Ah, its been quite some time. There are some posts (hopefully) coming soon. Anywho, lets get on to some articles:
Levin EG, Lustig Y, Cohen C, Fluss R, Indenbaum V, Amit S, Doolman R, Asraf K, Mendelson E, Ziv A, Rubin C, Freedman L, Kreiss Y, Regev-Yochay G. Waning Immune Humoral Response to BNT162b2 Covid-19 Vaccine over 6 Months. N Engl J Med. 2021 Oct 6. doi: 10.1056/NEJMoa2114583. Epub ahead of print. PMID: 34614326.
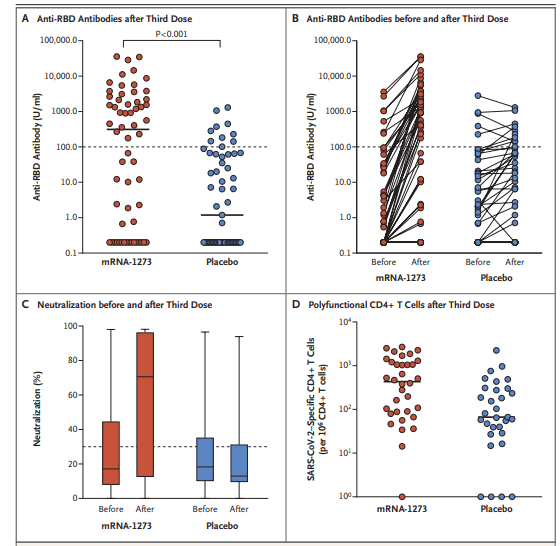
This is a prospective longitudinal cohort study of healthcare workers in Israel who receive 2 doses of the mRNA vaccine. Both IgG and neutralizing antibody levels were analyzed every month for 6 months after receipt of the second dose of the vaccine. 4868 people were recruited to participate, during which 20 had breakthrough covert infections. Overall there was a reduction in the IgG level each month decreasing by a factor of 18.3 at months 6. There was also a decrease in the neutralizing antibody titers however after month 3 the decrease was much lower.
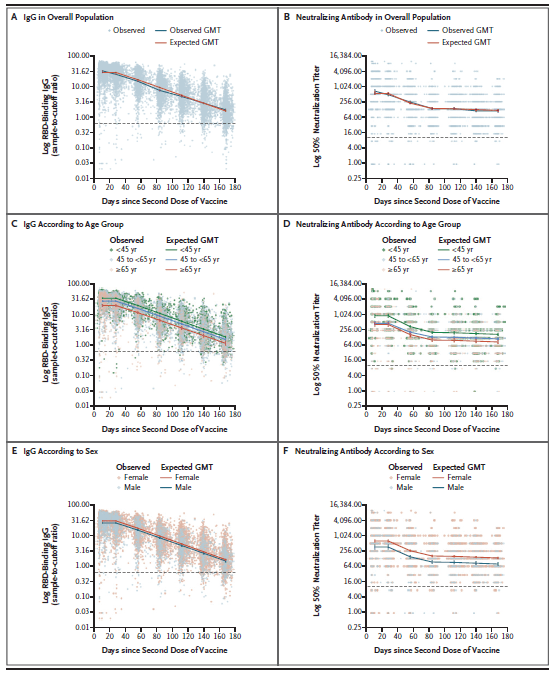
As can be seeing in the graphs above the decrease in the IgG overall plus steadily over the first 6 months however for neutralizing antibodies, decrease seems to slow down after 3 months and remains constant for the remainder of the study.. In those who were above the age of 65, the peak IgG and neutralizing antibody were significantly lower than those who are under 45. This pattern held through event at the end of the study at 6 months. Interestingly, the dose of had a BMI greater than 30 had greater peak IgG neutralizing antibodies and there and his study titers were also similarly higher than baseline.
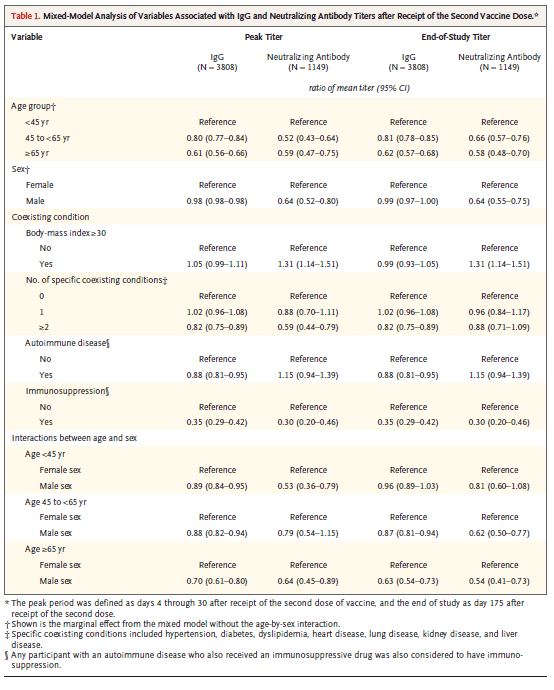
It is always difficult to extrapolate data on antibody titers into clinical scenarios, however the suggest that booster antibodies may not be a bad idea going forward, especially as this virus is most certainly going to circulate in the coming years. Likely, the same issue of New England Journal of Medicine actually has a study that answers question of clinical implications of decreasing antibody titers.
