This is a difficult post to go over since there is so much data to comb through that will likely change as the years go by (as with any topic I write about, but more so about therapy for influenza). Stay tuned as the flu season comes upon us, as this post may be updated accordingly. I would recommend looking through the quick notes (i.e. the TL;DR), since it can get non-sensical at times.
Influenza is an infection that we have sort of become numb to in the past few years. Aside from the 2009 H1N1 pandemic, seasonal influenza is one of those things we just “accept” for a lack of a better term. Despite this, it still kills a substantial number of patients and flu vaccines are moderately effective, at best (more on that in a future post). In the past few years, baloxavir has been touted as a new antiviral to combat acute influenza, partly because the data on oseltamivir is a bit all over the place, though there are some reports on early resistance. There is data that suggests it may shorten the duration of illness, but not reduce mortality in certain patient populations. Further, it may play a role in shortening length of hospital stay and reducing incidence of bronchitis, but does not prevent lower respiratory tract infections (depending on the study). In all fairness, it can be daunting to look over all this data. I’ll try to summarize the big studies on oseltamivir for acute influenza and post-exposure prophylaxis. Though if you have read through this paragraph and you do not want to read through any more, the takeaway is this: it is likely better to prescribe oseltamivir in all hospitalized patients and those who are at high risk of severe influenza, as well as healthy adults who come in within 48hrs of symptoms.
During this post, I’ll be talking about the neuramidinase inhibitor, oseltamivir. It is thought to inhibit the ability of the virus to exit host cells and to reduce symptom duration by reducing viral load and spread (1). Oseltamivir phosphate is the pro-drug of oseltamivir carboxylate (not really important). Early data came from experimental human studies, where doses of 100mg daily and 100mg twice per day were found to have a significant impact in viral load in both treatment and prophylaxis scenarios in healthy adult volunteers (2):
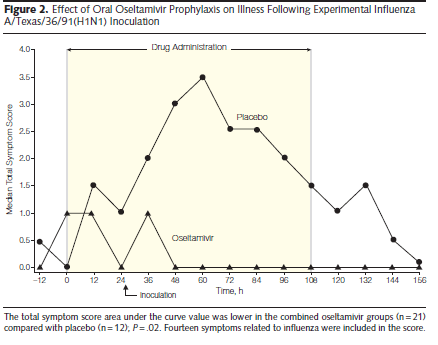
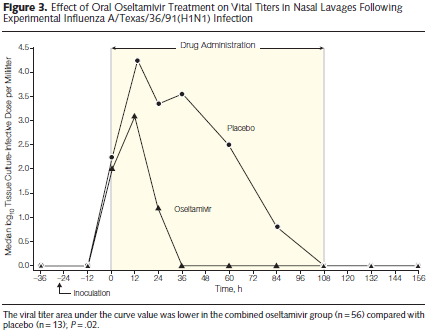
A follow up double-blind RCT, placebo controlled trial compared oseltamivir 75mg twice per day and 150mg twice per day given within 36 hours of illness onset in outpatients. In 629 patients who were randomized, oseltamivir resulted in a reduction of symptoms compared to placebo (103.3 hours in placebo vs 71.5 hours in the 75mg group and 69.9 hours in the 150mg group). This represents a reduction in the median symptom duration of illness by 30% (p-value 0.006) and median severity of illness by 40% (p-value <0.001).
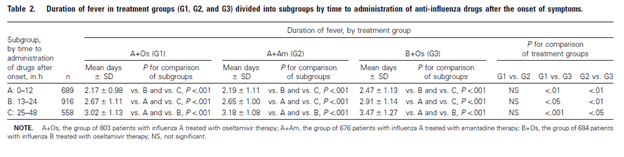
Early administration of oseltamivir seems to increase its benefits in treatment of influenza. For instance, an open-label, multicenter trial evaluated duration of symptoms as a function of time from treatment (4) found that earlier treatment was strongly associated with treatment duration in the outpatient setting:
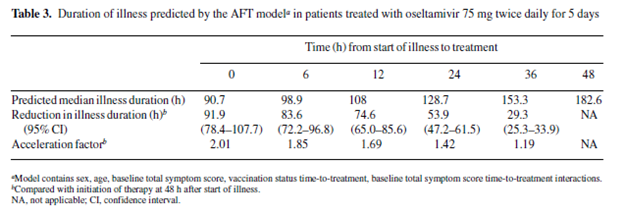
In another study (5), timing from symptom onset to initiation of therapy was associated with duration of fevers, with patients getting medication within 12 hours of symptom onset having shorter duration of fever compared to those getting drugs at 13-24hrs or 25-48hrs after onset:
Therapy with oseltamivir was also associated with lower rates of complications. For instance, a database that included 10 multicenter double-blind trials (6) that administered oseltamivir within n36 hours of presentation found that patients with influenza-confirmed cases and those who were influenza uninfected were more likely to have lower respiratory tract infection if they received placebo compared to oseltamivir, which was riven mostly by bronchitis:
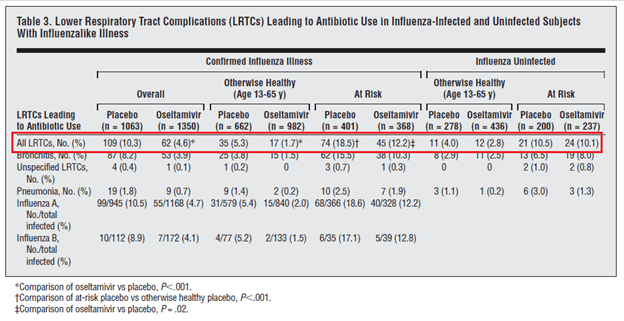
Though in this trial, there was no difference in hospitalization between groups.
