If I had to name an infection that gives people nightmares, besides COVID as of the time of writing this, would be Mucormycosis. These are moulds of the order Mucorales, which are comprised of multiple species, including Rhizopus spp, Mucor spp, and Lichtheimia spp (formerly of the genera Absidia and Mycocladus). Others include Rhizomucor spp, Cunninghamella spp, Apophysomyces spp, and Saksenae spp (1). The timely diagnosis of this mould is difficult, mostly because the risk factors for it are similar to risk factors of Aspergillus spp (namely, profound neutropenia, corticosteroid use, HSCT, hematological malignancies, and unique to Mucormycosis, poorly controlled diabetes):

Further complicating things is the fact that both BD glucan and galactomannan do not detect the antigen components of its cell wall (2) and fungal culture detects invasive moulds anywhere from 30 to 52% of the time (3). This is largely due to the friable nature of the nonseptated hyphae, which are frequently damaged during tissue manipulation (2). While I am not going to go into detail into the diagnosis of mucormycosis, you can see it is incredibly difficult and in many cases, the diagnosis is based on empiric evidence, such as imaging of sinuses or chest, lack of BD glucan or galactomannan (unless there is co-infections with other moulds), and histopathology (which can be modified by technical artifacts). In most cases, we end up having to rely on empiric therapy.
What Antifungals can we use?
In general, these are the antifungals you can use:
- Posaconazole
- Isavuconazole
- Amphotericin B
- Ecchinocandins can be used, but in general they should not be used unless you have someone smarter than you here.
Most of this data derives from in vitro studies testing MIC due to several methods, however there is no consensus as far as the “clinical breakpoints” are concerned (in other words, we do not know what to do with the MICs for each drug-bug combination and its relationship to outcomes and adverse events. So an MIC of 2 for amphotericin B doesn’t mean sensitive or resistant, though in my simple mind, lower = better). For instance, one retrospective study found that an MIC of <0.5 for amphotericin in Mucorales spp was associated with better outcomes at week 6 (4).
In one study of 37 isolates, the MIC was evaluated for 4 azoles (excluding isavuconazole) and amphotericin B (5):
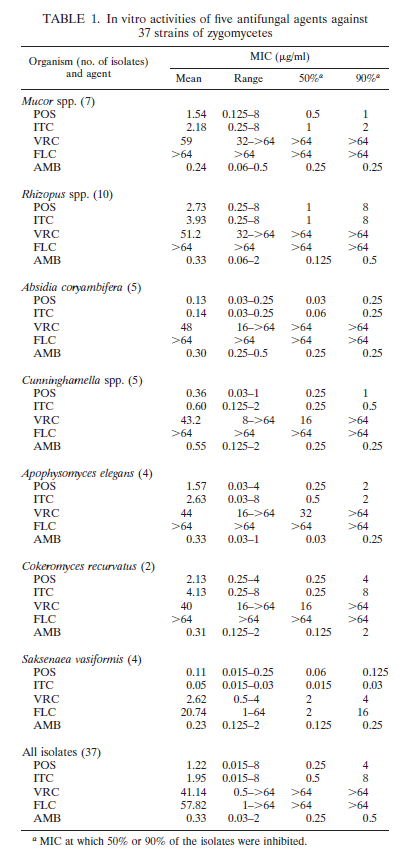
Posaconazole and amphotericin had the most consistent MIC across species (you may have noticed Itraconazole can has MICs comparable to that of the other 2 drugs, however given its issues with absorption, lets ignore it). Another in vitro study evaluated the isolates of Mucorales against EUCAAST and CLSI references methodologies with the more common antifungals used in these scenarios (6):
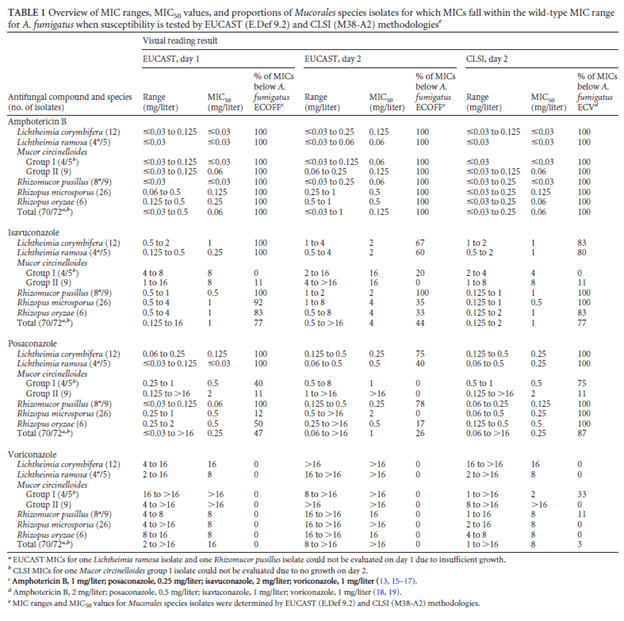
Again, the MICs are consistently low for amphotericin B, posaconazole, and with some exceptions, isavuconazole. Voriconazole tends to stay behind the other antifungals in this area.
