Enterococcal infections and bacteremia incidence have been increasing over the past decades, with enterococcal endocarditis representing 10% of all cases of infective endocarditis (1). This makes it the third most common organism implicated in IE, after Staphylococcus and streptococcus, and it is more prevalent in patients who are not drug abusers. Enterococcal bacteremia by itself has not been found to be associated with higher mortality, according to a prospective matched case control study, which found the crude mortality rate of 23% in enterococcal bacteremia to be not statistically different from a control group mortality of 17% (2). Infective endocarditis, however, carries a significant 1-year mortality rate of up to 28.9%, compared to that of other streptococci such as oral streptococci (14.6%) or group D streptococci (17.8%; 3).
TEE is one of the most sensitive modalities to evaluate for infective endocarditis but this is not always available or carries high risk in certain patient populations. As a result, clinical tools and scoring systems have emerged to risk stratify patients and avoid unneeded TEEs. A multicenter prospective study evaluated the risk factors for enterococcal bacteremia in 344 patients who had echocardiography performed (4). The prevalence in this cohort was 26.1%. Predictive factors for IE were prosthetic heart valve, community acquisition, >3 positive blood cultures, unknown source, and immunosuppression:
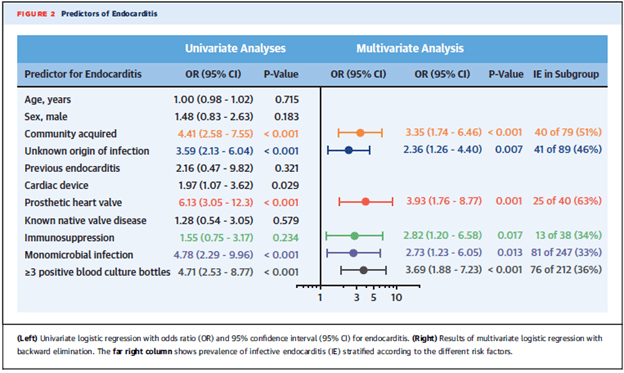
Given findings similar to these, a scoring system was developed to identify patients at low risk of IE and who did not require TEE. A single-center prospective cohort study (5) evaluated 1515 episodes of enterococcal bacteremia, of which 388 underwent TEE. IE was diagnosed in 65 patients (4.29% of all patients) and these cases were compared to 65 controls to identify variables associated with enterococcal endocarditis. These variables were analyzed in a logistic regression model and validated with bootstrapping. The multivariate analysis found that blood culture positivity in 3 cultures, history of heart valve disease, and unknown source of bacteremia were associated with the development of endocarditis and these variables were used to develop a 12-point scoring system, known as the NOVA score:

The higher the score, the more likely the probability of enterococcal endocarditis, as follows: 5 points, 23%; 6 points, 45.5%, 7 points 82.4%, 8 points, 66.7%, 9 points, 60%, 10 points 100%, 11 points 83.3%, 12 points, 90%. This score was validated with bootstrapping, achieving an ROC of 0.829 (95% CI 0.758-0.901). Notably, no patients with a cut-off value <4 had IE:

The authors estimate that the percentage of patients who could have avoided TEE ranged from 14.6% in a high prevalence population up to 27.7% in a low prevalence population:

The NOVA score was externally validated in a multicenter Danish study (6), though it was modified to have 2 out of 2 positive cultures as opposed to 3 to fit the practice. 647 patients were evaluated, of which 240 underwent echocardiography (235 had TTE and 116 had TEE). 78 of these were found to have enterococcal endocarditis by the modified Duke criteria. Univariate analysis found that male sex, community acquisition, known valvular disease, pacemaker, prior IE, prosthetic heart valve, and unknown source to be associated with IE.
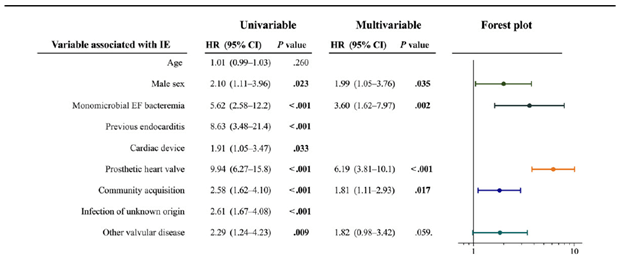
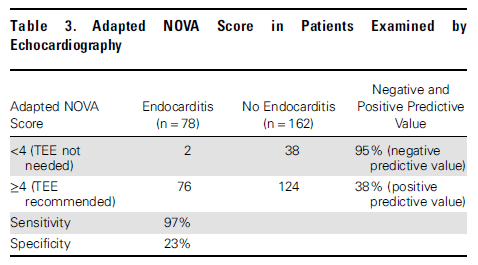
When applied, the NOVA score yielded a sensitivity of 97% and a specificity of 23%. Only 2 patients whose score were <4 had IE, one of them had only 1 blood culture performed and remained symptomatic, while the other one had only one culture set of 3 return positive, though he was a former IV drug user with prior IE.
An updated score was proposed in multicenter retrospective cohort (7). One cohort was used for calibration while another one was used for validation. The first cohort included 362 patients, of which 44 fulfilled criteria for infective endocarditis. Multivariate analysis found that symptom duration, number of positive blood cultures, unknown source of bacteremia, and heart murmur were associated with infective endocarditis.

