Beta-Lactamases make my head hurt. They are so many of them and they impact the therapeutic options for a lot of the infections that we treat. Due to this resistance mechanism, there has been an influx of new beta-lactam/beta-lactamase combinations (ceftazidime-avibactam, meropenem-vaborbactam, etc) to overcome this phenomena.
So first, this is a beta-lactam:
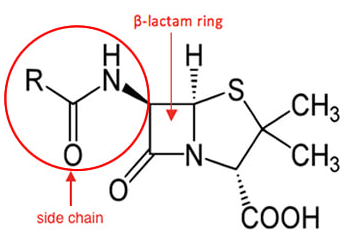
The penicillin-binding protein (which creates part of the cell-wall) thinks the ring is part of its building block to make that cell wall, but once it binds the beta-lactam ring, it cannot proceed further. There is no more cell wall synthesis -> big bug dies. Beta-lactamases hydrolyze this ring. Because of this, there is no longer a threat to the PBP, so it keeps doing its job. Some beta-lactamase inhibitors have a similar structure (the beta-lactam ring), so these beta-lactamases pick up the beta-lactamase inhibitors (confusing, I know) and the beta-lactam ring gets to work binding the PBP.
We need to distinguish between the different types of resistance mechanisms, as the BL/BLI combinations do not help in those other situations (1, 2, 3):
- Changes to the penicillin binding protein that lowers the affinity of Beta-lactams to work. This is how Strep pneumonia can develop resistance. One other notable bug is MRSA, as the mecA gene produces PBP2a which has lower affinity for penicillins and cephalosporins.
- Decreased expression of outer membrane proteins – essentially decreases the ability for antibiotics to diffuse through the porin channels.
- Efflux pumps – this is how MDR occurs in organisms such as Acinetobacter and Pseudomonas.
- Beta-lactamase – as the name suggest, these are enzymes that allow the hydrolyzation of beta-lactams, allowing resistance to these antibiotics.
While both gram positive and gram negative organisms can produce beta lactamses, these are frequently encountered in gram negative infections. One of the most well known gram positive beta-lactamases is penicillinase, which is encoded by the blaZ gene and is encountered in 86.5% of all Staph aureus isolates (2). This is the reason that penicillin cannot be used for MSSA, but the anti-staphylococcal beta lactams can still be used. The presence of beta-lactamases in bacteria is nothing new, in fact they’re probably older than humanity itself. For instance, one review noted that the estimated age of serine beta-lactamases is more than 2 billion years old, with plasmid encoded beta-lactamases appearing millions of years ago. It is with selective pressure that these have become prominent.
There are many beta-lactamases that have been encountered over the years, and the amount is staggering, with more being identified with each passing year:
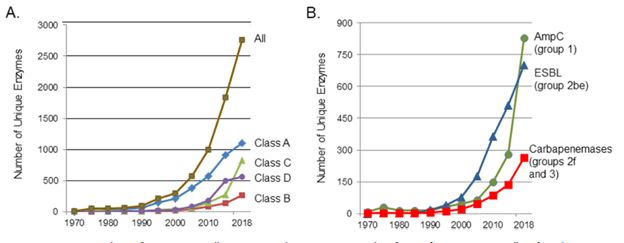
A lot of reviews tend to focus on the biochemical properties as well as the mechanism of actions (trust me, these are a painful read if this is not your interest), so I will try to give you the big picture idea to get you comfortable with this concept.
Classification:
There are 2 general classification schemes for beta-lactamses (1,3). The first one is the Ambler classification, which classifies beta-lactamases based on their amino acid sequence. It is not terribly helpful clinically. The second classification scheme is known as the Bush-Jacoby-Medeiros classification, and its based on substrate and inhibitor profile, which is a bit more helpful to keep things straight to a certain extent. In general, there are 2 broad types of beta-lactamases, based on their mechanism of action:
- The serine beta-lactamases (class A, C, and D)
- The Metallo-beta-lactamse (class B), which uses one or 2 Zinc ions.
