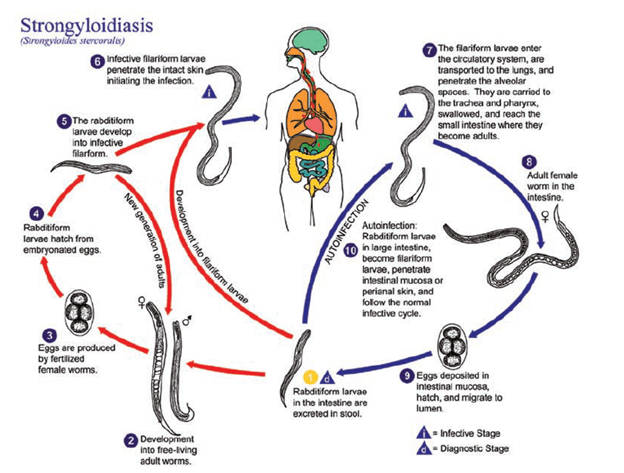
So what happens? The parasitic lifecycle occurs when the worm (usually within the soil) penetrates the human skin (usually the foot, if some one is walking barefoot) and gains access to their new host (1, 2).
- The larvae access the venous circulation
- They go into the pulmonary circulation and penetrate the alveolar spaces
- From here, they go through the bronchial tree, trachea, and larynx and reach the pharynx.
- After reaching the pharynx, they are swallowed (I know)
- The larvae make their way down the small intestine and they penetrate the mucosa and mature into adult females
- They deposit eggs, which hatch and are expected into stool.
- From here, one of 2 things happen
- Eggs hatch into rhabditoid larvae and migrate into the lumen, and end up in stool
- They mature into filariform larvae in the mucosa wall and penetrate the mucosal wall or perianal skin (causing pruritus -> fecal-oral route) and autoinfection
As such, patients can continue to be auto-infected for years and even decades. Usually, risk for acquiring infection include exposure to a combination of (1):
- Soil contamination with human feces
- Environmental conditions that allow survival
- Contact of human skin with contaminated soil
While I will not cover acute or chronic strongylodiasis, I should mention these tend to not cause many symptoms (1, 3). For instance, acute strongylodiasis tend to cause mild abdominal symptoms and once it gets into the chronic stage (usually by week 3, once it goes into the small intestine) the vast majority of patients tend to be asymptomatic. The immune system tends to keep the parasites in balance and once this balance shifts, one can see a large parasitic burden in either the lung or the intestine (due to higher depositing of eggs and more larvae penetrating the mucosa). As such (4)
- Hyperinfection – a large burden of parasites in a usual site of infection (either lung or intestine) that leads to severe symptoms
- Disseminated infection – usually presence of parasite outside of this, either blood stream, liver, CNS, etc.
What is this balance? Essentially, it boils down to the balance between T helper (Th1) and 2 (Th2) responses. In general, Th2 response leads to production of IL-5, eosinophils, and IgA, which is important for elimination of parasites (5). Infection with Strongyloidiasis leads to mast cell degranulation and direct killing of parasite by eosinophils, with mast cells being activated by IL-4 (also activated by Th2). Anything that increases Th2 response means more effective killing of parasites. Anything that decreases Th1 response means that, as a result of decreasing Th1 response, it will decrease parasite killing (or course, it is way more complicated than this, this sets up the major risk factors for hyperinfection):
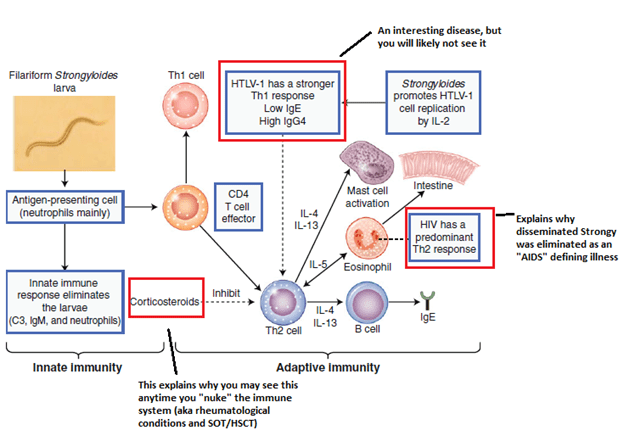
You can see what puts you at risk for hyperinfection: HTLV-1 infection, steroid therapy, and transplant.