Empiric therapy: Amphotericin vs Isavuconazole
The crux of the problem is getting effective antifungal therapy as soon as possible. In a retrospective study (7) of hematological patients with mucormycosis, delaying therapy beyond 6 days was associated with a 2-fold increase in mortality rate at 12 weeks (48.6% vs 82.9%, p-value 0.03):

Delaying therapy was associated with an OR of 8.1 (95% CI 1.7-38.2, p-value 0.008) in a multivariate analysis. Liposomal amphotericin B has been the most extensively used therapy here. Guidelines suggest (2) its use as first line therapy, with 5mg/kg being the usual dose that is recommended. By far, most of the “data” comes from case series (8). While the dosing is usually 5mg/kg, dosing as high as 10mg/kg have been used in certain case series (9). In a multicenter, randomized control trial (10), patients with proven or probable invasive mould infection were randomized to either 3mg/kg or 10mg/kg of liposomal amphotericin B for the first 14 days followed by 3mg/kg. 107 patients were in the standard dose group and 94 were in the high-dose group. Most of the patients had invasive aspergillosis. At the end of study drug treatment, survival was 93% in the standard dose group vs 88% in the high dose group (difference 4%, 95% CI -4% to 12%, p-value >0.05) but the rates of survival at week 12 were 72% and 59% respectively (difference 13%, 0.2% to 26%, p-value >0.05).
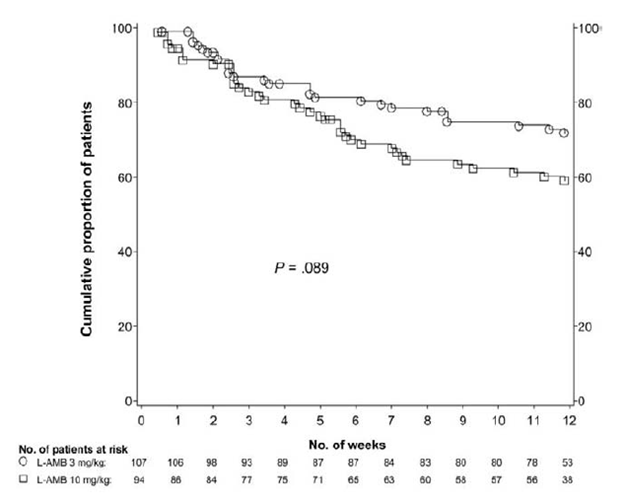
While this suggest no difference between standard and high dose, there were significantly more adverse events in the high dose group:
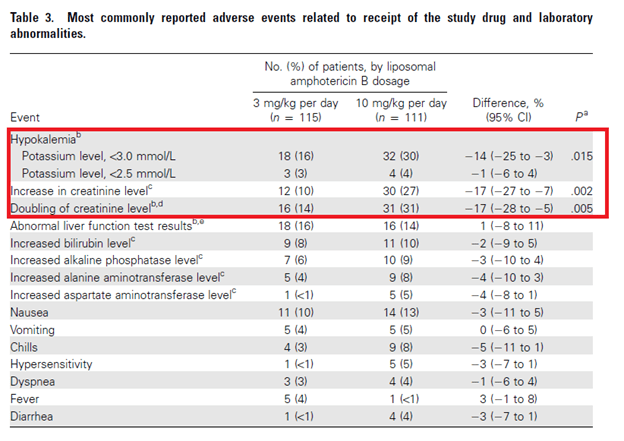
Recently, isavuconazole has surfaced as an alternative to liposomal amphotericin B for mucormycosis. It has had comparable results to voriconazole for aspergillus (11). A single arm, open label multicenter trial (12) evaluated the designed to determine the efficacy and safety of isavuconazole for the treatment of rare moulds. Patients were treated with isavuconazole and were matched with controls from the FungiScope Registry who received amphotericin B and were analyzed for 42 day all-cause mortality. 21 patients received isavuconazole for primary treatment and were matched with 33 FungiScope controls treated with amphotericin B. Day 42 crude all cause mortality was 7/21 (33%) in the primary treatment isavuconazole cases compared to 13/33 (39%) of amphotericin B-treated matched controls (weighted all-cause mortality: 33% vs 41%, p-value 0.595):

