If I had to choose an enemy to my pretend superhero that enemy would be AIDS-related Kaposi Sarcoma (well, Staphylococcus Aureus but KS is a close second). I’ve seen several cases that did not end up in a good outcome during my training and I can recall only one where everything went well. In the Western World, KS is not a disease that is commonly seen given the advent of HAART, however in Sub-Saharan Africa it is still a major cause of morbidity and mortality. I find this disease interesting, however I will not go into great detail about the chemotherapeutic aspects of this disease (this is why we have oncologists, after all. I cannot keep up with their chemotherapy abbreviations that use both brand and generic names. Choose one or the other!).
Introduction
The etiology of Kaposi’s sarcoma is HHV-8 (1), also known as Kaposi sarcoma-associated herpes virus. This type of malignancy is a multicentric angioproliferative neoplasm, and HHV-8 has been implicated in the development of other diseases such as multicentric Castleman’s disease. Risk factors for the development of KS includes severe immunosuppression, African or Sub-Saharan origin, and MSM. For instance, one retrospective analysis found that patients who were MSM or had lower CD4 cell counts were more likely to have KS (2):
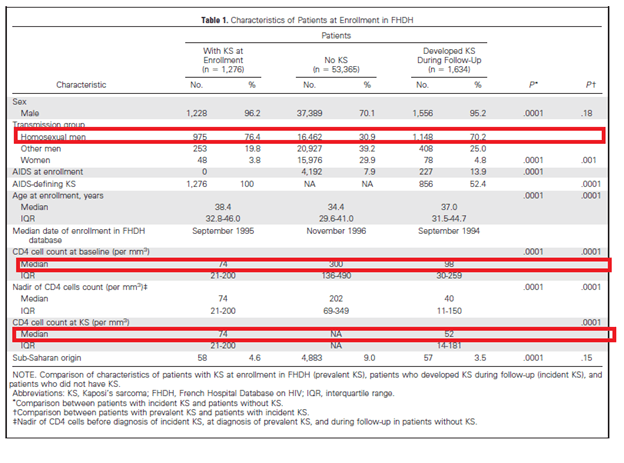
This pattern also held true in the EuroSIDA cohort (3):

As well as in a Swiss cohort (4):
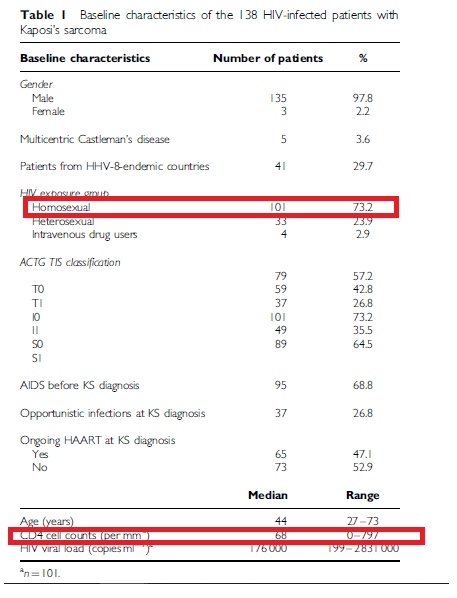
The reasons for MSM being a significant risk factor is unclear, as seen above, these patients have higher rates of KS even above those who acquired HIV via IV drug abuse. Other risk factors include immunosuppression (i.e. lower CD4 count), although the advent of widely available ART has improved CD4 counts and dropped viral loads. In a retrospective study (6) of 555 patients diagnosed with KS, the incidence of KS decreased with rising CD4 counts in both the naïve follow up group and the cART follow up group:
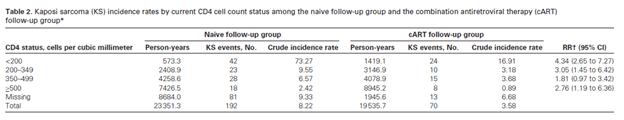
Further, the incidence of KS dropped the longer patients were on cART:
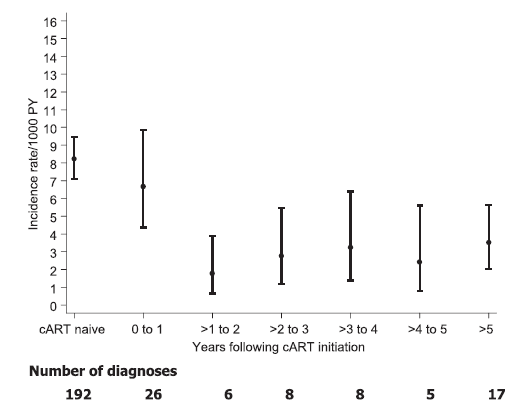
Though, if patients had a lower CD4 count despite being on cART, this was also associated with significant risk for developing KS:
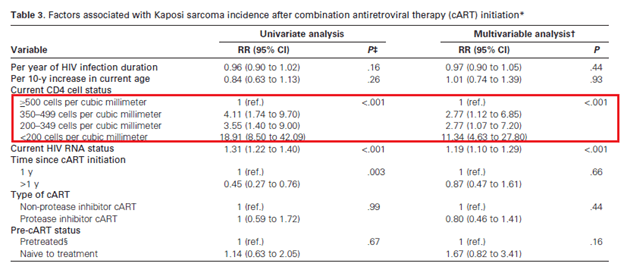
This was also seen in the EuroSIDA cohort (4), with those patients that tended to develop KS having lower CD4 counts at initiation of ART as well as 6 months into ART therapy:
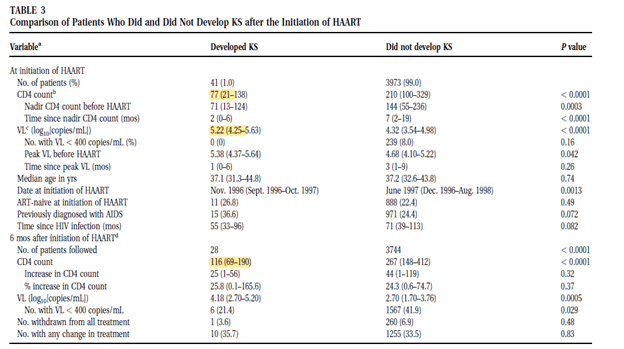
Steroids are also cited as a risk factor for development of KS, but this is limited to case studies. For instance, one letter to the editor (6) presented 2 patients who were given low dose prednisone for PMR and rheumatoid arthritis, while another case series (7) described 3 patients with unmasking of Kaposi Sarcoma in patients being treated for TB-IRIS.
Classification of Disease
The classification of AIDS-related Kaposi sarcoma used is the AIDS Clinical Trials group, which has important treatment and prognostic implications (8):

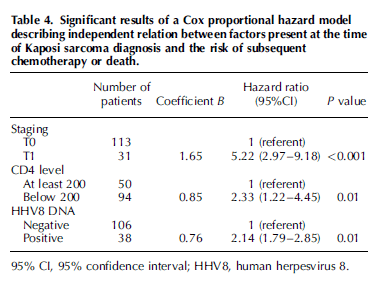
For instance, in a retrospective Swedish cohort (9), factors that were associated with a poor outcome (need for chemotherapy or death) were tumor stage and immune status:
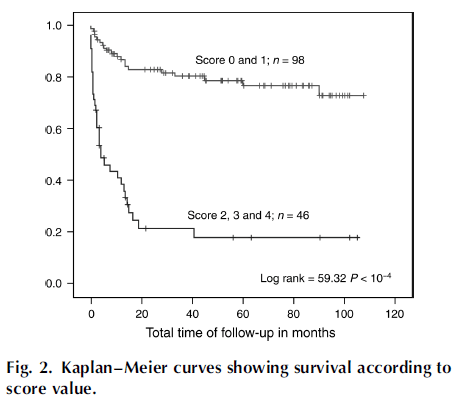
Using a scoring system (T1 = 2 points, low CD4 and positive HHV8 DNA = 1 point), the Kaplan-Meier curve demonstrated a significant increase in mortality with scores >2:
Indeed, British guidelines (8) and Cochrane reviews (10) recommend systemic chemotherapy in conjunction with ART in those patients with tumor stage 1.