Risk Factors for Hyperinfection
So there is no real “study” or “retrospective cohort” to tell us the specific risk factors, however, there are several patterns that arise. For instance, several case reports have suggested the link between HTLV-1 and strongylodiasis, however a Japanese cohort study was able to find a link between these entities (6). 31 HTLV-1 positive and 11 HTLV-1 negative patients were evaluated and found that 19 out of 31 patients with HTLV-1 were stool positive for S. stercolaris (61.3%) compared to 2 out of 11 HTLV-1 negative patients (18.2%). Further, there was a correlation between HTLV-1 proviral load and direct fecal smear positivity for strongylodiasis (p = +0.566, p<0.01).
Other risk factors include hematopoetic stem cell transplant and malignancies as well as solid organ transplant. A review (3) evaluated 54 renal transplant patients with Strongylodiasis, of which 33 presented with hyperinfection syndrome. A total of 44% of these patients died (though the presentation varied from disseminated to gastrointestinal strongylodiasis:
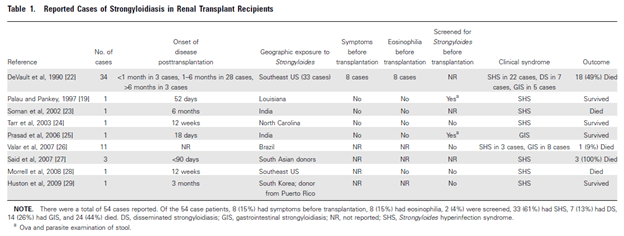
9 non-renal transplant patients were also evaluated, of which 8 presented with hyperinfection. Mortality rate was 78% in this small sample. That same review also evaluated 7 cases in HSCT patients, of which 6 had died. Another review on HSCT (7) from Houston, TX found 25 patients who had evidence of Strongyloidiasis. In this cohort, 6 had evidence of hyperinfection, with 4 of these having polymicrobial bacteremia with E. cloacae, K. pneumo, Ecoli, or E. faecalis. This represented a 24% rate of autoinfection.
One of the most comprehensive reviews (8) evaluated several risk factors, based mostly on cases:
- Corticosteroids – any dose, either high or low, has been implicated in the development of hyperinfection, likely due to the induction of eosinophilia. As mentioned before, this is due to downregulation of Th2 response
- Vinca drugs – vincristine is the main drug implicated, likely due to the decrease of intestinal motility
- Hypogammaglobulinemia – case reports have suggested the lower immunoglobulin leads to ongoing parasitemia
What about HIV? Per one review (8) there have been less than 30 cases of hyperinfection in relation to HIV, and another review (9) noted the elimination of Strongylodiasis as an AIDS-defining illness in the late 1980s. This may be due to the altered Th1 response rather than Th2. Notably, there is some case reports and mice studies suggesting that cyclosporine may have antiparasitic activity and be protective against hyperinfection syndrome, but how this pans out in clinical practice is poorly understood (3).
Symptoms
I am focusing mostly on the hyperinfection syndrome, but these tends to fall into 2 camps. Remember this is due to increased larval migration due to autoinfection in setting of decreased immunity, but they fall within these 2 organs (1,3,9)
- Pulmonary – ranges from asthma (cough, dyspnea) to pneumonia with diffuse bilateral infiltrates and consolidation. Other complications such as diffuse alveolar hemorrhage, necrotizing pneumonia, and eosinophilic pleural effusions have been described.