While this is not a giant cohort (indeed, this is not even a properly randomized trial), this is as best as we can do for the time being. Indeed, recruiting enough patients for a very where there is no real standard of care for a proper randomized trial is a herculean task that would take several years to accomplish (13). This suggests that isavuconazole may be a reasonable alternative for liposomal amphotericin B in certain scenarios, such as in those with renal failure.
Dual Therapy
I mentioned echinocandins cannot really be used for therapy in murcormycosis. Indeed, it has no in vitro activity against most moulds of this family, so it has no utility for empiric therapy. Despite this, murine models of Rhizopus oryzae have demonstrated limited echinocandin activity. In a murine model (14) of mice with DKA and R. oryzae, mice were treated with a combination of liposomal amphotericin and different doses of micafungin or anidulafungin. Liposomal amphotericin B was combined with micafungin at 1mg/kg/day or 3mg/kg/day in one arm and with anidulafungin 1mg/kg/day or 10mg/kg/day in another arm. In both, combination therapy resulted in a higher percentage of survival but only at certain doses (1mg/kg/day for micafungin and 10mg/kg/day for anidulafungin), suggesting a class effect:
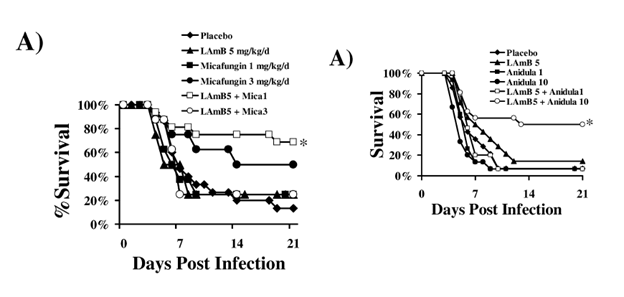
This effect was also seen in the neutropenic model:

A retrospective study of rhino-cerebral mucormycosis evaluated the outcomes of dual therapy compared to those who did not get dual therapy (15) with primary endpoint being success at 30 days after hospital discharge. In general, there was higher success rate in those treated with a combination of amphotericin B and echinocandin (usually caspofungin), though only 7 patients received the combination therapy. Notably, the use of ABLC was associated with worse treatment success when compared to AmB or lipid amphotericin B, but combination of ABLC plus caspofungin yielded more success than when used alone:
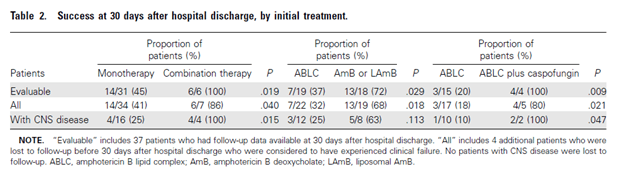
Bivariate and logistic regression analysis also demonstrated that combination therapy was successful in this group, though the numbers are fairly small to draw any definitive conclusions.