Chemaitelly H, Tang P, Hasan MR, AlMukdad S, Yassine HM, Benslimane FM, Al Khatib HA, Coyle P, Ayoub HH, Al Kanaani Z, Al Kuwari E, Jeremijenko A, Kaleeckal AH, Latif AN, Shaik RM, Abdul Rahim HF, Nasrallah GK, Al Kuwari MG, Al Romaihi HE, Butt AA, Al-Thani MH, Al Khal A, Bertollini R, Abu-Raddad LJ. Waning of BNT162b2 Vaccine Protection against SARS-CoV-2 Infection in Qatar. N Engl J Med. 2021 Oct 6. doi: 10.1056/NEJMoa2114114. Epub ahead of print. PMID: 34614327.
This is a prospective study evaluating the effectiveness of the Pfizer vaccine in Covid related hospitalization and that after the receipt of both the first and second doses. During the study., 947,035 people received at least 1 dose of the vaccine with weight 907,716 receiving both doses. Similarly, 564,196 people received at least 1 dose of the Moderna vaccine. Overall the over over 10,000 breakthrough infections and does receive 2 doses of the Pfister vaccine. In looking at the overall effectiveness against Covid infections, the effectiveness decrease from nearly 0 to 37% 2 weeks after the first dose. The efficacy reached a peak of 77.5% a month after the second dose, however the efficacy decrease from the first month after the second dose. The effectiveness was a low level of around 20% at month 5 and held month 7 after the second dose. Despite this however, peak effectiveness against any symptomatic infection was 82%, compared to 73% and those were asymptomatic. Despite these numbers however the effectiveness against severe, critical, or fatal disease due to Covid reached a peak of 96% in the first 2 months after the second dose. The lowest efficacy seeing during the study. Was 55.6% at month 7 however should be noted that even at month 6 the efficacy against severe or critical infections was roughly 88.9%.
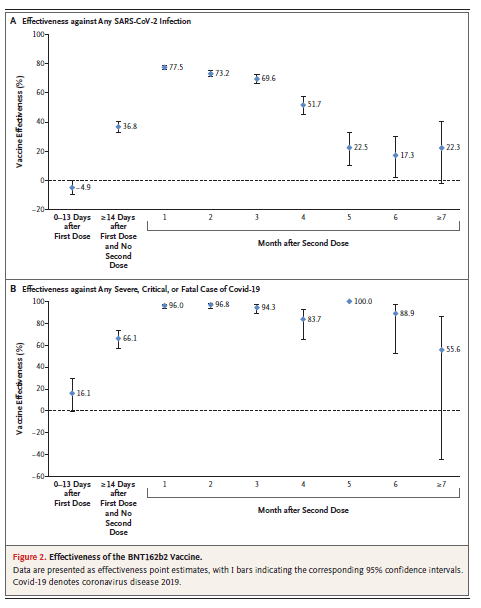
Sensitivity analysis as shown below, demonstrates that after the initial vaccination protection is not significant however this increases rapidly after the second dose. Further there is a significant drop off in efficacy in the months following the second dose, this does not seem to be the case for hospitalizations and death.
While looking at the efficacy of vaccinations in isolation suggest that the overall effectiveness of vaccines is going down, I think it is critical to look at the benefits and hospitalization and death. I think it is safe to say that these are heart outcomes that we are caring about, so the statin should provide some sort of relief that will be overall effectiveness against any type of infection seems to be going down, if protections again severe cases and death seem to be holding strong. Regardless it seems that booster shots may be in her future when it comes to this vaccine and I anticipate that this is going to be one at the shots that we had to get every year along with the flu vaccine.
Shah VP, Farah WH, Hill JC, Hassett LC, Binnicker MJ, Yao JD, Hassan Murad M. Association Between SARS-CoV-2 Cycle Threshold Values and Clinical Outcomes in Patients With COVID-19: A Systematic Review and Meta-analysis. Open Forum Infect Dis. 2021 Aug 31;8(9):ofab453. doi: 10.1093/ofid/ofab453. PMID: 34584900; PMCID: PMC8465328.
Now on to other Covid related articles. This was a meta-analysis that evaluated the association between cycling threshold and several clinical parameters, which included the need for hospitalization, to seek severity based on the WHO severity scale and in-hospital and 30-day mortality. A total of 18 studies were evaluated, however 15 of these were considered to be at a high risk about bias. Overall the meta-analysis did not find the difference in the cycling threshold value between hospitalized and non hospitalized patients (0.062; 95% CI, -1.933-2.056). In terms of the severity, patients with cycling threshold less than 25 or between 25 and 30 had an increased risk of severe disease compared to dose cycling thresholds were greater than 30 (OR, 2.31; 95% CI, 1.70-3.13; and OR, 1.45; 95% CI, 1.06-1.97).