- Gastrointestinal – ranges from mild dyspepsia to abdominal pain, hematemesis, or abdominal distention. Some reports of ulcerative enteritis with ulcers and fibrosis. Ileus or obstruction have been observed
A retrospective study of 133 patients (17) found that fever, respiratory symptoms, and GI symptoms were the most common manifestations:
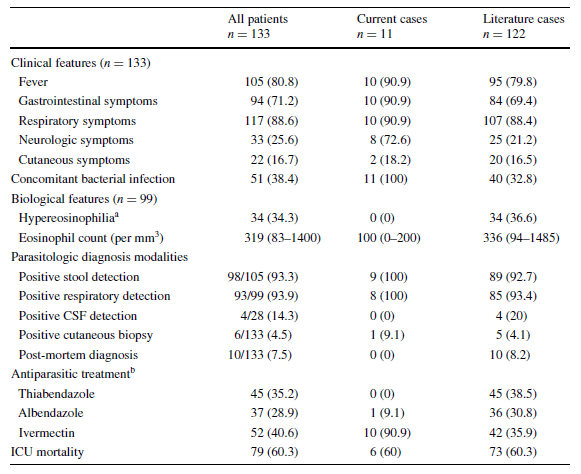
It boils down to the following: if you see a patient from an endemic area (usually South America) who had some GI symptoms (diarrhea), got steroids or had a transplant and now has GI bleed or severe abdominal pain with diffuse pulmonary infiltrates and respiratory failure, think of disseminated or hyperinfection with Strongyloidiasis. Another clue would be concurrent gram negative bacteremia. As mentioned above, many case reports have noted a superimposed infection with enteric gram-negative organisms including E. coli (10), Streptococcus bovis (11), and K. pneumoniae. This is likely due to damage to the GI wall and translocation of GI flora.
Diagnosis
Eosinophilia can be seen in up to 90% of cases (12) and many use the presence of eosinophilia as a clue to look for parasitic infections. This does not seem to hold up for hyperinfection syndrome, however. In a cohort of 33 patients (13) with strongylodiasis only 21 (63.6%) had peripheral eosinophilia. Another remote review (14) found that only a small percentage of patients who were immunocompromised and had hyperinfection with strongylodiasis had a high percentage of peripheral eosinophilia:

Notably, this also suggest that those who had low eosinophils had worse outcomes. Indeed, in the univariate analysis of a retrospective cohort (17) eosinophilia was found to be protective for ICU mortality (OR 0.28, 95% CI 0.12-0.66) as well a protective factor for shock univariate (OR 0.32 95% CI 0.14-0.77) and multivariate (0.21, 95% CI 0.06-0.70) analyses:
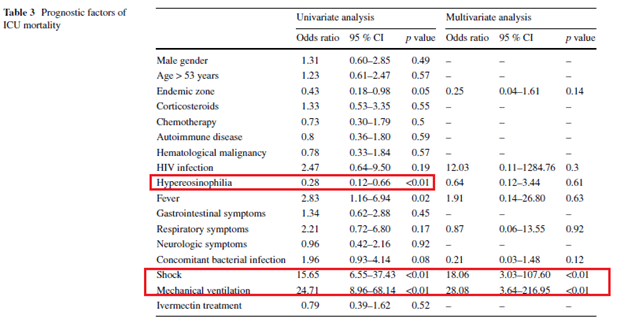
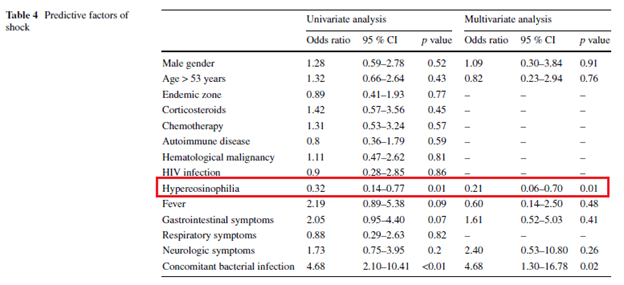
Another retrospective study (15) found that patients with immunosuppressive conditions had lower peripheral eosinophils compared to controls (HIV patients had 413 +/- 674 cells/uL, immunosuppressive drug patients 1073.2 +/- 1,642.2 cell u/L, healthy patients 1435 +/- 1825.8 cells u/L):
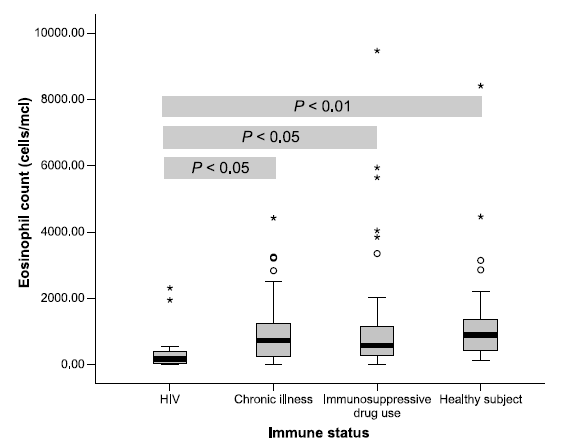
All this to say, in the absence of peripheral eosinophilia, the diagnosis of hyperinfection SHOULD NOT be discarded.
