Influenza season is right around the corner, and given the current circumstances, it is a terrifying thought to have both COVID and flu running around. Besides causing significant respiratory distress and subsequent ARDS, severe influenza leads to bacterial superinfection with organisms such as Staph aureus and Streptococcal pneumonia. This usually happens in 10-35% of cases (1) and leads to significant morbidity and mortality. There has been more reports and case series illustrating that invasive pulmonary aspergillosis complicates cases of severe influenza. This has been reported as far back as in the 70s, where 2 cases of influenza A were complicated by invasive pulmonary aspergillosis (2).
The pathophysiology behind this is poorly understood but is likely that damage to the epithelium by influenza and defective fungal hosts responses in the lung predispose to Aspergillus infection (1). Autopsy studies have also found sporulating heads of Aspergillus inside bronchi with invasive growth occurring.
Most of the data regarding this subject is retrospective, but it is compelling. For instance, one of the major cohorts (3) evaluated 432 patients admitted to the ICU with severe influenza and compared them to matched controls with bacterial pneumonia. They further evaluated 315 influenza patients without classic EORTC/MSG risk factors for IPA. Within patients who had the flu, male gender and prednisone were significant risk factors for development of IPA:

However, when comparing the influenza group and the pneumonia group, influenza was a significant risk factor for IPA, resulting with an aOR of 5.19 (95% CI 2.63-10.26):

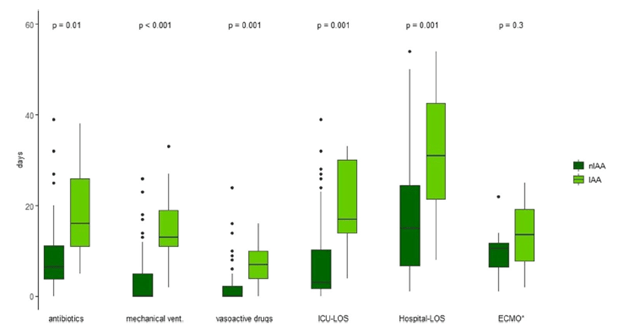
The incidence of IPA was 14% in the influenza group compared to 5% of the control group. Mortality was higher in the IPA group of patients with influenza (51% vs 28%). Further, BAL cultures were only positive in 63% of cases, with 88% of cases having a positive BAL galactomannan, and 65% having positive serum galactomannan. Another retrospective study from Switzerland (4) evaluated 81 patients with severe influenza and found 9 patients with IPA (11%) and found that these patients had bad outcomes when it came to median ICU length of stay (17 vs 3 days, p <0.01) and were more likely to have more antibiotic days, be longer on mechanical ventilation, or be on vasoactive drugs:
And in a retrospective cohort (5), corticosteroid use was found to be associated with influenza-related PA, with an OR of 14.4 (2.0-101.6, p-value 0.007). Further, more patients in this cohort with IPA were more likely to have bacteremia (67% vs 19%, p-value 0.006), though the overall cohort was fairly small, with a total of 40 patients, 9 of which had IPA. This was also found in another retrospective study (6), which compared flu patients with IPA and flu patients without. Those with IPA were more likely to be diabetic, have COPD/asthma, or have CHF, as well as having had corticosteroid use before the diagnosis. A logistic regression model found that age, systemic corticosteroid use before IPA, leukocytes >10 x 109/LL and lymphocytes <0.8 x 109/L were associated with acquisition of IPA while early neuraminidase use was associated with a decreased incidence of IPA:

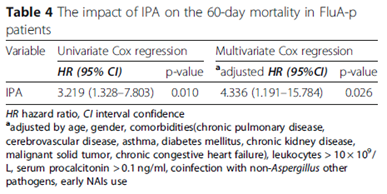
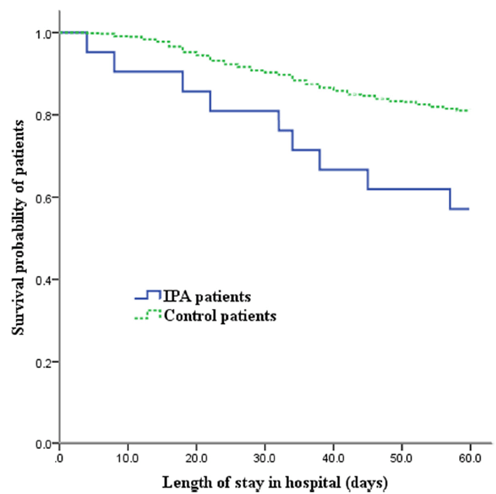
Further, the acquisition of IPA increased the mortality rate by up to four-fold:
One of the interesting things here is these patients do not have the classic risk factors for IPA. For instance, in one cohort study (7) the overall incidence of influenza associated IPA was 16%, with 23% of these having some sort of azole resistance. Mortality in healthy patients was 5/7 (71%) compared to 9/16 (56%) of those who had co-morbidities. Of these 23 cases, 7 had no co-morbidities whatsoever, 3 had hematology malignancies, 2 were transplant patients, and 4 had some sort of autoimmune disease. Another review (8) found 36 cases in patients who were immunocompetent. This becomes an issue, since the EORTC/MSG criteria for probable IPA requires 3 criteria to be met, which include the host factors, clinical features, and mycological criteria (9):

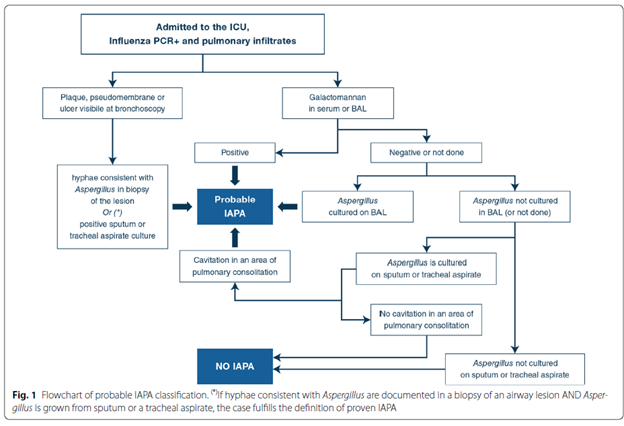
Of course, you can see the problem. If you go along with this diagnostic modality, you will miss patients who do not fall under the “classic” criteria such the severe influenza patients. Due to this, an alternative algorithm, the AspICU, was developed to be applied to the critically ill influenza patient (10).

The good thing about this is that it host factors are not a requirement to be diagnosed with putative. At the end of the day, getting a culture off sputum is difficult to interpret because everyone inhales spores daily, so we do not know if this is colonization or true infection in most cases (this is the reason why most ID folks will usually ignore a sputum culture with Aspergillus in someone with no imaging findings or no risk factors). This algorithm was evaluated using 115 patients from a multicenter observational cohort (10) using histopathological data and subsequently validated using a cohort of 524 patients. The overall sensitivity and specificity of this algorithm was 92% and 61%, respectively with a PPV and NPV of 61% and 92%. Comparing the AspICU ROC and the EORTC/MSG ROC, the area under the curve was 76% for the AspICU vs 57% for the EORTC/MSG:

Another proposed criteria specifically for influenza patients (1) was recently released, along with an algorithm to boot.
COVID-Associated Pulmonary Aspergillosis
This is largely limited to case reports, but given that we have very little in terms of therapeutics for severe COVID infection and that one of them puts you at risk for invasive mold infections, I think it is helpful to at least think about it. A retrospective chart review (11) evaluated 5 patients with COVID and pulmonary aspergillosis. All had severe ARDS, requiring proning, with one requiring vvECMO. None of the patients had any hematological malignancies, one had IV steroids for a total of 2 weeks, and 2 were on inhalational steroids for COPD. 3/5 patients in this cohort died. In another cohort (12) six patients were found to have IPA, and similar to the other cohort, none of them had SOT or hematological malignancies. 3 patients were given steroids prior to admission (one for cardiomyopathy, one for COPD, and another one for asthma).
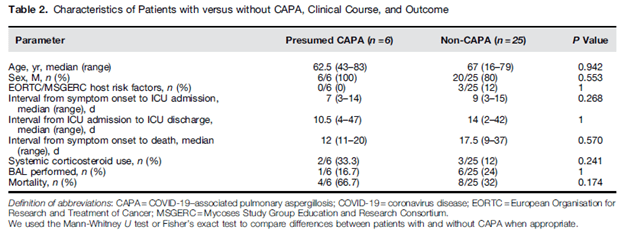
The largest cohort (13) evaluated 27 consecutive ventilated COVID patients and diagnosed putative IPA in 8 patients (30%) on day 3 after intubation. Putative diagnoses was considered if there was Aspergillus spp in BAL culture or if 2 other microbiological points were met (presence of Aspergillus spp in aspirate, +Aspergillus PCR in BAL, BA, or serum, GM >0.8 in BAL or >0.5 on serum, or BD glucan >80. None of the patients had classic risk factors and 6 of the 8 patients had received corticosteroids during the hospitalization. One review (1) suggests there is a difference between CAPA and flu-associated IPA, namely on their pathophysiology. For instance, the ACE-2 receptors are not commonly found in large airways and SARS-CoV2 has no know direct immune modulatory effect, suggesting there may not be a virus infection-related increase risk for IPA. The authors do note, however, this has not been extensively studied and while the virus itself may not increase the risk, it puts someone in a situation where there is higher risk for tissue invasion (i.e. severe COVID -> steroid administration + aspergillus colonization -> invasive disease).