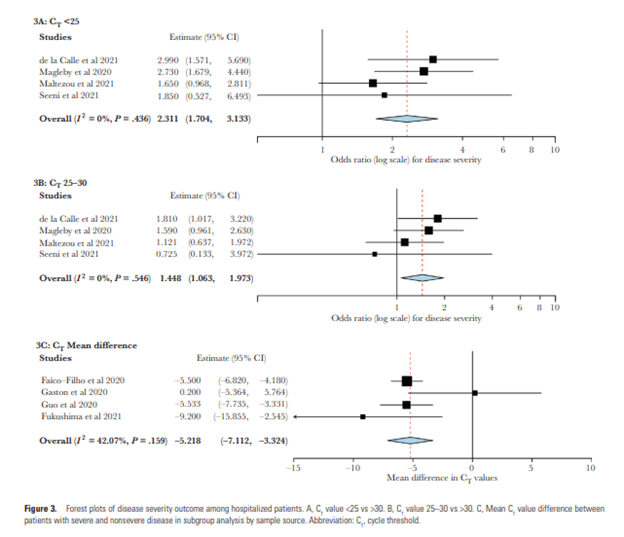
In terms of risk of mortality, those who have had a cycling threshold less than 25 had an increase risk of mortality compared to those of cycling thresholds was greater than 30 (OR, 2.95; 95% CI, 2.19-3.96). These results were similar to those 2 cycling thresholds were between 25 and 30 compared to those who cycling threshold was greater than 30 (OR, 1.59; 95% CI, 1.192 2.14).
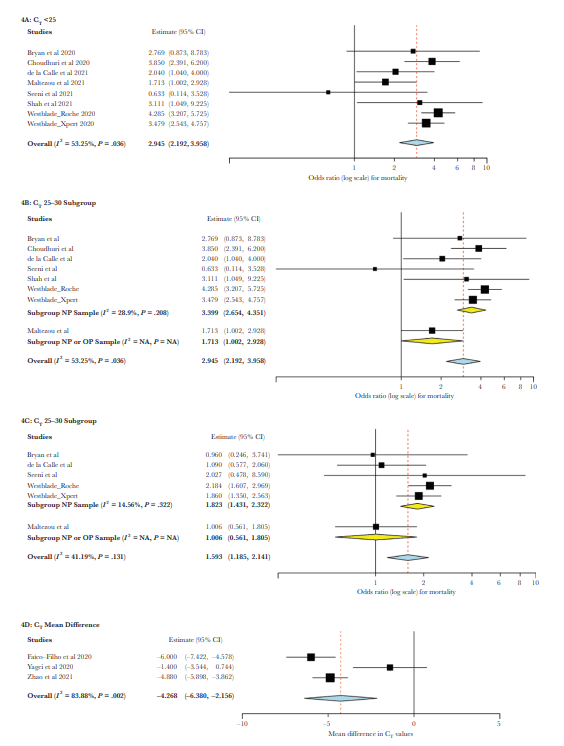
To reiterate the lower the cyclin threshold means that the PCR has to go through last cycles to amplify the virus to the point that it is detectable by the assay. This means that when the cycle threshold is lowered the viral load is actually higher. How this relates to the cytokine storm that is seen in severe Covid patients is unclear, however should probably not be surprising that those with more severe disease may still continue to shed virus as this is something that is also seen in patients with severe influenza.
Weinreich DM, Sivapalasingam S, Norton T, Ali S, Gao H, Bhore R, Xiao J, Hooper AT, Hamilton JD, Musser BJ, Rofail D, Hussein M, Im J, Atmodjo DY, Perry C, Pan C, Mahmood A, Hosain R, Davis JD, Turner KC, Baum A, Kyratsous CA, Kim Y, Cook A, Kampman W, Roque-Guerrero L, Acloque G, Aazami H, Cannon K, Simón-Campos JA, Bocchini JA, Kowal B, DiCioccio AT, Soo Y, Geba GP, Stahl N, Lipsich L, Braunstein N, Herman G, Yancopoulos GD; Trial Investigators. REGEN-COV Antibody Combination and Outcomes in Outpatients with Covid-19. N Engl J Med. 2021 Sep 29. doi: 10.1056/NEJMoa2108163. Epub ahead of print. PMID: 34587383.
This was this was a randomized control trial evaluating the antibiotic cocktail also known as regimen–cope and it is part of a larger trial that evaluated a different variety of patients, including those who are under the age of 18, and those who are pregnant and randomization. This study, patients were randomized in a 1: 1:1 ratio to receive either the IV antibiotic cocktail at a dose of 2400 mg or 8000 mg or placebo. During the trial, the methods were amended so that patients could receive a lower dose of the antibiotic cocktail either 1200 mg or 2400 mg along with placebo. The results of her palpitations and this article related to the lower dose compared with the placebo. The primary endpoint was the percentage of patients with at least 1 COVID-19 related hospitalization or death from any cause through day 29. 2519 patients with a least 1 risk factor for severe COVID-19 were assigned to either one of the 2 doses of the antibiotic cocktail or placebo. There were no significant differences in baseline characteristics between groups. For the primary outcome, Covid related hospitalization or death occurred in 1.3% of the 2400 mg group, compared to 4.6% of dose who were in the placebo group. Similarly 1% of the patients were randomized to the 1200 mg group as she had the primary endpoint compared to 3.2% and the corresponding placebo the relative risk reduction was 71.3% in the 2400 mg group and 70.4% in the 1200 mg group. The time to resolution of symptoms was the same in both antibody cocktail doses and was significantly shorter than the placebo.