An overview of traditional microbiological methods of diagnosis is as follows (3, 5).
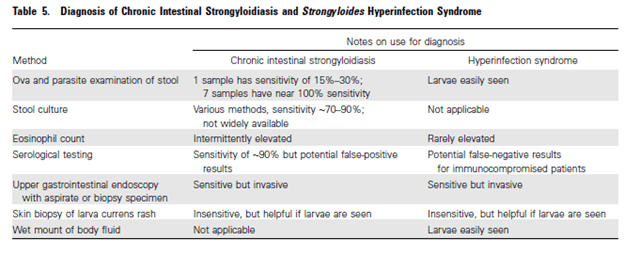
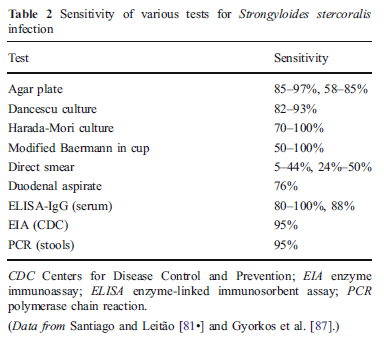
Keep in mind the sensitivities are for acute strongylodiasis and chronic strongylodiasis. The standard of diagnosis is stool microscopy exam for parasite and ova. Despite this, the sensitivity of one stool sample ranges from 30-50% (12), with three consecutive stool samples improving sensitivity to up to 95%. This is because adult parasites may produce only a handful of eggs per day (8). The most common culture method is the blood agar plate, in which serpiginous tracks of bacterial growth along the paths of motile larvae become apparent after 1-2 days of incubation (8). ELISA serological assays measure IgG response to Strongyloides antigen extracts in serum but these can take up to 4- weeks before they turn up positive (16). While they are a great way to screen someone, they offer little in ways of figuring out acute infection.
All of the above applies to chronic strongylodiasis. In hyperinfection, because of the increased parasitic load, any sample should be teeming with larvae, making the diagnosis much simpler. A retrospective study of hyperinfection syndrome (17) found that 93.3% of patients in a 133 patient cohort had a positive stool O&P, while respiratory samples were positive in 93.9% of samples.
Therapy:
The most common therapeutic agent used is Ivermectin. A review (1) noted the cure rates for ivermectin were 74-84% compared to 48% for albendazole. Further, ivermectin is preferred in these scenarios because it is -cidal in both the larval and adult forms of Strongylodiasis. In chronic infection, ivermectin is typically given for 2 days with repeat in about 2 weeks if stool O&P is still positive (3, 5, 12). For hyperinfection, treatment is 200ug/kg daily until there is no larvae present in stool, which can take up to 2 weeks. This is followed by one dose 2 weeks after clearance of larvae to ensure eradication (duration of 1 autoinfection cycle is about 2 weeks). Most of this is based case reports and expert opinion, though ivermectin is fairly tolerable making this a viable option for therapy:
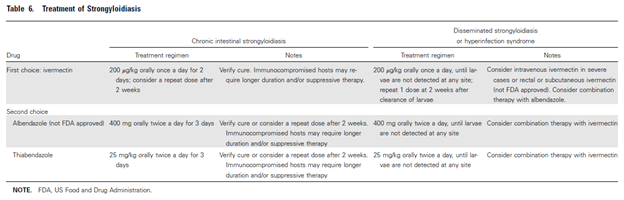
Ivermectin can also be given in IV as well rectally. Data has also suggested that ivermectin levels ranging from 11.4 to 49.6ng/L have correlated with eradication of Strongylodiasis (4). Along with this, reduction of immunosuppressives (if feasible) has been recommended.
References:
- Krolewiecki A, Nutman TB. Strongyloidiasis: A Neglected Tropical Disease. Infect Dis Clin North Am. 2019;33(1):135-151. doi:10.1016/j.idc.2018.10.006
- Barros, Nicolas & Montes, Martin. (2014). Infection and Hyperinfection with Strongyloides stercoralis: Clinical Presentation, Etiology of Disease, and Treatment Options. Current Tropical Medicine Reports. 1. 223-228. 10.1007/s40475-014-0030-y.