References:
- Verweij PE, Rijnders BJA, Brüggemann RJM, Azoulay E, Bassetti M, Blot S, Calandra T, Clancy CJ, Cornely OA, Chiller T, Depuydt P, Giacobbe DR, Janssen NAF, Kullberg BJ, Lagrou K, Lass-Flörl C, Lewis RE, Liu PW, Lortholary O, Maertens J, Martin-Loeches I, Nguyen MH, Patterson TF, Rogers TR, Schouten JA, Spriet I, Vanderbeke L, Wauters J, van de Veerdonk FL. Review of influenza-associated pulmonary aspergillosis in ICU patients and proposal for a case definition: an expert opinion. Intensive Care Med. 2020 Aug;46(8):1524-1535. doi: 10.1007/s00134-020-06091-6. Epub 2020 Jun 22. PMID: 32572532; PMCID: PMC7306567.
- Fischer JJ, Walker DH. Invasive pulmonary aspergillosis associated with influenza. JAMA. 1979 Apr 6;241(14):1493-4. PMID: 372590.
- Schauwvlieghe AFAD, Rijnders BJA, Philips N, Verwijs R, Vanderbeke L, Van Tienen C, Lagrou K, Verweij PE, Van de Veerdonk FL, Gommers D, Spronk P, Bergmans DCJJ, Hoedemaekers A, Andrinopoulou ER, van den Berg CHSB, Juffermans NP, Hodiamont CJ, Vonk AG, Depuydt P, Boelens J, Wauters J; Dutch-Belgian Mycosis study group. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: a retrospective cohort study. Lancet Respir Med. 2018 Oct;6(10):782-792. doi: 10.1016/S2213-2600(18)30274-1. Epub 2018 Jul 31. PMID: 30076119.
- Waldeck F, Boroli F, Suh N, Wendel Garcia PD, Flury D, Notter J, Iten A, Kaiser L, Schrenzel J, Boggian K, Maggiorini M, Pugin J, Kleger GR, Albrich WC. Influenza-associated aspergillosis in critically-ill patients-a retrospective bicentric cohort study. Eur J Clin Microbiol Infect Dis. 2020 Oct;39(10):1915-1923. doi: 10.1007/s10096-020-03923-7. Epub 2020 Jun 3. PMID: 32494955; PMCID: PMC7266735.
- Wauters J, Baar I, Meersseman P, Meersseman W, Dams K, De Paep R, Lagrou K, Wilmer A, Jorens P, Hermans G. Invasive pulmonary aspergillosis is a frequent complication of critically ill H1N1 patients: a retrospective study. Intensive Care Med. 2012 Nov;38(11):1761-8. doi: 10.1007/s00134-012-2673-2. Epub 2012 Aug 16. PMID: 22895826; PMCID: PMC7079899.
- Chen L, Han X, Li Y, Zhang C, Xing X. Invasive pulmonary aspergillosis in immunocompetent patients hospitalised with influenza A-related pneumonia: a multicenter retrospective study. BMC Pulm Med. 2020 Sep 9;20(1):239. doi: 10.1186/s12890-020-01257-w. PMID: 32907585; PMCID: PMC7479745.
- van de Veerdonk FL, Kolwijck E, Lestrade PP, Hodiamont CJ, Rijnders BJ, van Paassen J, Haas PJ, Oliveira Dos Santos C, Kampinga GA, Bergmans DC, van Dijk K, de Haan AF, van Dissel J, van der Hoeven HG, Verweij PE; Dutch Mycoses Study Group. Influenza-Associated Aspergillosis in Critically Ill Patients. Am J Respir Crit Care Med. 2017 Aug 15;196(4):524-527. doi: 10.1164/rccm.201612-2540LE. Epub 2017 Apr 7. PMID: 28387526.
- Shah MM, Hsiao EI, Kirsch CM, Gohil A, Narasimhan S, Stevens DA. Invasive pulmonary aspergillosis and influenza co-infection in immunocompetent hosts: case reports and review of the literature. Diagn Microbiol Infect Dis. 2018 Jun;91(2):147-152. doi: 10.1016/j.diagmicrobio.2018.01.014. Epub 2018 Feb 2. PMID: 29454654; PMCID: PMC5970059.
- Donnelly JP, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE, Clancy CJ, Wingard JR, Lockhart SR, Groll AH, Sorrell TC, Bassetti M, Akan H, Alexander BD, Andes D, Azoulay E, Bialek R, Bradsher RW, Bretagne S, Calandra T, Caliendo AM, Castagnola E, Cruciani M, Cuenca-Estrella M, Decker CF, Desai SR, Fisher B, Harrison T, Heussel CP, Jensen HE, Kibbler CC, Kontoyiannis DP, Kullberg BJ, Lagrou K, Lamoth F, Lehrnbecher T, Loeffler J, Lortholary O, Maertens J, Marchetti O, Marr KA, Masur H, Meis JF, Morrisey CO, Nucci M, Ostrosky-Zeichner L, Pagano L, Patterson TF, Perfect JR, Racil Z, Roilides E, Ruhnke M, Prokop CS, Shoham S, Slavin MA, Stevens DA, Thompson GR, Vazquez JA, Viscoli C, Walsh TJ, Warris A, Wheat LJ, White PL, Zaoutis TE, Pappas PG. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020 Sep 12;71(6):1367-1376. doi: 10.1093/cid/ciz1008. PMID: 31802125; PMCID: PMC7486838.
- Blot SI, Taccone FS, Van den Abeele AM, Bulpa P, Meersseman W, Brusselaers N, Dimopoulos G, Paiva JA, Misset B, Rello J, Vandewoude K, Vogelaers D; AspICU Study Investigators. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients. Am J Respir Crit Care Med. 2012 Jul 1;186(1):56-64. doi: 10.1164/rccm.201111-1978OC. Epub 2012 Apr 19. Erratum in: Am J Respir Crit Care Med. 2012 Oct 15;186(8):808. PMID: 22517788.
- Koehler P, Cornely OA, Böttiger BW, Dusse F, Eichenauer DA, Fuchs F, Hallek M, Jung N, Klein F, Persigehl T, Rybniker J, Kochanek M, Böll B, Shimabukuro-Vornhagen A. COVID-19 associated pulmonary aspergillosis. Mycoses. 2020 Jun;63(6):528-534. doi: 10.1111/myc.13096. Epub 2020 May 15. PMID: 32339350; PMCID: PMC7267243.
- van Arkel ALE, Rijpstra TA, Belderbos HNA, van Wijngaarden P, Verweij PE, Bentvelsen RG. COVID-19-associated Pulmonary Aspergillosis. Am J Respir Crit Care Med. 2020;202(1):132-135. doi:10.1164/rccm.202004-1038LE
- Alanio A, Dellière S, Fodil S, Bretagne S, Mégarbane B. Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Lancet Respir Med. 2020 Jun;8(6):e48-e49. doi: 10.1016/S2213-2600(20)30237-X. Epub 2020 May 20. PMID: 32445626; PMCID: PMC7239617.

1 comments on “Influenza, COVID-19, Pulmonary Aspergillosis, Lions, Tigers, And Bears – Oh My!”