In a multicountry data set of 308 patients with H5N1 influenza (7), oseltamivir treatment was found to be an independent variable in a cox regression analysis associated with improved mortality, HR 0.34 (95% CI 0.25-0.48). Further, survival difference between those who got oseltamivir vs those who didn’t was greatest if therapy was started within 2 days of symptom onset:
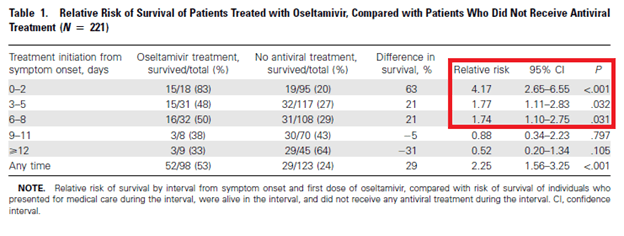
In a Canadian cohort of 327 adult patients with influenza (8), multivariate analysis found that oseltamivir therapy was associated with reduced mortality in lab-confirmed influenza cases that required hospitalization:

Though notably, absolute 15-day mortality was not different between the group who got oseltamivir and the group who did not (however, this was not controlled for anything).
Several meta-analyses have been performed to look at the efficacy of oseltamivir. The earliest meta-analysis performed in 2003 (9) evaluated 9 randomized control trials for time to alleviation of symptoms. They found that in a healthy population, the median difference in symptom alleviation between oseltamivir and placebo was 20.69 hours (95% CI 33.97 to 7.41), though in high risk there was no statistical significance.
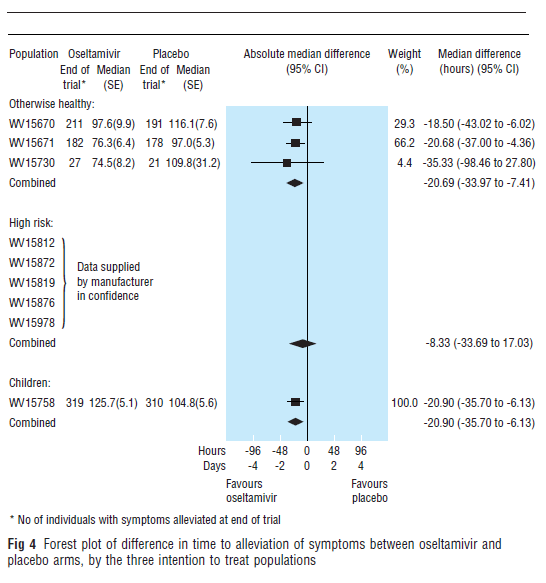
The same pattern held true even in flu-confirmed cases.
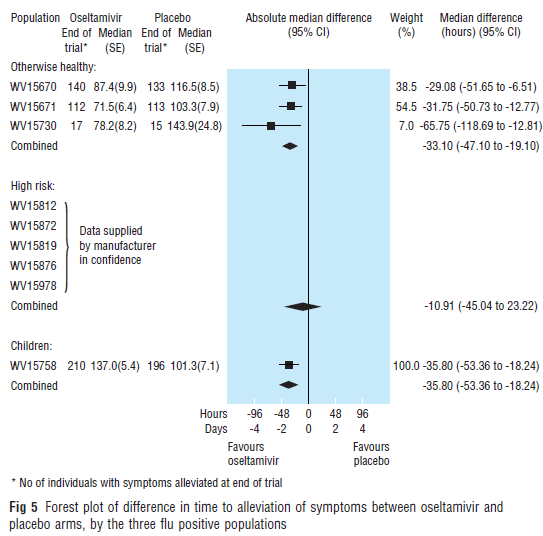
In healthy patients (10) a meta-analysis of 1118 patients treated with oseltamivir and 679 controls found that evidence of shortening duration of symptoms if taken within 48 hours (HR 1.20, 95% CI 1.06-1.35).