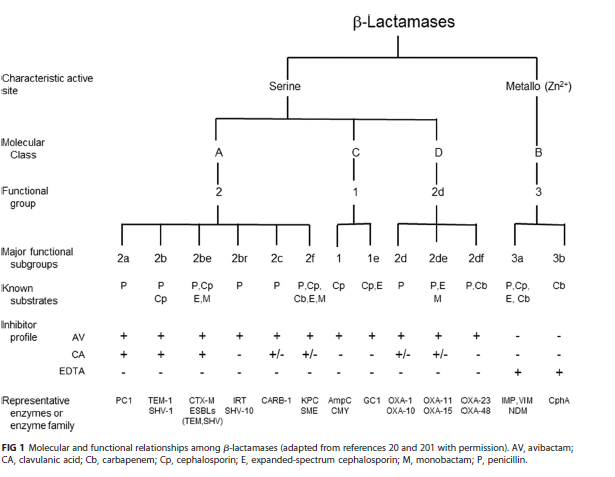
Grouping beta-lactamases based on the Bush Jacoby-Medeiros classification yields a scheme like this (5):
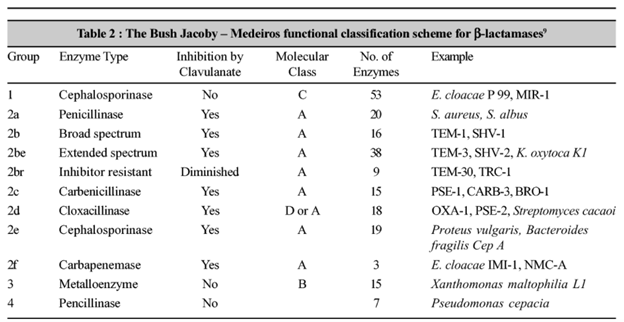
You will notice a few things. First, group 1 and group 4 have relatively narrow substrates. Group 2 essentially hits all major beta-lactams (2a – the first antibiotic, penicillin, 2b – b for broad spectrum, e for extended spectrum , if that helps) and almost all are inhibited by Clavulanate. Another way to put it:
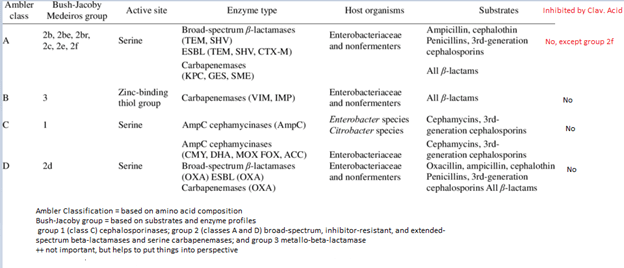
If you need a way to remember, group A is inhibited by Clavulanic acid (look at the resistance patterns). The second generation cephalosporins are resistant in presence of group C beta-lactamases (i.e. cefotetan, cefoxitin; this becomes important later). Further,
- Class A -> group 2
- Class B -> group 3
- Class C -> group 1
- Class D -> group 2d
Class A beta-lactamases (group 2, except 2d) – usually inhibited by most beta-lactamse inhibitors, including clavulanate, tazobactam, and to a lesser degree sulbactam. They are generally found in plasmids and can be encoded in chromosomes or integrons. The number of inhibitor needed for each type of beta-lactamase varies, which can be measured by the turnover number (number of inhibitor molecules hydrolyzed per unit time by one enzyme). For instance, sulbactam’s turnover number is 10,000 for TEM-1 and 13,000 for SHV-1, respectively. These also include the ESBL beta-lactamases, which hydrolyze penicillins, aztreonam, and most cephalosporins (with one exception).
Before moving on, we need to define extended-spectrum beta-lactams. These refer to the 3rd and 4th generation cephalosporins, which include ceftriaxone, ceftazidime, and cefepime. So ESBL beta-lactamases hydrolyze: penicillins, first generation cephalosporins, third and fourth generation cephalosporins, and aztreonam. Anything missing here?
ESBL cannot hydrolyze
- Cephamycins
- Carbapenems
- Beta-lactamase inhibitors
Within this group, there is also carbapenemases, though this is not the more well-known so I won’t speak on it in this review, but you are welcome to review the first 4 sources if you want more details about this.
- Clinically important beta-lactamases
- TEM, SHV, CTX-M – found in E.coli and Klebsiella spp
- KPC, GES, Nmc-A/IMI, SME – carbapenemases. The most important being KPC, encoded by blaKPC gene.