Role of ART
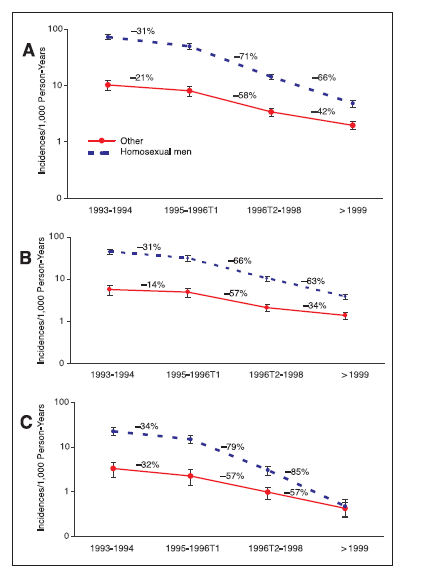
The impact of ART has had on the incidence of KS is not to be understated. For instance, the incidence rates of KS has been dropping since the early 90s as seen in several studies. A cohort study (11) found the incidence of KS dropped since 1993:
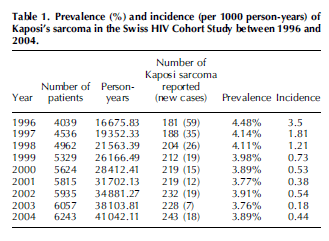
Looking at the previously quoted Swiss study (8) the incidence of KS dropped since the mid 1990s, though the prevalence remained the same:
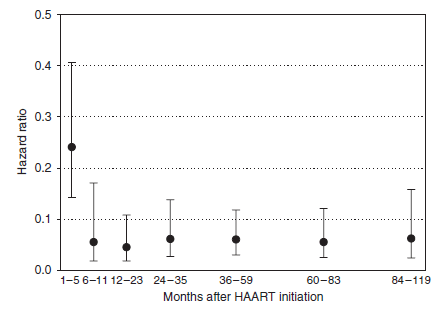
This was also seen in a retrospective study (12), where the HR for development KS dropped following ART initiation:
As well having a significant reduction in incidence:
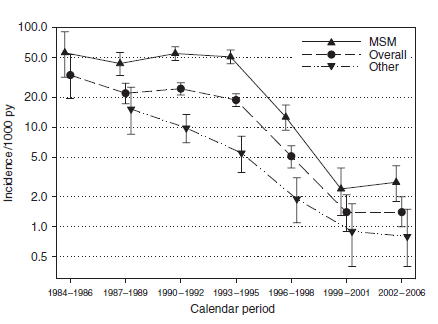
A similar trend was seen in the EuroSIDA cohort (4), with incidence decreasing from 24.7 per 1000 person-years follow up (PYFU) in 1994 down to 1.7 per 1000 PFYU in 2002.
How ART plays a role beyond immunomodulation in the regression of KS is unknown. One review (13) suggested that immune reconstitution, inhibition of HIV-1 replication and decrease of Tat protein, and reduction of intracellular cytokine production may impart anti-angiogenic effects against KS. The transactivating protein, aka Tat, has been found to induce growth of KS spindle cells in vitro, protect KSHV-positive cell lines from chemotherapy, and is angiogenic in transgenic mice (13), suggesting this plays a role in the pathogenesis of AIDS-related KS. Indeed, for some time it was believed that PI-based ART regimens were better suited for KS given its findings in blocking the development of angio-proliferation by blocking basic fibroblast growth factor and vascular endothelial growth factor. Despite this, clinical data suggest that PI-based ART regimens are not required for improvement. For instance, in a retrospective study (14) of over 56,000 HIV infected patients, the HR for PI and NNRTI based ART regimens were similar, though NRTI had a higher risk ratio, it was still associated with lower risk:

Another retrospective cohort (15) of 138 patients with HIV-1 found that PI-based regimens and non-PI based regimens had similar efficacy over a 24 month period for KS remission:
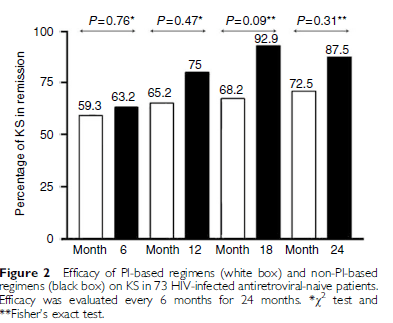
Any type of ART regimens seem to work well, though as noted previously, the risk of KS depends on the CD4 count and VL following ART initiation (12):