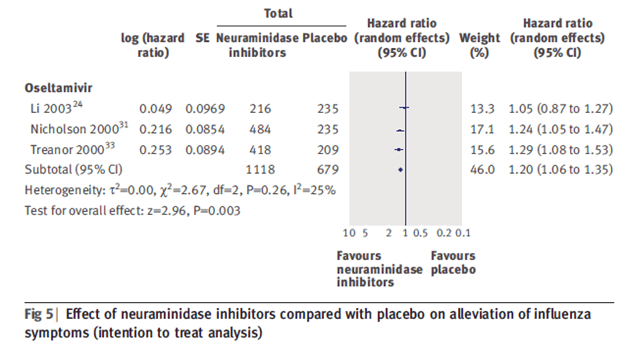
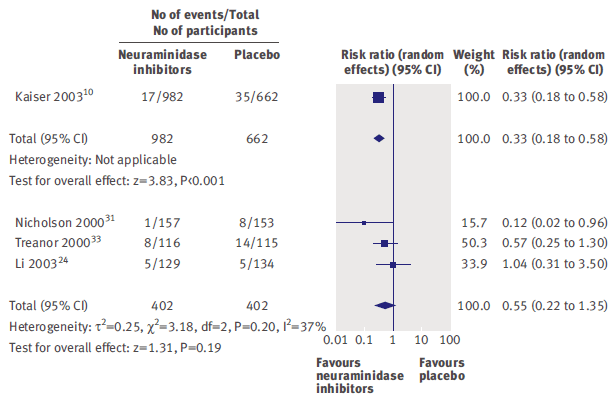
The same meta-analysis did not find evidence of benefit for preventing complications such as pneumonia or bronchitis:
In a Cochrane review (1), early therapy with oseltamivir reduced the time to first alleviation of symptoms by 16.8 hours (95% CI 8.4 to 25.1), representing a 10% reduction from 7 days to 6.3 days. Further, it was found to reduce incidence of investigator-mediated, unverified pneumonia (RR 0.55, 95% CI 0.33 to 0.9), however it had no impact on hospitalization rate (RR 0.92, 95% CI 0.57 to 1.50). Another cohort in ICU patients in Greece compared oseltamivir within 2 days vs after 2 days and the impact of timing on mortality (15). Notably, there was no difference in mortality regardless of when the therapy was given (likely due to how sick these patients were already) but those who had influenza A/H3N2 were more likely to benefit from therapy:
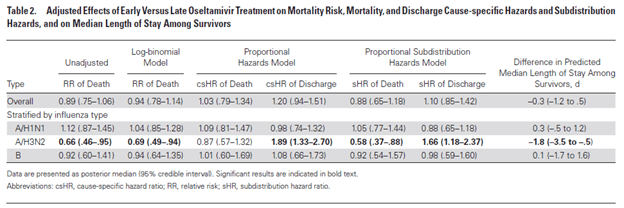
Impact of Oseltamivir on H1N1
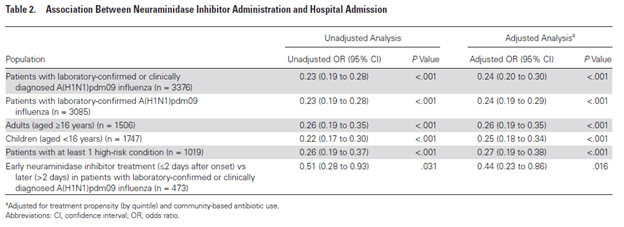
The use of oseltamivir increased in 2009 with the rise of the H1N1 pandemic strain. A meta-analysis (11) evaluated over 3300 patients in the ambulatory setting who were deemed high risk of hospitalization. The adjusted likelihood of hospitalization in those who received oseltamivir was lower when compared to those who did not receive it (aOR 0.24, 95% CI 0.2 to 0.30). This pattern held true for those had one high risk condition and both adults and children:
In a cohort study (12) of 1461 patients admitted with radiographically confirmed pneumonia, in-hospital mortality was higher in patients who did not receive antiviral therapy (18.2%) than those who got it within 2 days (2.9%), those who got it between 2-5 days (4.6%) and those who got it >5 days (4.9%). Further, cox-regression analysis demonstrated survival benefit to the initiation of oseltamivir at any point from symptom onset.