Class B beta-lactamases (group 3) – aka the zinc-dependent metallo-beta-lactamases. These are typically very broad spectrum, meaning they hydrolyze penicillins, cephalosporins, and carbapenems. One exception is aztreonam, which has hydrolytic stability. This class includes the new kid on the block, New Delhi MBL (metallo-beta-lactamse), which induces resistance to carbapenems (encoded by blaNDM–1)
Class C beta-lactamases (group 1) – these also known as the AmpC beta-lactamases, which are plasmid-borne. These organisms tend to be resistant to penicillins, most cephalosporins, and noticeably beta-lactamase inhibitors such as clavulanate and tazobactam. One exception is the relatively new (well probably old but you know, medicine moves fast) avibactam. The main characteristic of this class is they tend to be “repressed” so it won’t show up in susceptibility testing. These are induced, typically by things such as cefazolin, amoxicillin, ampicillin, cefoxitin, and imipenem. Therapy with these antibiotics will lead to the “induction” of ampC beta-lactamases and subsequent resistance. One exception is cefepime, which is poorly hydrolyzed by ampC and is actually more stable.
AmpC Producers:
- Strong inducers: benzylpenicillin, ampicillin, amoxicillin, cefazolin, cefoxitin, imipenem, clavulonate (yes, really).
- For instance, a pharmacodynamic study (10) found that out of 10 clinical isoaltes of PseA, AmpC expression was seen in 8 samples, compared to tazobactam which failed to induce AmpC.
- Another study (11) found that clavulonate, but not tazobactam or sulbactam, was able to induce beta-lactamases in Enterobacter spp, Pseudomonas, Serratia marcescens, Citrobacter freundii at high concentrations.
- Weak inducers: cefoxatime, ceftriaxone, ceftazidime, cefepime, cefuroxime, piperacillin, aztreonam
Class D beta-lactamases – also known as oxacillinases, with OXA enzymes being able to act as ESBL and carbapenamases. These prefer to hydrolyze oxacillin (hence the name), but several of them are able to hydrolyze carabapenems, including OXA-23, OXA-24/40, OXA-48, OXA-143, and OXA-51. A nice (albeit overly complicated) review is available (7).
Beta-Lactamase Inhibitors
The original 3 BLI include clavulanic acid, sulbactam, and tazobactam and these have activity against class A enzymes, with the notable exception being KPC, which also belongs in class A. Further, these have no activity against class B, C, or D beta-lactamase. As a result, the diazabicyclooctanones (DBOs, whatever that means) were created which gave us Avibactam. This is a non-beta lactam, beta-lactamse inhibitor and is able to inhibit class C and A beta lactamases, including KPC. Another DBO down the pipeline is zidebactam (undergoing clinical trials as of the time of writing this). The other family of BLI that have been released into the market have been the boronate-baseed compounds, of which Vaborbactam is a part of. It is able to inhibit class A, including KPC, but has no activity to class C or D.
One beta-lactamase class that none of the above is able to tackle is class B, of which NDM-1 is part of. You may have heard of this one, as it is one of the beta-lactamases that confers resistance to a large number of beta lactams with the exception of aztreonam and cefidericol. An experimental rat model of P. aeruginosa COL-1 (which was a class B BL) demonstrated high MIC to imipenem, cefepime, ceftazdime, while being susceptible to aztreonam and moderately susceptible to pip-tazo (8). Unfortunately, we do not have any beta-lactamase inhibitor against this class of BLs (though there are some therapeutic options, see later).
ampC Production
As mentioned previously, these beta-lactamases have the feature of being “repressed” and can get “expressed” under the right circumstances. The mechanism of the “de-repression” is beyond me, but you can review it if you so choose (6). The presence of beta-lactam therapy confers a stimulus that leads to the expression of ampC. The most potent inducers include the aminopenicillins, narrow-spectrum cephalosporins, and the cephamycins. Weak inducers include piperacillin-tazobactam, aztreonam, and the extended-spectrum cephalosporins. Despite this distinction, some AmpC producers such as E. cloacae complex, C. feundii, and S. marcesens hydrolyze the strong inducers at basal levels (i.e. before beta-lactam exposure).
ampC genes have been identified in gram negative organisms, which are known as the SPACE-M organisms:
- Serratia marcescens
- Providentia stuartii/Pseudomonas aeruginosa
- Acinetobacter
- Citrobacter freundii
- Enterobacter cloacae
- Morganella morganii
Despite this, not all organisms within this cluster express AmpC homogeneously. For instance, de-repressed Serratia, Morganella, and Providentia strains express AmpC levels that are roughly 10 fold lower than de-repressed E. cloacae complex or Citrobacter isolates. This leads to some interesting treatment options for these organisms. For instance, since cefepime tends to be more stable and less likely to get hydrolyzed by AmpC producers, this is a reasonable treatment option for infections caused by Morganella spp.