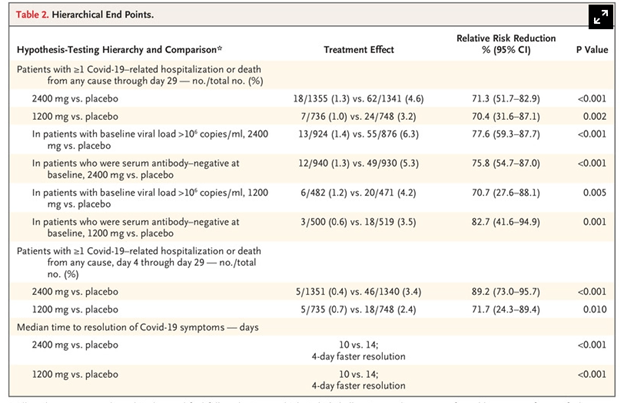
When he came to the safety endpoints, 4% of those in the placebo group had serious adverse events compared to anywhere from 1.1 to 1.7% in any of the 3 antibody cocktail groups. It should be noted however that these adverse events were considered to be complications of COVID-19 rather than the placebo itself.
I think these are reassuring news in terms of Covid therapeutics especially when it comes to this we are at high risk of complications. 1 thing that I would be curious about will be the establishment of infusion centers for this to meet certain criteria in the outpatient setting, and who would be eligible to receive this cocktail. I cannot imagine that this will be an affordable option to the vast majority of people who are at high risk.
Diaz GA, Parsons GT, Gering SK, Meier AR, Hutchinson IV, Robicsek A. Myocarditis and Pericarditis After Vaccination for COVID-19. JAMA. 2021 Sep 28;326(12):1210-1212. doi: 10.1001/jama.2021.13443. PMID: 34347001; PMCID: PMC8340007.
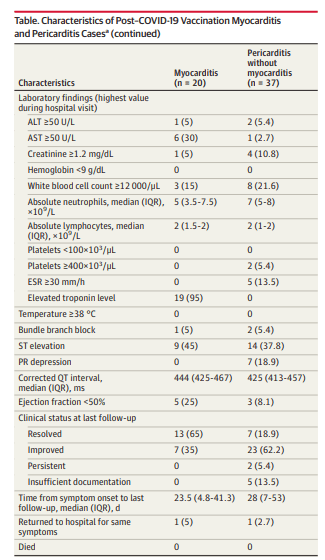
This was actually making any use recently. This was a retrospective cohort study that evaluated adverse events after vaccination. Data from 40 hospital was evaluated which included over 2 million individual individuals who received at least 1 dose of the COVID-19 vaccination. Most received one of the mRNA vaccine either at the Pfizer or Moderna. Overall myocarditis developed in 20 patients and pericarditis developing 37 patients. When comparing the prevaccine and post vaccine., There was a significant increase in the mean numbers of pericarditis and myocarditis. The mean monthly number of myocarditis was 16.93 vaccine compared to 78.8 after vaccination was available. Similarly, there was an increase in the cases of perimyocarditis from a mean monthly case number of 27.3-49.1. Of the 20 patients with myocarditis all of either had resolved symptoms improve while the vast majority of patients with pericarditis that improved at the time of the publication of the study. Despite these findings I do not think this should discourage people from taking the vaccinations given the overall benefit that the cephalad and decreasing the incidence of hospitalization and death.
Hall VG, Ferreira VH, Ku T, Ierullo M, Majchrzak-Kita B, Chaparro C, Selzner N, Schiff J, McDonald M, Tomlinson G, Kulasingam V, Kumar D, Humar A. Randomized Trial of a Third Dose of mRNA-1273 Vaccine in Transplant Recipients. N Engl J Med. 2021 Sep 23;385(13):1244-1246. doi: 10.1056/NEJMc2111462. Epub 2021 Aug 11. PMID: 34379917; PMCID: PMC8385563.
And one last Covid paper. This was a double-blind randomized control trial evaluating at third dose of the Moderna vaccine in patients who had received a solid organ transplant. Patients were randomized to to receive either a third dose of the vaccine or placebo 2 months after the second dose of the vaccine. The primary outcome was the serologic response as characterized by the anti-– receptor–binding domain antibody at month 4. 120 solid organ transplant recipients were enrolled, and at month for antibody level of at least 100 units/mL was present in 55% of the vaccine patients compared to 18% of this to receive placebo (relative risk, 3.1; 95% CI 1.7-5.8). Virus neutralization was also significantly higher in the vaccine group with a medium percent virus neutralization being 71% compared to 13% in the placebo group.
All right I think that is enough Covid for today.