Given these findings, duration of symptoms >7 days and embolization were added to the existing NOVA score to create the DENOVA score, where each variable was given one point a piece. In the calibration cohort, the ROC for DENOVA had an AUC of 0.95 (95% CI 0.94-0.97) for a cut-off value of >3 compared to an AUC of 0.91 (95% CI 0.89-0.95) for cutoff of >4.
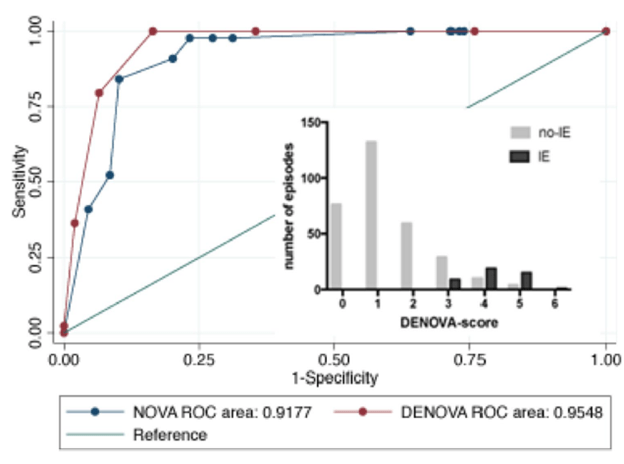
Sensitivity for DENOVA was 100% and specificity was 83% compared to the NOVA, which had a sensitivity of 100% and specificity of 29%. In the validation cohort, 252 patients were evaluated, with 26 being classified and IE. The AUC of the DENOVA score in the validation cohort was 0.95 (95% CI 0.93-0.98) compared to the NOVA with AUC of 0.92 (95% CI 0.88-0.95), with sensitivity and specificity being 100% and 85% for the DENOVA score in the validation cohort and NOVA score having a sensitivity and specificity of 100% and 35% respectively.
TL;DR
- NOVA score is a useful tool for figuring out who to get a TEE
- In combination with clinical judgement, it may avoid unnecessary TEE
- While there is a concern for sensitivity here, remember that sustained bacteremia is worth 5 points, so that buys you a TEE by this score. It is useful for those cases where you are not quite sure (maybe you heard a murmur and there is no obvious source of bacteremia).
References:
- Murdoch DR, Corey GR, Hoen B, Miró JM, Fowler VG Jr, Bayer AS, Karchmer AW, Olaison L, Pappas PA, Moreillon P, Chambers ST, Chu VH, Falcó V, Holland DJ, Jones P, Klein JL, Raymond NJ, Read KM, Tripodi MF, Utili R, Wang A, Woods CW, Cabell CH; International Collaboration on Endocarditis-Prospective Cohort Study (ICE-PCS) Investigators. Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: the International Collaboration on Endocarditis-Prospective Cohort Study. Arch Intern Med. 2009 Mar 9;169(5):463-73. doi: 10.1001/archinternmed.2008.603. PMID: 19273776; PMCID: PMC3625651.
- Caballero-Granado FJ, Becerril B, Cuberos L, Bernabeu M, Cisneros JM, Pachón J. Attributable mortality rate and duration of hospital stay associated with enterococcal bacteremia. Clin Infect Dis. 2001 Feb 15;32(4):587-94. doi: 10.1086/318717. Epub 2001 Feb 7. PMID: 11181122.
- Chirouze C, Athan E, Alla F, Chu VH, Ralph Corey G, Selton-Suty C, Erpelding ML, Miro JM, Olaison L, Hoen B; International Collaboration on Endocarditis Study Group. Enterococcal endocarditis in the beginning of the 21st century: analysis from the International Collaboration on Endocarditis-Prospective Cohort Study. Clin Microbiol Infect. 2013 Dec;19(12):1140-7. doi: 10.1111/1469-0691.12166. Epub 2013 Mar 20. PMID: 23517406.
- Dahl A, Iversen K, Tonder N, Hoest N, Arpi M, Dalsgaard M, Chehri M, Soerensen LL, Fanoe S, Junge S, Hoest U, Valeur N, Lauridsen TK, Fosbol E, Hoi-Hansen T, Bruun NE. Prevalence of Infective Endocarditis in Enterococcus faecalis Bacteremia. J Am Coll Cardiol. 2019 Jul 16;74(2):193-201. doi: 10.1016/j.jacc.2019.04.059. PMID: 31296291.
- Bouza E, Kestler M, Beca T, Mariscal G, Rodríguez-Créixems M, Bermejo J, Fernández-Cruz A, Fernández-Avilés F, Muñoz P; Grupo de Apoyo al Manejo de la Endocarditis. The NOVA score: a proposal to reduce the need for transesophageal echocardiography in patients with enterococcal bacteremia. Clin Infect Dis. 2015 Feb 15;60(4):528-35. doi: 10.1093/cid/ciu872. Epub 2014 Nov 7. Erratum in: Clin Infect Dis. 2015 May 21;: PMID: 25381321.
- Dahl A, Lauridsen TK, Arpi M, Sørensen LL, Østergaard C, Sogaard P, Bruun NE. Risk Factors of Endocarditis in Patients With Enterococcus faecalis Bacteremia: External Validation of the NOVA Score. Clin Infect Dis. 2016 Sep 15;63(6):771-5. doi: 10.1093/cid/ciw383. Epub 2016 Jun 15. PMID: 27307506.
- Berge A, Krantz A, Östlund H, Nauclér P, Rasmussen M. The DENOVA score efficiently identifies patients with monomicrobial Enterococcus faecalis bacteremia where echocardiography is not necessary. Infection. 2019 Feb;47(1):45-50. doi: 10.1007/s15010-018-1208-3. Epub 2018 Sep 3. PMID: 30178077.