ESBL – Extended Spectrum Beta Lactamases
You may have heard of this phrase. As I mentioned before, the extended-spectrum beta-lactams refer to the third and fourth generation cephalosporins. These beta-lactamases confer resistance to these, including aminopenicillins and the narrow-spectrum beta-lactams, but retain susceptibility to carbapenems (except for some of the forthcoming carbapenem resistant organisms).
How are you able to differentiate between AmpC producers and ESBL? There are several microbiological options, such as the disk potentiation test, double-disk synergy test, and using an ETEST with inhibitors and look for deformation of the ellipse of inhibition, known as the phantom zone. From a clinical perspective, it is helpful to look at the susceptibility pattern. There are a few ways to differentiating AmpC and ESBL:
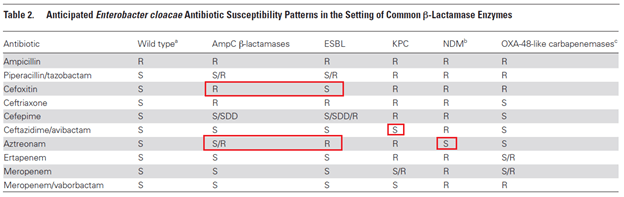
- AmpC producers are resistant to cephamycins aka second generation cephalosporins (look for an R in cefoxitin)
- ESBL have high MIC to cefepime and can at times be resistant
- Aztreonam is resistant in ESBL
Clinical Data:
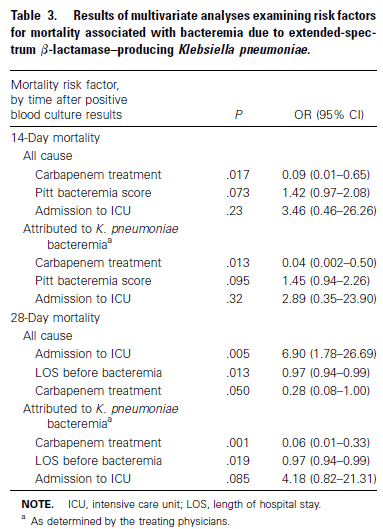
How do you approach therapy for ESBLs or AmpC producers? In general, therapy for ESBLs with “susceptible” cephalosporins are not a good idea. In a retrospective cohort (12) half of the patients with ESBL infections who were treated with a cephalosporin failed therapy. Further, they analyzed 36 patients in the literature and 54% of these had clinical failure when treated with a cephalosporin. A prospective, observational study (13) evaluated therapy for culture positive with ESBL-producing K. pneumoniae, comparing carbapenems with non-carbapenems (primarily, ciprofloxacin, BL/BLI, amikacin). Use of carabpenems was associated with a lower 14 day mortality due to ESBL-producing K pneumoniae bacteremia (4.8% vs 27.6%, OR 0.173, 95% CI 0.039-0.755). Further, carbapenem use was independently associated with lower 14 and 28 day mortality in a multivariate analysis:
Compared to carbapenems, quinolones tend to fare worse. One reasonably sized retrospective study (14) found that carbapenems resulted in favorable response in 8 out of 10 cases of ESBL-producing K. pneumoniae compared to 2 out of 7 patients in the ciprofloxacin group, though this was a small cohort. A post-hoc analysis (15) evaluated the outcomes of patients with ESBL-E.coli bacteremia treated with BL/BLI or carbapenems from 6 prospective cohort studies. 287 patients were included, 103 included in the empirical therapy cohort and 174 included in the definitive therapy cohort. In empirical therapy, mortality at day 30 for BL/BLI was 5.9% vs 9.4% for those whose therapy was changed to a carbapenem (p-value 0.6). In the definitive therapy cohort, mortality at day 14 was 5.6% for the BL/BLI group vs 11.7% for the carbapenem group, and 9.3% for the BL/BLI group vs 16.7% for the carbapenem group at day 30 (p >0.2). Cox regression analysis did not find empirical or definitive therapy with BL/BLI to be associated with any difference in outcomes in comparison to carbapenems.
These data, however is prospective. The large Merino trial (16), which was a multicenter, randomized, open-label trial evaluated meropenem with piperacillin-tazobactam in CTX-non susceptible K. pneumoniae and E.coli. Primary outcome was all-cause 30 day mortality, with 378 patients being randomized (trial was stopped due to a significant difference in the primary outcome being observed). 191 patients got meropenem vs 188 for piperacillin-tazobactam. Primary outcome was met in 12.3% of the pip-tazo group (23/187) compared to 3.7% in the meropenem group (7/191; risk difference 8.6%, p-value 0.9 for non-inferiority).