Graham H R Smith, W Keith Henry, Daniel Podzamczer, Maria Del Mar Masiá, Christopher J Bettacchi, Keikawus Arasteh, Hans Jaeger, Marie-Aude Khuong-Josses, Maria Luisa Montes-Ramírez, Hans-Jürgen Stellbrink, Yazdan Yazdanpanah, Gary J Richmond, Kenneth C Sutton, Feifan Zhang, Cynthia C McCoig, Marty H St Clair, Kati Vandermeulen, Rodica Van Solingen-Ristea, Kimberly Y Smith, David A Margolis, William R Spreen, Efficacy, Safety, and Durability of Long-Acting Cabotegravir and Rilpivirine in Adults With Human Immunodeficiency Virus Type 1 Infection: 5-Year Results From the LATTE-2 Study, Open Forum Infectious Diseases, Volume 8, Issue 9, September 2021, ofab439, https://doi.org/10.1093/ofid/ofab439
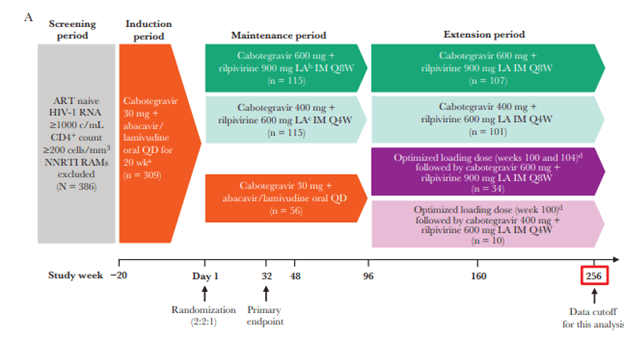
Now on to more interesting and fun articles. This was a study that evaluated the long term efficacy and safety of the combination of injectable cabotegravir and ripilvirine dosed at a interval of either every 4 weeks or every 8 weeks over a 5-year period. There was an induction period in eligible patients where they receive once daily oral cabotegravir, abacavir, and lamivudine for 20 weeks with daily recovering added during the last 4 weeks. Those were virally suppressed week for were then randomized in a 2:2:1 fashion to receive intramuscular injections every 8 weeks or every 4 weeks of the study drug, or remain in the oral therapy group. Cabotegravir and rilpilvirine were dosed at 600mg and 900 mg respectively when given every 8 weeks while they were dosed at 400 mg and 600 mg at every 4 weeks. After week 96, patients who were randomized to injectable therapy were able to continue into an extension. While those who were initially randomized to an oral therapy could switch to an injectable therapy either at every 8 weeks or every 4 weeks. Primary endpoint was a proportion of patients and the intention to treat urinalysis who should have viral suppression at week 32. The image below highlights the parent trial:
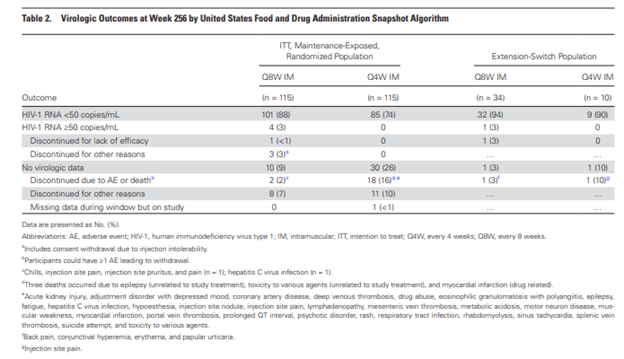
262 patients enter the extension phase, with patients who underwent an oral to injectable switch having a higher baseline CD4 and lower baseline viral load. It should be noted however that the extension switch group had a significantly lower number of patients. At the end of the study, 88% of patients in the q 8 weeks and 74% of patients in the every 4 week group achieved viral suppression. Similarly, in those who switched from oral to IM therapy, 94% of the q8w and 90% of the q4w cohort achieved viral suppression.
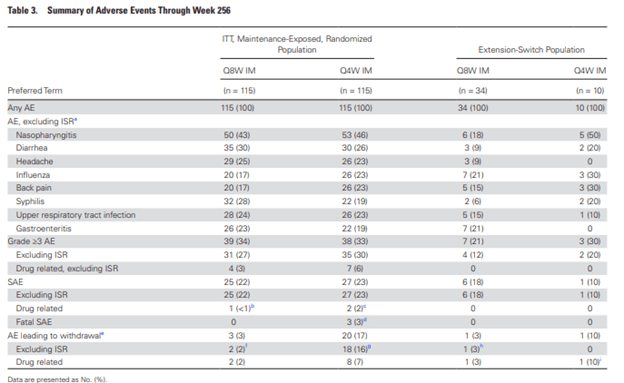
Adherence was over 90% and all injection groups. In terms of safety, all patients experience at least 1 adverse events. Surprisingly, the most common side effect was nasopharyngitis which occurred in 45% of patients. 67% of those who were initially randomized to injections and 77% who were subsequently switched to injections had a max grade 1/2 adverse events.
The future of ART looks quite bright with new data on 2-drug regimens and injectable drugs coming around. It is an exciting time for HIV care.
Thind SK, Shibib DR, Gentry CA. The Effect of Nomenclature Revision of Streptococcus bovis to Streptococcus gallolyticus on Subsequent Colon Cancer Screening. Open Forum Infect Dis. 2021 Sep 23;8(9):ofab426. doi: 10.1093/ofid/ofab426. PMID: 34568510; PMCID: PMC8458264.