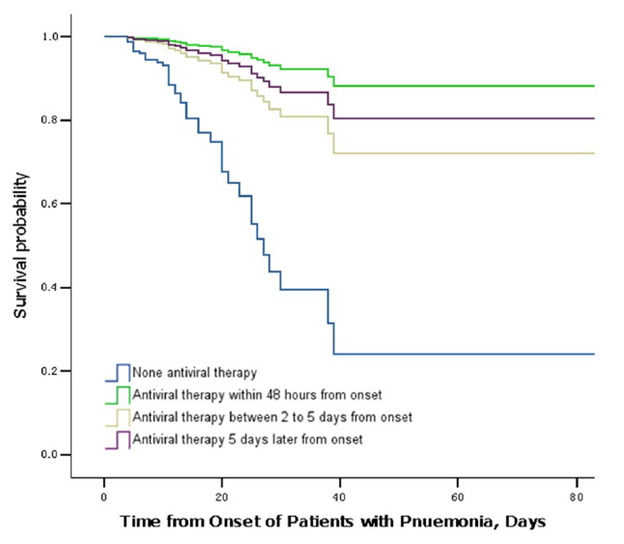
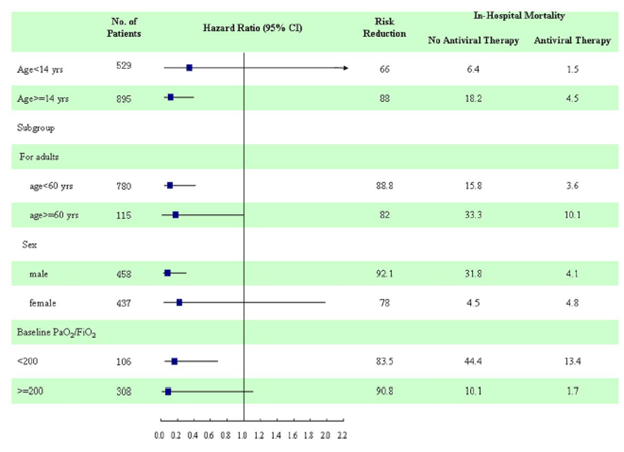
In subgroup analysis, patients younger than 60, males, and those with baseline P/F ratios <200 had significant risk reductions:
A large observational study of critically ill influenza patients (13) evaluated 1676 patients who received oseltamivir and 183 who did not and found that therapy was associated with improve survival, 75% in the group who got oseltamivir vs 58% who did not, however there was little adjustment done and per the authors, they were unable to measure disease severity to adjust for it.
Several meta-analyses have evaluated the impact of oseltamivir on H1N1 mortality. A re-analysis of the FLU-CIN dataset which accounted for time-dependent dynamics of an initial meta-analysis (14) found that oseltamivir therapy was associated with shorter length of stay (aHR 2, 95% CI 1.73-2.13) but did not impact inpatient mortality (aHR 1.04, 95% CI 0.62-1.75):
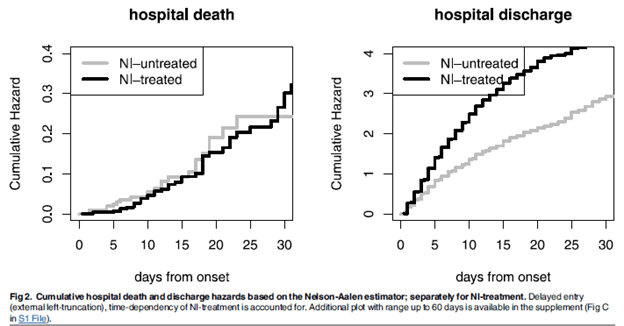
This effect was more pronounced with a higher CURB-65 score and age 16-64, but not age >65. Another meta-analysis evaluated over 29,000 patients (16) found that neuramidinase inhibitor was associated with reduced mortality after adjustment for propensity score, steroid and antibiotic therapy (aOR 0.81, 95% CI 0.70-0.93), which held true when including only laboratory confirmed cases (aOR 0.82, 95% CI 0.7-0.95). In this analysis, starting therapy beyond 2 days was not associated with reduction in mortality in all patients (aOR 1.20, 95% CI 0.93-1.54) but was associated with reduced mortality in those admitted to the ICU (aOR 0.65, 95% CI 0.46-0.93).