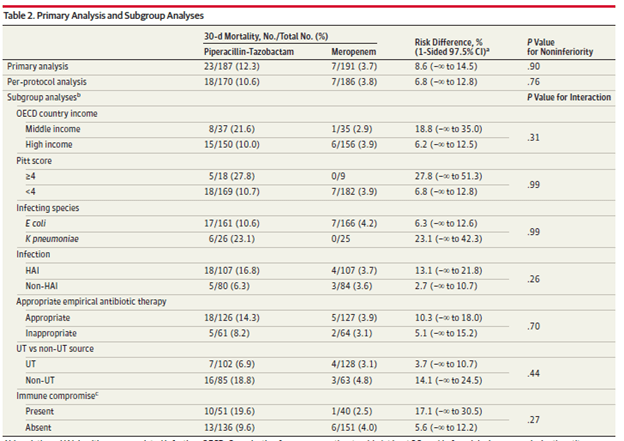
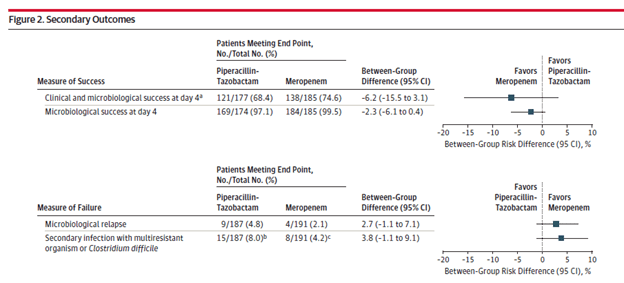
Secondary outcomes tended to favor meropenem, but these did not reach statistical significance.
Given these data, first line therapy for ESBL-producing gram negative infections (especially bacteremias) should be carbapenems. This seems to be a class effect. For instance, 2 studies evaluated the efficacy of ertapenem (the one carbapenem that doesn’t cover pseudomonas) in ESBL-producing Enterobacteriaceae infections. The retrospective INCREMENT project (17) evaluated clinical response rate at day 14 and all-cause mortality at day 30 in patients who were treated with either ertapenem or another carbapenem for ESBL Enterobacteriacae bacteremia. In the empiric cohort, cure was seen at day 14 in 90.6% of ertapenem patients vs 75.5% of other carbapenems (p-value 0.06, absolute difference 15.1%, 95% CI 1.9-25.9%). In the targeted therapy cohort, cure rates were 89.8% in the ertapenem group vs 82.6% in the other carbapenem group (p-value 0.02, absolute difference 7.2%, 95% CI 1.2-13.14%). Multivariate analysis did not show ertapenem being associated with cure or improvement at day 14 compared to other carbapenems (OR 1.04, 95% CI 0.44-2.50). Mortality between those who got ertapenem or other carbapenems was not statistically different at 30 days.
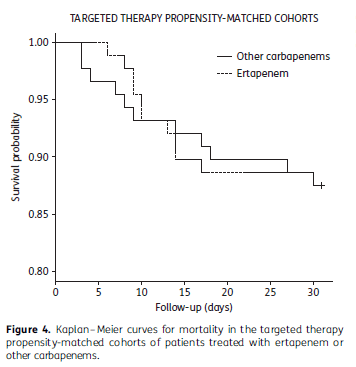
In another retrospective study (18) evaluating therapy for ESBL E.coli and K. pneuomniae, multivariate analysis did not show that the type of carbapenem was associated with increased mortality (aOR 0.26, p-value 0.12) and after controlling for propensity score, ertapenem was not associated with increased risk of death (OR 0.5, 95% CI 0.12 to 2.1). This all suggest that any carbapenem is a good option for ESBL bacteremias. Which is nice to have a good once a day carbapenem for possible OPAT.