ART vs ART + Chemotherapy
As noted previously, chemotherapy is usually recommended for patients with advanced tumor staging. In these patients, the combination of chemotherapy and ART usually improves outcomes. A prospective, single-center, RCT compared ART vs ART + chemotherapy (16). This trial involved patients with cutaneous or visceral disease, though 89% had visceral disease. Patients were given stavudine, lamivudine, and nevirapine and chemotherapy consisted of doxorubicin, bleomycin, and vincristine every 3 weeks. Primary outcome was KS response at 12 months. 59 patients were randomized to ART and 53 randomized to ART and chemotherapy. There was a lot of cross over, with 22% in the ART group receiving chemotherapy and 28% of the chemo group getting only ART. Patients in the chemotherapy group had a better response in comparison to the ART only group, which was mostly driven by partial response. Those in the ART group had progression of disease. Notably, there was no difference in mortality between the groups:
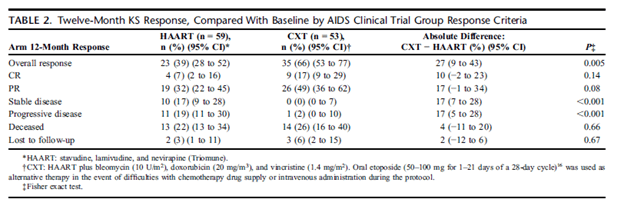
Progression free survival was better for the chemotherapy group vs the ART only group (HR 0.52, 95% CI 0.31-0.88):
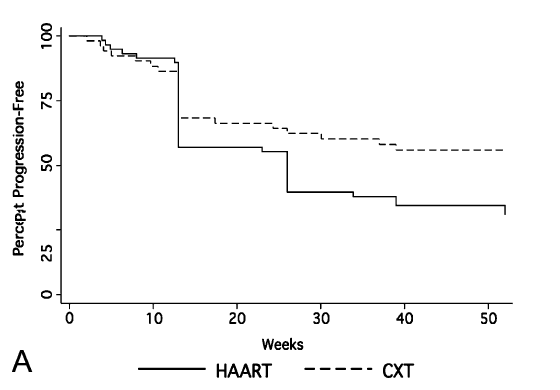
In a small open label, randomized controlled trial of 28 patients (17), 13 patients were randomized to chemotherapy + ART vs 15 in the ART group alone. At 48 weeks, patients in the ART + chemo group had a significantly higher response rate when compared to the ART only group:
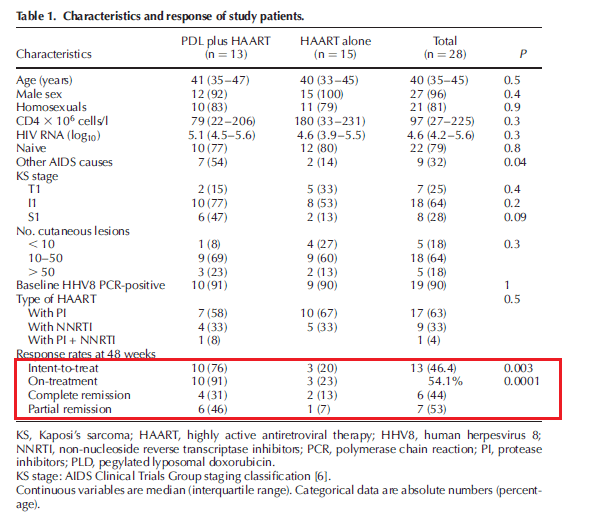
The survival benefit of combination therapy was also evaluated in an analysis of a phase II trial (18) comparing the efficacy and safety of bleomycin/vincristine and gemcitabine. All patients were on combination with ART. Overall survival was 85.7% (60 out of a total of 70 patients) at the end of the 3 year period which was better than the studies the authors quoted which included survival rates of 57-60%.

One thing to note is that most of these patients had good performance status, with an exclusion criteria being an ECOG of >2 so these patients may have done much better due to this fact. Regardless this seems to suggest that in more advanced disease, combination with chemotherapy seems to be a reasonable approach in combination with ART.