It becomes difficult to figure out what to do with these data. On the one hand, there is good evidence that in healthy outpatients, starting oseltamivir early will lead to a modest reduction in symptoms and overall improvement by about a day or so. In terms of inpatients, it tends to be tricky. It would be beneficial to give sick patients (i.e. those requiring oxygen), oseltamivir at any point after their illness, as these patients tend to benefit somewhat (if not by discharge, mortality, it seems). Otherwise, it becomes somewhat difficult to tell. IDSA guidelines (17) suggest that patients be started on influenza therapy if:
- They’re hospitalized with influenza, no matter how long they’ve been sick
- Outpatients if they have progressive disease
- Outpatients if they are at risk of severe disease (i.e immunocompromised and chronic medical conditions)
- Folks aged >65
- Those who have been ill <2 days before starting therapy
- Patient’s whose family members are at high risk of complications
I would suggest that any patient admitted to the hospital would benefit from oseltamivir, as will outpatients who come in early. Those who are ok and are beyond 5 days it becomes a bit dicey, however given the overall tolerability it seems like a safe bet to pursue therapy.
Prophylaxis
Prophylaxis for influenza tends to be something that is not commonly practiced. Pre-exposure prophylaxis is not recommended outside of those who are not vaccinated and are at high risk of complications (17). In terms of post-exposure prophylaxis, usually those who tend to be at high risk of complications are the ones who are recommended to undergo prophylaxis, usually NAI. There is data suggesting its efficacy, however. In a multicenter RCT (18), oseltamivir was compared to placebo in household contacts at risk of influenza within 48hrs of exposure. evaluating efficacy of oseltamivir 75mg daily for seven days in preventing influenza in household contacts vs placebo within 48hrs of exposure. Protective efficacy was high for individuals (89%, 95% CI 71-96%) as well as for households (86%, 95% CI 60-95%). This pattern held true in contacts of an influenza positive and influenza negative index case:
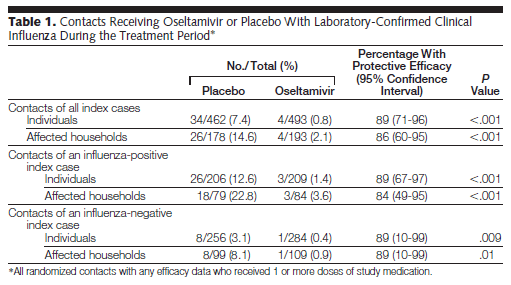
Further, the frequency of viral shedding was significantly lower in the oseltamivir group (54/668 in placebo vs 9/699). A meta-analysis found that oseltamivir was associated with protection against influenza in household contacts (10). A Cochrane analysis (1) found that symptomatic influenza was overall lower in the oseltamivir arm when compared to the placebo arms (RR ).45, 95% CI 0.30 to 0.67) but this did not hold true for asymptomatic influenza (RR 1.14, 95% CI 0.39 to 3.33). Finally, a review (9) found a 90% reduction (95% CI .71 to 0.96) in the incidence of laboratory confirmed influenza in post-exposure prophylaxis.
High Dose/Prolonged Duration
Guidelines do not recommend higher dose of oseltamivir for routine use. Indeed, a prospective study (19) conducted on 75mg and 150mg of oseltamivir did not find any difference in viral RNA negativity (44.7% vs 40.2%) and no difference in hospitalization duration, time to discontinuation of supplemental oxygen, or time to resolution of fever. Further, a randomized double blind study (20) did not find any evidence of benefit of 150mg dose of oseltamivir when it comes to viral RNA negativity (72.3% in standard dose arm, 95% CI 0.65 to 0.79 vs 68.2%, 95% CI 0.601 to 0.75) or clinical failure. Despite this, the guidelines do suggest prolongation of therapy from 5 days to 10 days in certain populations. Part of the reason for this is not because of any clinical benefit to the patient, but rather due to the prolonged shedding that can occur in these populations. As such, 10 day therapies are used in ICU patients to prevent viral shedding (as has been my experience). What is the data behind this? Quite honestly, none. This has to do with the fact that sicker patients shed