New Delhi Metallo-beta-lactamases
I mentioned before that NDM-1, the class B beta-lactamases, is not able to be inhibited by the original 3 beta-lactamase inhibitors or even avibactam. Aztreonam is the only beta-lactam that is susceptible to NDMs. As a result, we have very little in terms of treating these gram negative infections. One combination that has been proposed has been aztreonam + ceftazidime/avibactam. The combination has show to be synergistic in isolates of NDM-producing K. pneumonaie (19) and one retrospective analysis of a particularly nasty K. pneumoniae (NDM-1 + OXA 48 + CTX-M-15) which affected 10 patients found that the combination was successful in 6 out patients (20). Time-kill curves of an MBL-containing K. pneumoniae isolate (21) found that the combination of aztreonam and ceftaz/avibactam at different CAZ/AVI combination resulted in better bacterial killing than CAZ/AVI alone:
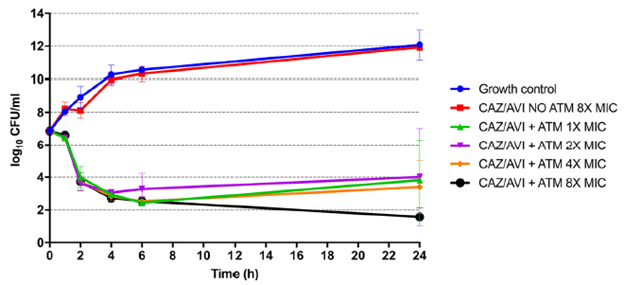
Further, in the same study, using an in vivo mice thigh model, the combination was found to have an almost four-fold reduction in bacterial load at a lower dose of CAZ/AVI in combination compared to CAZ/AVI alone:

Beyond this, other antibiotics that have been proposed have been cefidericol, a polymyxin with a second-active agent such as meropenem if the MIC permits or tigecycline. For instance, one study evaluated 16 isolates of carbapenem-resistant K. pneumoniae which found susceptibility of Polymyxin B and Tigecycline (22). Another study evaluated carbapenem-resistant Enterobacteriacae and A. baumannii isolates and found that both tigecycline and eravacycline were more active for these organisms, with eravacycline being 2 to 4-fold more active than tigecycline (23). a non-inferiority, randomized control trial (24) evaluated the safety and efficacy of eravacycline compared to ertapenem for complicated intra-abdominal infections. In the mITT population, clinical cure rates were 87% (235/270) for eravacycline compared to 88.8% (238/268) for ertapenem (difference -1.80%, 95% CI -7.4 to 3.8%).
Appendix:
An additional way to picture the classification
References:
- Bush K, Jacoby GA. Updated functional classification of beta-lactamases. Antimicrob Agents Chemother. 2010 Mar;54(3):969-76. doi: 10.1128/AAC.01009-09. Epub 2009 Dec 7. PMID: 19995920; PMCID: PMC2825993.
- Bush K. Past and Present Perspectives on β-Lactamases. Antimicrob Agents Chemother. 2018 Sep 24;62(10):e01076-18. doi: 10.1128/AAC.01076-18. PMID: 30061284; PMCID: PMC6153792.
- Drawz SM, Bonomo RA. Three decades of beta-lactamase inhibitors. Clin Microbiol Rev. 2010 Jan;23(1):160-201. doi: 10.1128/CMR.00037-09. PMID: 20065329; PMCID: PMC2806661.
- Bonomo RA. β-Lactamases: A Focus on Current Challenges. Cold Spring Harb Perspect Med. 2017 Jan 3;7(1):a025239. doi: 10.1101/cshperspect.a025239. PMID: 27742735; PMCID: PMC5204326.
- Chaudhary U, Aggarwal R. Extended spectrum -lactamases (ESBL) – an emerging threat to clinical therapeutics. Indian J Med Microbiol. 2004 Apr-Jun;22(2):75-80. PMID: 17642700.
- Tamma PD, Doi Y, Bonomo RA, Johnson JK, Simner PJ; Antibacterial Resistance Leadership Group. A Primer on AmpC β-Lactamases: Necessary Knowledge for an Increasingly Multidrug-resistant World. Clin Infect Dis. 2019 Sep 27;69(8):1446-1455. doi: 10.1093/cid/ciz173. PMID: 30838380; PMCID: PMC6763639.
- Tamma PD, Doi Y, Bonomo RA, Johnson JK, Simner PJ; Antibacterial Resistance Leadership Group. A Primer on AmpC β-Lactamases: Necessary Knowledge for an Increasingly Multidrug-resistant World. Clin Infect Dis. 2019 Sep 27;69(8):1446-1455. doi: 10.1093/cid/ciz173. PMID: 30838380; PMCID: PMC6763639.
- Bellais S, Mimoz O, Léotard S, Jacolot A, Petitjean O, Nordmann P. Efficacy of beta-lactams for treating experimentally induced pneumonia due to a carbapenem-hydrolyzing metallo-beta-lactamase-producing strain of Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2002 Jun;46(6):2032-4. doi: 10.1128/aac.46.6.2032-2034.2002. PMID: 12019134; PMCID: PMC127240.