Another retrospective study of hematological patients evaluated the outcomes of patients with mucormycosis treated with monotherapy or combination therapy (16). Primary outcome was survival 6 weeks after treatment. 106 patients were identified, of which 47 received monotherapy and 59 received combination therapy (LAMB + posaconazole or LAMB + caspofungin). The propensity score-adjusted probability of 6-week survival for those who got monotherapy was 56% compared to those who got combination therapy, 60% (p-value 0.71), suggesting no benefit for combination therapy:

Step Down Therapy
If you opt to use liposomal amphotericin B for induction therapy, how long do you treat before stepping down to PO isavuconazole or posaconazole? No one really knows. In general, therapy can be weeks to months long. One review (2) suggested the use of 3 weeks of IV amphotericin followed by stepdown therapy for maintenance therapy. Total duration depends on symptom and radiological resolution, as well as reversal of underlying immunosuppression (read: stopping chemo and return of neutrophils). The practice seems to be to administer posaconazole if using this as stepdown and overlap it for 1-2 weeks with amphotericin B to achieve a trough of >1ug/mL, with some preferring >2ug/mL (2). While I have seen people get isavuconazole troughs, no one knows what an appropriate trough is.
Other Interesting Tidbits:
The rapid recovery of neutrophils is an important component of adjunctive therapy. For instance, neutrophil recovery (16) was found to be a predictor of mortality on a combination study (38% vs 89% mortality, p-value 0.001) and in another study in its multivariate analysis, PMN recovery had an OR of 0.07 (95% CI 0.01-0.5) for mortality (7).
Another therapeutic adjunct has been iron-chelators. There is a link between iron availability and risk of mucor and a multicenter randomized trial attempted to evaluate the safety of the combination of deferasirox and LAmB (17). 20 patients were randomized, 11 to deferasirox and 9 to placebo, with only 6 patients in the deferasirox group getting at least 4 doses of the study medications compared to 8 of the placebo trial, and only 4 patients completed the 14 days of therapy in the treatment group vs 7 in the placebo arm. Mortality rate at 30 days was 45% in the deferasirox arm vs 11% in the placebo arm (p-value 0.1) and 82% vs 22% at 90 days, respectively (p-value 0.01).
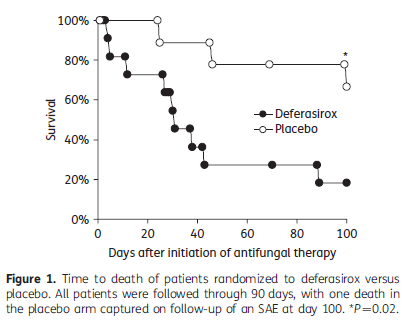
There were more adverse events in the deferasirox arm (12 vs 7) as well. While this was a phase II trial with issues, these data do not suggest the use of iron chelation as initial therapy.
TL;DR
- There is no RCTs for the therapy of mucormycosis, due to the rarity of this infection. Most of it comes from case series
- Delay of appropriate therapy beyond 6 days is associated with worse prognosis
- Amphotericin B or isavuconazole as empiric therapy are reasonable choices. While data is lacking, going beyond Liposomal Amphotericin B at 5mg/kg will not yield better results
- Liposomal amphotericin B > all others
- Posaconazole is also a reasonable option as a stepdown therapy (overlap it with LAmB for at least a week and get a trough of at least >1ug/mL)
- Duration of therapy depends on imaging and clinical characteristics, as well as removal of risk factors (i.e. no more DKA, restoring of neutrophils). Typically this is several weeks long, with one review suggesting at least 3 weeks of LAmB.