Does changing the name of organisms lead to change in care? This study evaluated the change in name of Strep bovis to strep gallolyticus and its impact on people ordering colonoscopies to evaluate for colon cancer. Using data from the VA, this retrospective study was carried out from 2002 through 2017 and compared the rates of colonoscopies ordered in those whose organism was labeled as S. bovis or S. gallolyticus. 1181 unique cases of streptococcus bacteremias were evaluated. Primary outcome was intent for colonoscopy rather than the receipt of the colonoscopy instead. Overall, the intent was present in 62% of all cases, with no difference between the S. gallolyticus group (66.5%) or the S. bovis group (62%). This held true even when looking at those over or under the age of 75.
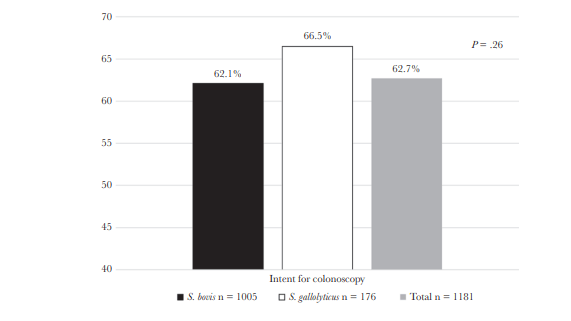
Similarly, there was no difference in the rate of GI consults between cohorts, though there was a higher rate of ID consultation in the S. gallolyticus cohort regardless of age (61% vs 49% if aged >75; 66% vs 49% if aged <75). There was a higher rate of consults for both GI/ID in the Strep gallolyticus group (85% vs 78%), but no change in mortality. Univariate and multivariate analysis did not find that the name change was associated with intent for colonoscopy, but it did find that ID consultation had a positive effect:
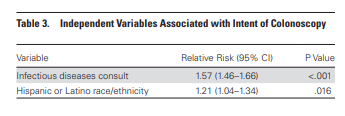
I think the name change has been present enough for people to realize the association between S. gallolyticus bacteremia and colon cancer, but I still hear S. bovis is the one that is thought instead.
Boxhoorn L, van Dijk SM, van Grinsven J, Verdonk RC, Boermeester MA, Bollen TL, Bouwense SAW, Bruno MJ, Cappendijk VC, Dejong CHC, van Duijvendijk P, van Eijck CHJ, Fockens P, Francken MFG, van Goor H, Hadithi M, Hallensleben NDL, Haveman JW, Jacobs MAJM, Jansen JM, Kop MPM, van Lienden KP, Manusama ER, Mieog JSD, Molenaar IQ, Nieuwenhuijs VB, Poen AC, Poley JW, van de Poll M, Quispel R, Römkens TEH, Schwartz MP, Seerden TC, Stommel MWJ, Straathof JWA, Timmerhuis HC, Venneman NG, Voermans RP, van de Vrie W, Witteman BJ, Dijkgraaf MGW, van Santvoort HC, Besselink MG; Dutch Pancreatitis Study Group. Immediate versus Postponed Intervention for Infected Necrotizing Pancreatitis. N Engl J Med. 2021 Oct 7;385(15):1372-1381. doi: 10.1056/NEJMoa2100826. PMID: 34614330.
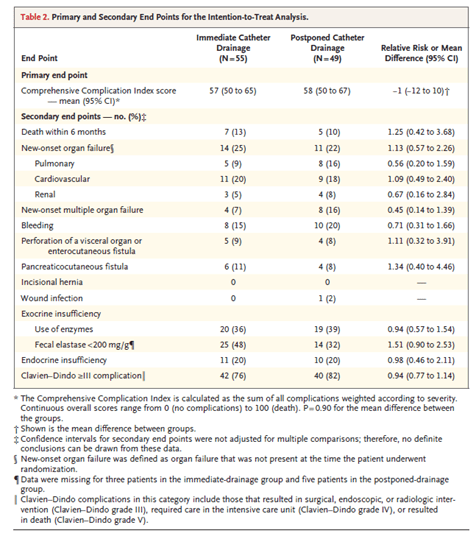
Now moving on to pancreatitis. This was a multicenter, randomized superiority trial designed to investigate with immediate drainage of infected pancreatic or peripancreatic necrosis was superior to the lady drainage. Caroline suggest that drainage of peripancreatic necrosis to be delayed until the necrosis becomes encapsulated. In the study, patients were randomized in 1:1 ratio to either immediate catheter drainage or postpone catheter drainage. Dose in the immediate drainage group were treated with antibiotics and drainage within 24 hours of randomization will dose in the delayed drainage group catheter procedure delayed until the station of walled off necrosis. Primary endpoint was a score on the comprehensive complication index with higher scores up to 100 suggesting more complications. For follow-up for 6 months urinalysis were performed and attention to treat fashion. 104 patients where randomly assigned with no significant differences in her baseline characteristics. Overall, there was no difference between the groups in terms of the mean comprehensive complication index score (57 in the immediate drainage group in 58 and post bone drainage prescription). Further there was no difference in the length of hospital stay or ICU stay. There was a higher number of interventions performed in the immediate drainage drip compared to the possible drainage group (4.4 versus 2.6). A lower number of patients in the postpone drainage group required and necrosectomy (11 versus 28).