- Roxby AC, Gottlieb GS, Limaye AP. Strongyloidiasis in transplant patients. Clin Infect Dis. 2009 Nov 1;49(9):1411-23. doi: 10.1086/630201. PMID: 19807271; PMCID: PMC2913967.
- Kassalik M, Mönkemüller K. Strongyloides stercoralis hyperinfection syndrome and disseminated disease. Gastroenterol Hepatol (N Y). 2011;7(11):766-768.
- Marcos LA, Terashima A, Canales M, Gotuzzo E. Update on strongyloidiasis in the immunocompromised host. Curr Infect Dis Rep. 2011 Feb;13(1):35-46. doi: 10.1007/s11908-010-0150-z. PMID: 21308453.
- Satoh M, Kiyuna S, Shiroma Y, Toma H, Kokaze A, Sato Y. Predictive markers for development of strongyloidiasis in patients infected with both Strongyloides stercoralis and HTLV-1. Clin Exp Immunol. 2003 Sep;133(3):391-6. doi: 10.1046/j.1365-2249.2003.02224.x. PMID: 12930366; PMCID: PMC1808792.
- Safdar A, Malathum K, Rodriguez SJ, Husni R, Rolston KV. Strongyloidiasis in patients at a comprehensive cancer center in the United States. Cancer. 2004 Apr 1;100(7):1531-6. doi: 10.1002/cncr.20120. PMID: 15042689.
- Keiser PB, Nutman TB. Strongyloides stercoralis in the Immunocompromised Population. Clin Microbiol Rev. 2004;17(1):208-217. doi:10.1128/cmr.17.1.208-217.2004
- Vasquez-Rios G, Pineda-Reyes R, Pineda-Reyes J, Marin R, Ruiz EF, Terashima A. Strongyloides stercoralis hyperinfection syndrome: a deeper understanding of a neglected disease. J Parasit Dis. 2019 Jun;43(2):167-175. doi: 10.1007/s12639-019-01090-x. Epub 2019 Feb 6. PMID: 31263320; PMCID: PMC6570730.
- Linder JD, Mönkemüller KE, Lazenby AJ, Wilcox CM. Streptococcus bovis bacteremia associated with Strongyloides stercoralis colitis. Gastrointest Endosc. 2000;52:796-798.
- Smallman LA, Young JA, Shortland-Webb WR, Carey MP, Michael J. Strongyloide stercoralis hyperinfestation syndrome with Escherichia coli meningitis: report of two cases. J Clin Pathol. 1986;39:366-370.
- Barros, N., Montes, M. Infection and Hyperinfection with Strongyloides stercoralis: Clinical Presentation, Etiology of Disease, and Treatment Options. Curr Trop Med Rep 1, 223–228 (2014)
- González A, Gallo M, Valls ME, Muñoz J, Puyol L, Pinazo MJ, Mas J, Gascon J. Clinical and epidemiological features of 33 imported Strongyloides stercoralis infections. Trans R Soc Trop Med Hyg. 2010 Sep;104(9):613-6. doi: 10.1016/j.trstmh.2010.06.001. Epub 2010 Jul 16. PMID: 20637483.
- Igra-Siegman Y, Kapila R, Sen P, Kaminski ZC, Louria DB. Syndrome of hyperinfection with Strongyloides stercoralis. Rev Infect Dis. 1981 May-Jun;3(3):397-407. doi: 10.1093/clinids/3.3.397. PMID: 7025145.
- Vaiyavatjamai P, Boitano JJ, Techasintana P, Tungtrongchitr A. Immunocompromised group differences in the presentation of intestinal strongyloidiasis. Jpn J Infect Dis. 2008 Jan;61(1):5-8. PMID: 18219126.
- Mejia R, Nutman TB. Screening, prevention, and treatment for hyperinfection syndrome and disseminated infections caused by Strongyloides stercoralis. Curr Opin Infect Dis. 2012 Aug;25(4):458-63. doi: 10.1097/QCO.0b013e3283551dbd. PMID: 22691685; PMCID: PMC3430846.
- Geri G, Rabbat A, Mayaux J, Zafrani L, Chalumeau-Lemoine L, Guidet B, Azoulay E, Pène F. Strongyloides stercoralis hyperinfection syndrome: a case series and a review of the literature. Infection. 2015 Dec;43(6):691-8. doi: 10.1007/s15010-015-0799-1. Epub 2015 May 26. PMID: 26008854.