more viruses for longer, hence the rationale behind prolonging therapy. In a cohort study comparing immunocompromised and immunocompetent patients (21), those who were immunocompromised had prolonged viral shedding, a mean of 19 days vs 6.4 days. In another cohort study (22), patients who had severe pneumonia from influenza were more likely to have higher viral RNA concentration at presentation and the time to nasopharyngeal swab negativity from the time of therapy was longer in patients with severe pneumonia compared to those with mild pneumonia (6 days vs 2 days). The duration of RNA positivity was much longer in tracheal aspirates, with a mean of 11 days following initiation of oseltamivir therapy:
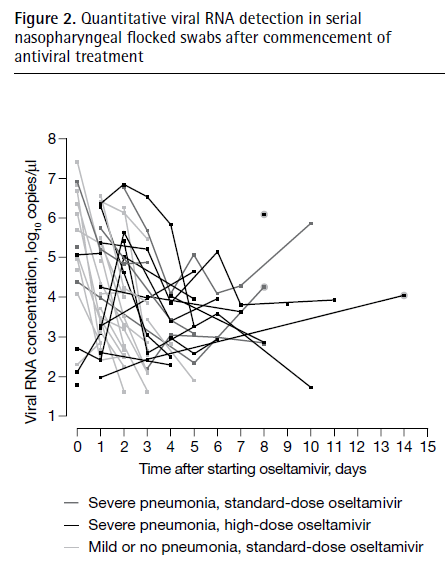
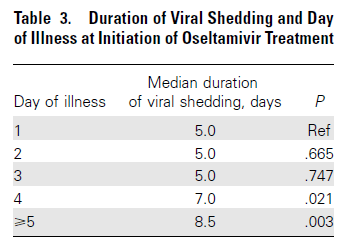
Given this, it may be reasonable to prolong duration of therapy to at least 10 days in immunocompromised patients, with the rationale that higher viral loads are correlated more severe disease. Despite this, however, there is some data in immunocompetent patients that suggest starting oseltamivir after 4 days of symptoms prolongs median duration of viral shedding (23):
TL;DR
- Oseltamivir reduces duration of illness by about a day in outpatients if given within 48hrs of symptom onset
- It is also associated with shorter hospitalization, with some studies suggesting mortality benefit and others suggesting reduction in certain complications such as pneumonia
- By far, recommendations are to proceed with oseltamivir if someone is admitted, regardless of when their illness started
- No role for prophylaxis UNLESS the patient is at high risk of complications
- No use for dosing beyond 75mg twice daily
- Immunosuppressed patients and severe presentations are associated with prolonged viral shedding, and these may benefit from prolonged therapy
References:
- Jefferson T, Jones MA, Doshi P, Del Mar CB, Hama R, Thompson MJ, Spencer EA, Onakpoya I, Mahtani KR, Nunan D, Howick J, Heneghan CJ. Neuraminidase inhibitors for preventing and treating influenza in healthy adults and children. Cochrane Database Syst Rev. 2014 Apr 10;2014(4):CD008965. doi: 10.1002/14651858.CD008965.pub4. PMID: 24718923; PMCID: PMC6464969.
- Hayden FG, Treanor JJ, Fritz RS, Lobo M, Betts RF, Miller M, Kinnersley N, Mills RG, Ward P, Straus SE. Use of the oral neuraminidase inhibitor oseltamivir in experimental human influenza: randomized controlled trials for prevention and treatment. JAMA. 1999 Oct 6;282(13):1240-6. doi: 10.1001/jama.282.13.1240. PMID: 10517426.
- Treanor JJ, Hayden FG, Vrooman PS, et al. Efficacy and Safety of the Oral Neuraminidase Inhibitor Oseltamivir in Treating Acute Influenza: A Randomized Controlled Trial. JAMA. 2000;283(8):1016–1024. doi:10.1001/jama.283.8.1016
- Aoki FY, Macleod MD, Paggiaro P, Carewicz O, El Sawy A, Wat C, Griffiths M, Waalberg E, Ward P; IMPACT Study Group. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003 Jan;51(1):123-9. doi: 10.1093/jac/dkg007. PMID: 12493796.
- Kawai N, Ikematsu H, Iwaki N, Satoh I, Kawashima T, Maeda T, Miyachi K, Hirotsu N, Shigematsu T, Kashiwagi S. Factors influencing the effectiveness of oseltamivir and amantadine for the treatment of influenza: a multicenter study from Japan of the 2002-2003 influenza season. Clin Infect Dis. 2005 May 1;40(9):1309-16. doi: 10.1086/429241. Epub 2005 Mar 16. PMID: 15825034.
- Kaiser L, Wat C, Mills T, Mahoney P, Ward P, Hayden F. Impact of oseltamivir treatment on influenza-related lower respiratory tract complications and hospitalizations. Arch Intern Med. 2003 Jul 28;163(14):1667-72. doi: 10.1001/archinte.163.14.1667. PMID: 12885681.
- Adisasmito W, Chan PK, Lee N, Oner AF, Gasimov V, Aghayev F, Zaman M, Bamgboye E, Dogan N, Coker R, Starzyk K, Dreyer NA, Toovey S. Effectiveness of antiviral treatment in human influenza A(H5N1) infections: analysis of a Global Patient Registry. J Infect Dis. 2010 Oct 15;202(8):1154-60. doi: 10.1086/656316. PMID: 20831384.