Yun Woo Lee, Seongman Bae, Eunmi Yang, Hyemin Chung, Eunsil Kim, Jiwon Jung, Min Jae Kim, Yong Pil Chong, Sung-Han Kim, Sang-Ho Choi, Sang-Oh Lee, Yang Soo Kim, Clinical and Microbiological Characteristics of Hospital-Acquired Methicillin-Resistant Staphylococcus aureus Bacteremia Caused by a Community-Associated PVL-Negative Strain, Open Forum Infectious Diseases, Volume 8, Issue 9, September 2021, ofab424, https://doi.org/10.1093/ofid/ofab424
This study compared the clinical outcomes of CA-MRSA isolates that carried the PVL gene and those who did not. Specifically, ST72-CCmecIV, which does not carry the PVL gene was compared to ST5-SCCmecII, which does carry it. This Korean prospective study compared 583 hospital acquired cases. When comparing the presentation between the cohorts, those with the PVL-carrying strain were older, less likely to have hematological malignancies, more likely to have had prior antibiotic treatment and MRSA colonization, and more likely to have CVC placement. Further, a higher percentage of the non-PVL cohort had non-eradicable foci of infection such as pneumonia (43% vs 37%). Those with the PVL-carrying strain were more likely to be in the ICU (36% vs 13%), however there was no significant difference in 30-day or 90-day mortality, with multivariate analysis finding that the strain was not a contributor to 30-day mortality:
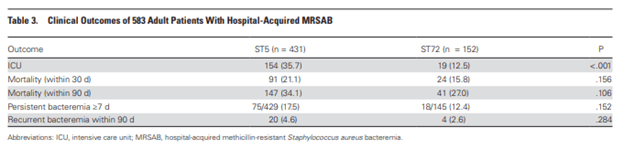
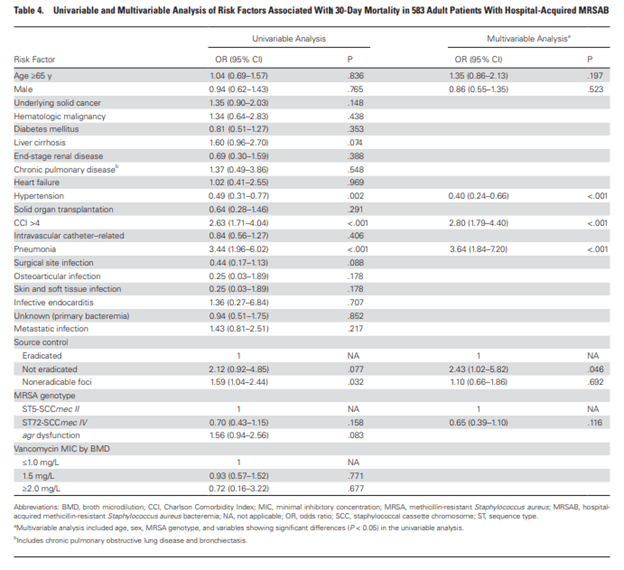
Meanwhile, higher co-morbidities index, pneumonia as site of infection, non-eradicated foci were all associated with higher mortality. Another interesting bit was that the PVL-carrying strain had higher vancomycin MIC and was more likely to be resistant to several antibiotics, including clindamycin, ciprofloxacin, and gentamicin:
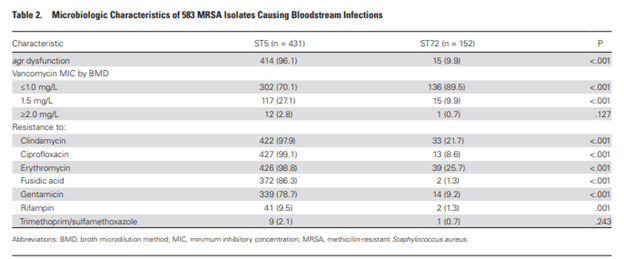
I still content PVL-carrying strains are more terrifying, especially with those resistant profiles. The non-eradicable bit in the non-PVL strain was a bit surprising, but seems the PVL-group was sicker to begin with.
Saccente M, Krishnan G. Comparison of Blood (1->3)-β-d-Glucan Levels in AIDS-Related Pneumocystis jirovecii Pneumonia and AIDS-Related Progressive Disseminated Histoplasmosis. Clin Infect Dis. 2021 Sep 15;73(6):1100-1102. doi: 10.1093/cid/ciab277. PMID: 33786591.
How does beta-D glucan differ between PJP and pulmonary histoplasmosis in AIDS patients? This was a retrospective study evaluating patients with HIV who had either PJP/PCP or pulmonary histoplasmosis and had a BD glucan measured using the fungitell assay. 47 patients with histoplasmosis and 41 patients with PCP were compared. Overall, the median BD glucan was higher in those with PCP compared with those of histoplasmosis (500 vs 465, p =0.017). Further, a higher percentage of patients with PJP had a BD glucan >500:
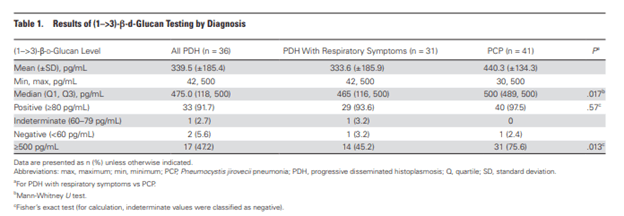
With a cut-off of 80 pg/mL or greater, sensitivity of BD glucan was 92%, 94%, and 98% for pulmonary histoplasmosis, pulmonary histoplasmosis with respiratory symptoms, and PJP, respectively. So BD glucan is higher in those with PJP, but I do not find this to be necessarily helpful in the clinical setting. Either way, I think this is an interesting study.