- Tooke CL, Hinchliffe P, Bragginton EC, Colenso CK, Hirvonen VHA, Takebayashi Y, Spencer J. β-Lactamases and β-Lactamase Inhibitors in the 21st Century. J Mol Biol. 2019 Aug 23;431(18):3472-3500. doi: 10.1016/j.jmb.2019.04.002. Epub 2019 Apr 5. PMID: 30959050; PMCID: PMC6723624.
- Lister PD, Gardner VM, Sanders CC. Clavulanate induces expression of the Pseudomonas aeruginosa AmpC cephalosporinase at physiologically relevant concentrations and antagonizes the antibacterial activity of ticarcillin. Antimicrob Agents Chemother. 1999 Apr;43(4):882-9. doi: 10.1128/AAC.43.4.882. PMID: 10103195; PMCID: PMC89221.
- Weber DA, Sanders CC. Diverse potential of beta-lactamase inhibitors to induce class I enzymes. Antimicrob Agents Chemother. 1990 Jan;34(1):156-8. doi: 10.1128/aac.34.1.156. PMID: 2327752; PMCID: PMC171539.
- Paterson DL, Ko WC, Von Gottberg A, Casellas JM, Mulazimoglu L, Klugman KP, Bonomo RA, Rice LB, McCormack JG, Yu VL. Outcome of cephalosporin treatment for serious infections due to apparently susceptible organisms producing extended-spectrum beta-lactamases: implications for the clinical microbiology laboratory. J Clin Microbiol. 2001 Jun;39(6):2206-12. doi: 10.1128/JCM.39.6.2206-2212.2001. PMID: 11376058; PMCID: PMC88112.
- Paterson DL, Ko WC, Von Gottberg A, Mohapatra S, Casellas JM, Goossens H, Mulazimoglu L, Trenholme G, Klugman KP, Bonomo RA, Rice LB, Wagener MM, McCormack JG, Yu VL. Antibiotic therapy for Klebsiella pneumoniae bacteremia: implications of production of extended-spectrum beta-lactamases. Clin Infect Dis. 2004 Jul 1;39(1):31-7. doi: 10.1086/420816. Epub 2004 Jun 8. PMID: 15206050.
- Endimiani A, Luzzaro F, Perilli M, Lombardi G, Colì A, Tamborini A, Amicosante G, Toniolo A. Bacteremia due to Klebsiella pneumoniae isolates producing the TEM-52 extended-spectrum beta-lactamase: treatment outcome of patients receiving imipenem or ciprofloxacin. Clin Infect Dis. 2004 Jan 15;38(2):243-51. doi: 10.1086/380645. Epub 2003 Dec 19. PMID: 14699457.
- Rodríguez-Baño J, Navarro MD, Retamar P, Picón E, Pascual Á; Extended-Spectrum Beta-Lactamases–Red Española de Investigación en Patología Infecciosa/Grupo de Estudio de Infección Hospitalaria Group. β-Lactam/β-lactam inhibitor combinations for the treatment of bacteremia due to extended-spectrum β-lactamase-producing Escherichia coli: a post hoc analysis of prospective cohorts. Clin Infect Dis. 2012 Jan 15;54(2):167-74. doi: 10.1093/cid/cir790. Epub 2011 Nov 4. PMID: 22057701.
- Harris PNA, Tambyah PA, Lye DC, Mo Y, Lee TH, Yilmaz M, Alenazi TH, Arabi Y, Falcone M, Bassetti M, Righi E, Rogers BA, Kanj S, Bhally H, Iredell J, Mendelson M, Boyles TH, Looke D, Miyakis S, Walls G, Al Khamis M, Zikri A, Crowe A, Ingram P, Daneman N, Griffin P, Athan E, Lorenc P, Baker P, Roberts L, Beatson SA, Peleg AY, Harris-Brown T, Paterson DL; MERINO Trial Investigators and the Australasian Society for Infectious Disease Clinical Research Network (ASID-CRN). Effect of Piperacillin-Tazobactam vs Meropenem on 30-Day Mortality for Patients With E coli or Klebsiella pneumoniae Bloodstream Infection and Ceftriaxone Resistance: A Randomized Clinical Trial. JAMA. 2018 Sep 11;320(10):984-994. doi: 10.1001/jama.2018.12163. Erratum in: JAMA. 2019 Jun 18;321(23):2370. PMID: 30208454; PMCID: PMC6143100.