- McGeer A, Green KA, Plevneshi A, Shigayeva A, Siddiqi N, Raboud J, Low DE; Toronto Invasive Bacterial Diseases Network. Antiviral therapy and outcomes of influenza requiring hospitalization in Ontario, Canada. Clin Infect Dis. 2007 Dec 15;45(12):1568-75. doi: 10.1086/523584. PMID: 18190317.
- Cooper NJ, Sutton AJ, Abrams KR, Wailoo A, Turner D, Nicholson KG. Effectiveness of neuraminidase inhibitors in treatment and prevention of influenza A and B: systematic review and meta-analyses of randomised controlled trials. BMJ. 2003 Jun 7;326(7401):1235. doi: 10.1136/bmj.326.7401.1235. PMID: 12791735; PMCID: PMC161558.
- Jefferson Tom, Jones Mark, Doshi Peter, Del Mar Chris. Neuraminidase inhibitors for preventing and treating influenza in healthy adults: systematic review and meta-analysis BMJ 2009; 339 :b5106
- Venkatesan S, Myles PR, Leonardi-Bee J, Muthuri SG, Al Masri M, Andrews N, Bantar C, Dubnov-Raz G, Gérardin P, Koay ESC, Loh TP, Memish Z, Miller E, Oliva ME, Rath BA, Schweiger B, Tang JW, Tran D, Vidmar T, Waight PA, Nguyen-Van-Tam JS. Impact of Outpatient Neuraminidase Inhibitor Treatment in Patients Infected With Influenza A(H1N1)pdm09 at High Risk of Hospitalization: An Individual Participant Data Metaanalysis. Clin Infect Dis. 2017 May 15;64(10):1328-1334. doi: 10.1093/cid/cix127. PMID: 28199524; PMCID: PMC5411393.
- Yang SG, Cao B, Liang LR, Li XL, Xiao YH, Cao ZX, Jia HY, Yu HJ, Xu Z, Gu L, Yang YD, Chen Y, Du WB, Yan XX, Liang ZA, Zhang W, Zhang CL, Chen W, Guo CP, Jiang XL, Yang M, Deng GM, Yu KJ, Hu K, Zou Q, Li LJ, Wang C; National Influenza A Pandemic (H1N1) 2009 Clinical Investigation Group of China. Antiviral therapy and outcomes of patients with pneumonia caused by influenza A pandemic (H1N1) virus. PLoS One. 2012;7(1):e29652. doi: 10.1371/journal.pone.0029652. Epub 2012 Jan 20. PMID: 22276122; PMCID: PMC3262784.
- Louie JK, Yang S, Acosta M, Yen C, Samuel MC, Schechter R, Guevara H, Uyeki TM. Treatment with neuraminidase inhibitors for critically ill patients with influenza A (H1N1)pdm09. Clin Infect Dis. 2012 Nov;55(9):1198-204. doi: 10.1093/cid/cis636. Epub 2012 Jul 26. PMID: 22843781.
- Wolkewitz M, Schumacher M. Neuraminidase Inhibitors and Hospital Mortality in British Patients with H1N1 Influenza A: A Re-Analysis of Observational Data. PLoS One. 2016 Sep 1;11(9):e0160430. doi: 10.1371/journal.pone.0160430. Erratum in: PLoS One. 2017 Apr 6;12 (4):e0175660. PMID: 27583403; PMCID: PMC5008789.
- Lytras T, Mouratidou E, Andreopoulou A, Bonovas S, Tsiodras S. Effect of Early Oseltamivir Treatment on Mortality in Critically Ill Patients With Different Types of Influenza: A Multiseason Cohort Study. Clin Infect Dis. 2019 Nov 13;69(11):1896-1902. doi: 10.1093/cid/ciz101. PMID: 30753349.