Claeys KC, Schlaffer K, Smith R, Hitchcock S, Jiang Y, Evans S, Johnson JK, Leekha S. Day at the Races: Comparing BioFire FilmArray Blood Culture ID Panels With Verigene Blood Culture Panel in Gram-Negative Bloodstream Infections Using DOOR-MAT Analysis. Clin Infect Dis. 2021 Sep 15;73(6):1103-1106. doi: 10.1093/cid/ciab262. PMID: 33772269; PMCID: PMC8442774.
This was a proof-of-concept studying evaluating 3 different rapid diagnostic tests (BioFire FilmArray, BioFire BCID2 and Verigene Blood culture assay) in gram-negative bacteremias and to compare the theoretical antibiotic prescribing. Gold-standard assay for susceptibility was via VITEK MS/VITEK 2. Theoretical antibiotic therapy decisions was made by an ID- trained doctor who reviewed RDT results from each panel, which were made based on each patient variable and further compared with what actually happened. 103 blood cultures were evaluated, which yielded 108 organisms. Verigene panel had a positive percent agreement (PPA) of 98.8%, BioFire had a PPA of 97.8%, and BioFire BCID 2 had a PPA of 96.9%. Using a DOOR-Mat spectrum scoring system (see below, but basically, higher the better), Verigene had a higher mean score (83.8) compared to BioFire BCID (59.9) and 89.7% BioFire BCID 2.
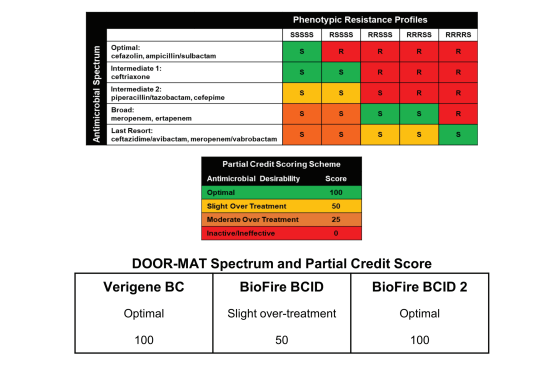
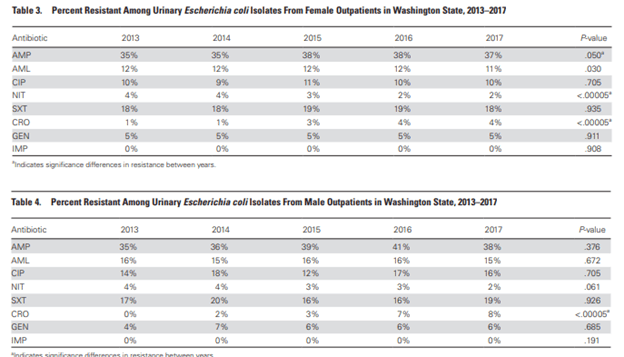
Frisbie L, Weissman SJ, Kapoor H, D’Angeli M, Salm A, Radcliff J, Rabinowitz P. Antimicrobial Resistance Patterns of Urinary Escherichia coli Among Outpatients in Washington State, 2013-2017: Associations With Age and Sex. Clin Infect Dis. 2021 Sep 15;73(6):1066-1074. doi: 10.1093/cid/ciab250. PMID: 33768228.
This was a retrospective cross-sectional study evaluating antibiotic susceptibility for E.coli isolates from urinary specimens in outpatients in Washington State during a 5 year period. 24,215 patients were included in the cohort, with ampicillin and TMP-SMX being more likely to be resistant with regards to oral antibiotics for all ages. Women >50 had a higher percentage of resistance across all antibiotics except for TMP-SMX, while men >50 had an increase resistance for Cipro, ceftriaxone, gentamycin, and imipenem. Ampicillin and amox-clav had lower resistance percentages in men >50 compared to other age groups.
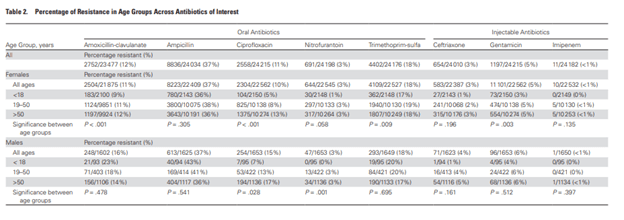
Resistance for ceftriaxone increased for both men and women over the study period, however rest remained the same.
Using logistic regression, age was correlated with higher odds of resistance for amox-clav, Cipro, ceftriaxone, and gentamicin for females, and for amox-clav and Cipro for men:

I am a bit surprised that Cipro resistance was not higher, but I am assuming people are not writing for it as much. Ampicillin resistance is not surprising, but I think macrobid should be a go-to for many going forward.