- Gutiérrez-Gutiérrez B, Bonomo RA, Carmeli Y, Paterson DL, Almirante B, Martínez-Martínez L, Oliver A, Calbo E, Peña C, Akova M, Pitout J, Origüen J, Pintado V, García-Vázquez E, Gasch O, Hamprecht A, Prim N, Tumbarello M, Bou G, Viale P, Tacconelli E, Almela M, Pérez F, Giamarellou H, Cisneros JM, Schwaber MJ, Venditti M, Lowman W, Bermejo J, Hsueh PR, Mora-Rillo M, Gracia-Ahulfinger I, Pascual A, Rodríguez-Baño J; REIPI/ESGBIS/INCREMENT Group. Ertapenem for the treatment of bloodstream infections due to ESBL-producing Enterobacteriaceae: a multinational pre-registered cohort study. J Antimicrob Chemother. 2016 Jun;71(6):1672-80. doi: 10.1093/jac/dkv502. Epub 2016 Feb 22. PMID: 26907184; PMCID: PMC4867097.
- Collins VL, Marchaim D, Pogue JM, Moshos J, Bheemreddy S, Sunkara B, Shallal A, Chugh N, Eiseler S, Bhargava P, Blunden C, Lephart PR, Memon BI, Hayakawa K, Abreu-Lanfranco O, Chopra T, Munoz-Price LS, Carmeli Y, Kaye KS. Efficacy of ertapenem for treatment of bloodstream infections caused by extended-spectrum-β-lactamase-producing Enterobacteriaceae. Antimicrob Agents Chemother. 2012 Apr;56(4):2173-7. doi: 10.1128/AAC.05913-11. Epub 2012 Jan 30. PMID: 22290982; PMCID: PMC3318320.
- Jayol A, Nordmann P, Poirel L, Dubois V. Ceftazidime/avibactam alone or in combination with aztreonam against colistin-resistant and carbapenemase-producing Klebsiella pneumoniae. J Antimicrob Chemother. 2018 Feb 1;73(2):542-544. doi: 10.1093/jac/dkx393. PMID: 29165563.
- Shaw E, Rombauts A, Tubau F, Padullés A, Càmara J, Lozano T, Cobo-Sacristán S, Sabe N, Grau I, Rigo-Bonnin R, Dominguez MA, Carratalà J. Clinical outcomes after combination treatment with ceftazidime/avibactam and aztreonam for NDM-1/OXA-48/CTX-M-15-producing Klebsiella pneumoniae infection. J Antimicrob Chemother. 2018 Apr 1;73(4):1104-1106. doi: 10.1093/jac/dkx496. PMID: 29272413.
- Marshall S, Hujer AM, Rojas LJ, Papp-Wallace KM, Humphries RM, Spellberg B, Hujer KM, Marshall EK, Rudin SD, Perez F, Wilson BM, Wasserman RB, Chikowski L, Paterson DL, Vila AJ, van Duin D, Kreiswirth BN, Chambers HF, Fowler VG Jr, Jacobs MR, Pulse ME, Weiss WJ, Bonomo RA. Can Ceftazidime-Avibactam and Aztreonam Overcome β-Lactam Resistance Conferred by Metallo-β-Lactamases in Enterobacteriaceae? Antimicrob Agents Chemother. 2017 Mar 24;61(4):e02243-16. doi: 10.1128/AAC.02243-16. PMID: 28167541; PMCID: PMC5365724.
- Bratu S, Tolaney P, Karumudi U, Quale J, Mooty M, Nichani S, Landman D. Carbapenemase-producing Klebsiella pneumoniae in Brooklyn, NY: molecular epidemiology and in vitro activity of polymyxin B and other agents. J Antimicrob Chemother. 2005 Jul;56(1):128-32. doi: 10.1093/jac/dki175. Epub 2005 May 25. PMID: 15917285.
- Livermore DM, Mushtaq S, Warner M, Woodford N. In Vitro Activity of Eravacycline against Carbapenem-Resistant Enterobacteriaceae and Acinetobacter baumannii. Antimicrob Agents Chemother. 2016 May 23;60(6):3840-4. doi: 10.1128/AAC.00436-16. PMID: 27044556; PMCID: PMC4879382.
- Solomkin J, Evans D, Slepavicius A, Lee P, Marsh A, Tsai L, Sutcliffe JA, Horn P. Assessing the Efficacy and Safety of Eravacycline vs Ertapenem in Complicated Intra-abdominal Infections in the Investigating Gram-Negative Infections Treated With Eravacycline (IGNITE 1) Trial: A Randomized Clinical Trial. JAMA Surg. 2017 Mar 1;152(3):224-232. doi: 10.1001/jamasurg.2016.4237. PMID: 27851857.

1 comments on “A Crash Course of Beta-Lactamases and Beta-Lactamase Inhibitors”