- Muthuri SG, Venkatesan S, Myles PR, Leonardi-Bee J, Al Khuwaitir TS, Al Mamun A, Anovadiya AP, Azziz-Baumgartner E, Báez C, Bassetti M, Beovic B, Bertisch B, Bonmarin I, Booy R, Borja-Aburto VH, Burgmann H, Cao B, Carratala J, Denholm JT, Dominguez SR, Duarte PA, Dubnov-Raz G, Echavarria M, Fanella S, Gao Z, Gérardin P, Giannella M, Gubbels S, Herberg J, Iglesias AL, Hoger PH, Hu X, Islam QT, Jiménez MF, Kandeel A, Keijzers G, Khalili H, Knight M, Kudo K, Kusznierz G, Kuzman I, Kwan AM, Amine IL, Langenegger E, Lankarani KB, Leo YS, Linko R, Liu P, Madanat F, Mayo-Montero E, McGeer A, Memish Z, Metan G, Mickiene A, Mikić D, Mohn KG, Moradi A, Nymadawa P, Oliva ME, Ozkan M, Parekh D, Paul M, Polack FP, Rath BA, Rodríguez AH, Sarrouf EB, Seale AC, Sertogullarindan B, Siqueira MM, Skręt-Magierło J, Stephan F, Talarek E, Tang JW, To KK, Torres A, Törün SH, Tran D, Uyeki TM, Van Zwol A, Vaudry W, Vidmar T, Yokota RT, Zarogoulidis P; PRIDE Consortium Investigators, Nguyen-Van-Tam JS. Effectiveness of neuraminidase inhibitors in reducing mortality in patients admitted to hospital with influenza A H1N1pdm09 virus infection: a meta-analysis of individual participant data. Lancet Respir Med. 2014 May;2(5):395-404. doi: 10.1016/S2213-2600(14)70041-4. Epub 2014 Mar 19. PMID: 24815805; PMCID: PMC6637757.
- Timothy M Uyeki, Henry H Bernstein, John S Bradley, Janet A Englund, Thomas M File, Jr, Alicia M Fry, Stefan Gravenstein, Frederick G Hayden, Scott A Harper, Jon Mark Hirshon, Michael G Ison, B Lynn Johnston, Shandra L Knight, Allison McGeer, Laura E Riley, Cameron R Wolfe, Paul E Alexander, Andrew T Pavia, Clinical Practice Guidelines by the Infectious Diseases Society of America: 2018 Update on Diagnosis, Treatment, Chemoprophylaxis, and Institutional Outbreak Management of Seasonal Influenza, Clinical Infectious Diseases, Volume 68, Issue 6, 15 March 2019, Pages e1–e47, https://doi.org/10.1093/cid/ciy866
- Welliver R, Monto AS, Carewicz O, Schatteman E, Hassman M, Hedrick J, Jackson HC, Huson L, Ward P, Oxford JS; Oseltamivir Post Exposure Prophylaxis Investigator Group. Effectiveness of oseltamivir in preventing influenza in household contacts: a randomized controlled trial. JAMA. 2001 Feb 14;285(6):748-54. doi: 10.1001/jama.285.6.748. PMID: 11176912.
- Lee N, Hui DS, Zuo Z, Ngai KL, Lui GC, Wo SK, Tam WW, Chan MC, Wong BC, Wong RY, Choi KW, Sin WW, Lee EL, Tomlinson B, Hayden FG, Chan PK. A prospective intervention study on higher-dose oseltamivir treatment in adults hospitalized with influenza a and B infections. Clin Infect Dis. 2013 Dec;57(11):1511-9. doi: 10.1093/cid/cit597. Epub 2013 Sep 17. PMID: 24046309.
- South East Asia Infectious Disease Clinical Research Network. Effect of double dose oseltamivir on clinical and virological outcomes in children and adults admitted to hospital with severe influenza: double blind randomised controlled trial. BMJ. 2013 May 30;346:f3039. doi: 10.1136/bmj.f3039. PMID: 23723457; PMCID: PMC3668094.
- Memoli MJ, Athota R, Reed S, Czajkowski L, Bristol T, Proudfoot K, Hagey R, Voell J, Fiorentino C, Ademposi A, Shoham S, Taubenberger JK. The natural history of influenza infection in the severely immunocompromised vs nonimmunocompromised hosts. Clin Infect Dis. 2014 Jan;58(2):214-24. doi: 10.1093/cid/cit725. Epub 2013 Nov 1. PMID: 24186906; PMCID: PMC3871797.
- Lee N, Chan PK, Wong CK, Wong KT, Choi KW, Joynt GM, Lam P, Chan MC, Wong BC, Lui GC, Sin WW, Wong RY, Lam WY, Yeung AC, Leung TF, So HY, Yu AW, Sung JJ, Hui DS. Viral clearance and inflammatory response patterns in adults hospitalized for pandemic 2009 influenza A(H1N1) virus pneumonia. Antivir Ther. 2011;16(2):237-47. doi: 10.3851/IMP1722. PMID: 21447873.
- Ling LM, Chow AL, Lye DC, Tan AS, Krishnan P, Cui L, Win NN, Chan M, Lim PL, Lee CC, Leo YS. Effects of early oseltamivir therapy on viral shedding in 2009 pandemic influenza A (H1N1) virus infection. Clin Infect Dis. 2010 Apr 1;50(7):963-9. doi: 10.1086/651083. PMID: 20180701.