- Small role for combination therapy with echinocandins and LAmB, but data is sparse
- No use for iron chelators
References:
- Cornely OA, Alastruey-Izquierdo A, Arenz D, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19(12):e405-e421. doi:10.1016/S1473-3099(19)30312-3
- Kontoyiannis DP, Lewis RE. How I treat mucormycosis. Blood. 2011;118(5):1216-1224. doi:10.1182/blood-2011-03-316430
- Tarrand JJ, Lichterfeld M, Warraich I, et al. Diagnosis of invasive septate mold infections. A correlation of microbiological culture and histologic or cytologic examination. Am J Clin Pathol. 2003;119(6):854-858. doi:10.1309/EXBV-YAUP-ENBM-285Y
- Lamoth F, Damonti L, Alexander BD. Role of Antifungal Susceptibility Testing in Non-Aspergillus Invasive Mold Infections. J Clin Microbiol. 2016;54(6):1638-1640. doi:10.1128/JCM.00318-16
- Sun QN, Fothergill AW, McCarthy DI, Rinaldi MG, Graybill JR. In vitro activities of posaconazole, itraconazole, voriconazole, amphotericin B, and fluconazole against 37 clinical isolates of zygomycetes. Antimicrob Agents Chemother. 2002;46(5):1581-1582. doi:10.1128/aac.46.5.1581-1582.2002
- Arendrup MC, Jensen RH, Meletiadis J. In Vitro Activity of Isavuconazole and Comparators against Clinical Isolates of the Mucorales Order. Antimicrob Agents Chemother. 2015;59(12):7735-7742. doi:10.1128/AAC.01919-15
- Chamilos G, Lewis RE, Kontoyiannis DP. Delaying amphotericin B-based frontline therapy significantly increases mortality among patients with hematologic malignancy who have zygomycosis. Clin Infect Dis. 2008;47(4):503-509. doi:10.1086/590004
- Shoham S, Magill SS, Merz WG, et al. Primary treatment of zygomycosis with liposomal amphotericin B: analysis of 28 cases. Med Mycol. 2010;48(3):511-517. doi:10.3109/13693780903311944
- Lanternier F, Poiree S, Elie C, et al. Prospective pilot study of high-dose (10 mg/kg/day) liposomal amphotericin B (L-AMB) for the initial treatment of mucormycosis. J Antimicrob Chemother. 2015;70(11):3116-3123. doi:10.1093/jac/dkv236
- Cornely OA, Maertens J, Bresnik M, et al. Liposomal amphotericin B as initial therapy for invasive mold infection: a randomized trial comparing a high-loading dose regimen with standard dosing (AmBiLoad trial). Clin Infect Dis. 2007;44(10):1289-1297. doi:10.1086/514341
- Maertens JA, Raad II, Marr KA, et al. Isavuconazole versus voriconazole for primary treatment of invasive mould disease caused by Aspergillus and other filamentous fungi (SECURE): a phase 3, randomised-controlled, non-inferiority trial. Lancet. 2016;387(10020):760-769. doi:10.1016/S0140-6736(15)01159-9
- Marty FM, Ostrosky-Zeichner L, Cornely OA, et al. Isavuconazole treatment for mucormycosis: a single-arm open-label trial and case-control analysis. Lancet Infect Dis. 2016;16(7):828-837. doi:10.1016/S1473-3099(16)00071-2
- Perfect JR, Cornely OA, Heep M, et al. Isavuconazole treatment for rare fungal diseases and for invasive aspergillosis in patients with renal impairment: Challenges and lessons of the VITAL trial. Mycoses. 2018;61(7):420-429. doi:10.1111/myc.12769
- Ibrahim AS, Gebremariam T, Fu Y, Edwards JE Jr, Spellberg B. Combination echinocandin-polyene treatment of murine mucormycosis. Antimicrob Agents Chemother. 2008;52(4):1556-1558. doi:10.1128/AAC.01458-07
- Reed C, Bryant R, Ibrahim AS, et al. Combination polyene-caspofungin treatment of rhino-orbital-cerebral mucormycosis. Clin Infect Dis. 2008;47(3):364-371. doi:10.1086/589857
- Kyvernitakis A, Torres HA, Jiang Y, Chamilos G, Lewis RE, Kontoyiannis DP. Initial use of combination treatment does not impact survival of 106 patients with haematologic malignancies and mucormycosis: a propensity score analysis. Clin Microbiol Infect. 2016;22(9):811.e1-811.e8. doi:10.1016/j.cmi.2016.03.029
- Spellberg B, Ibrahim AS, Chin-Hong PV, et al. The Deferasirox-AmBisome Therapy for Mucormycosis (DEFEAT Mucor) study: a randomized, double-blinded, placebo-controlled trial. J Antimicrob Chemother. 2012;67(3):715-722. doi:10.1093/jac/dkr375