IRIS
I mentioned before that in AIDS patients who are started on ART, they run the risk of developing immune reconstitution inflammatory syndrome if they are co-infected with either tuberculosis or cryptococcus. These tend to be either unmasking (never knew you had an opportunistic infection until your immune system kicked into overdrive) or paradoxical (your underlying OI got worse despite being treated for it). If you recall that post, starting ART in tuberculosis is not associated with worse outcomes from IRIS (unless you had tuberculous meningitis), while ART in cryptococcus meningitis is associated with higher mortality due to IRIS. Not surprisingly, KS is also associated with IRIS. To reiterate, the clinical case definition for IRIS is at least a 1 log10 drop in conjunction with the following (19):

A prospective study (20) evaluated risk factors for KS-IRIS. 300 patients were diagnosed with KS, of which 150 were treated with ART alone. Of these, 10 had rapid clinical progression of KS during the first 2 months of starting ART. These patients had higher CD3 counts at KS diagnosis and were m0ore likely to have KS-associated edema. Further, patients were kept on ART though 5 of these patients required chemotherapy or radiotherapy. In another cohort study (21) of ART-naïve HIV-patients who were diagnosed with KS, 436 patients were evaluated to determine the risk factors of KS-IRIS. Multivariate analysis demonstrated that T1 KS stage (HR 2.96, 95% CI 1.26-6.94), viral load >5 log10 copies/mL (HR 2.14, 95% CI 1.25-3.67), and using ART alone (HR 2.97, 95% CI 1.02-8.7) were associated with KS-IRIS:
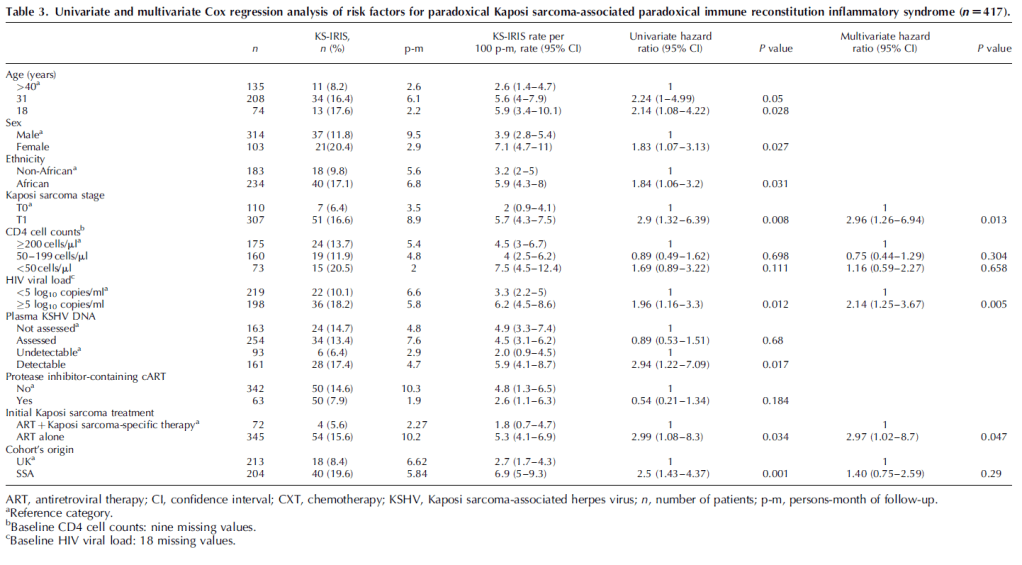
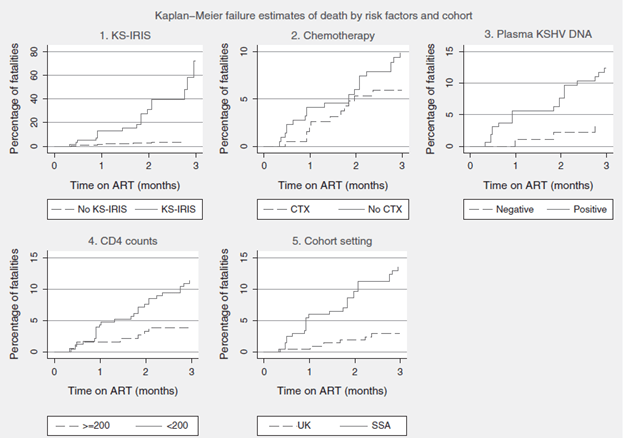
Of this cohort, there were 58 patients with KS-IRIS. The mortality rate in the KS-IRIS cohort was 32.9% (19/58, 95% CI 20.3-45.2) and 11.1% in the non-IRIS group (40/359, 95% CI 7.8-14.4). The cause of death was known 13 of the 19 patients and it was attributed to IRIS in 6/13 (46.1%). Overall, chemotherapy, no KS-IRIS, and lower CD4 counts were associated with decreased mortality. Notably, the UK part of the cohort had significantly lower mortality when compared to the sub-Saharan Africa part of the cohort:
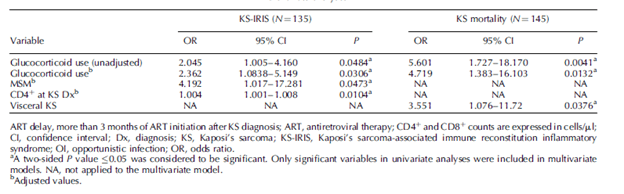
One additional risk factor seems to be the use of steroids. A retrospective study (22) evaluated the outcomes related to KS-IRIS and KS-related mortality. 145 HIV positive patients were evaluated, of which 60 received glucocorticoids. In multivariable analysis, the use of glucocorticoids was associated with a higher incidence of KS-IRIS and KS-related mortality:
Chemotherapy
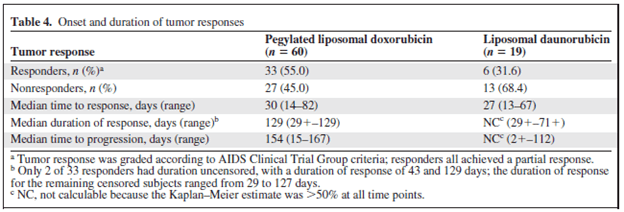
As I mentioned at the beginning of the post, the subject of chemotherapy is one that is best left to oncology. Having said that, there are a few interesting things to note. When it comes to systemic chemotherapy, it seems that either liposomal doxorubicin or liposomal daunorubicin have been employed with success. A prospective, double blind, multicenter RCT (23) evaluated pegylated liposomal doxorubicin with liposomal daunorubicin with primary outcome being the proportion of patients with clinical benefit. Patients were randomized in a 3:1 ratio to pegylated liposomal doxorubicin (n= 60) or liposomal daunorubicin (n =19). 80% of patients in the pegylated liposomal doxorubicin and 63.2% of those in the liposomal daunorubicin achieved a clinical response:

Tumor response was also higher in the pegylated liposomal doxorubicin:
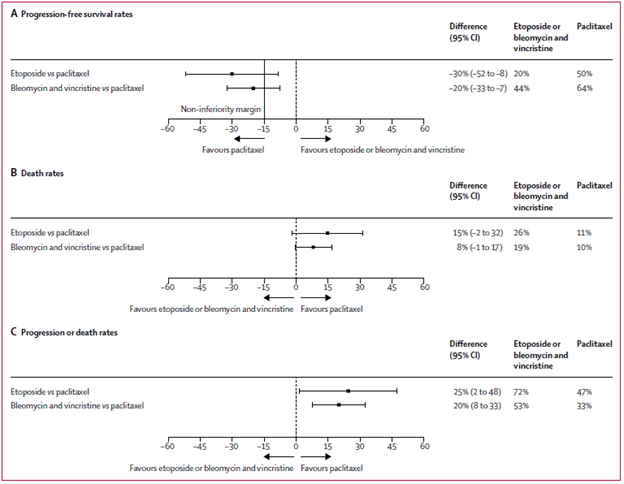
Notably, pegylated liposomal doxorubicin was associated with higher rates of neutropenia, but had similar side effect profile to that of liposomal daunorubicin:
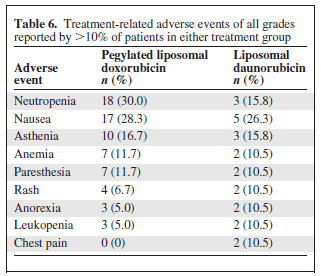
Paclitaxel has also been used with similar efficacy. A RCT evaluated paclitaxel with pegylated liposomal doxorubicin with primary endpoint being progression-free survival (24). 73 patients were randomized in 1:1 fashion (37 to paclitaxel, 37 in the doxorubicin arm; notably, the planned number of patients was 240 but this was not achieved due to slow recruitment). There was no difference in the progression free survival (17.5 months in the paclitaxel arm vs 12.2 months in the pegylated liposomal doxorubicin, p-value 0.66) nor in the overall survival rate (79% in the paclitaxel arm vs 78% in the doxorubicin arm, p-value 0.748):
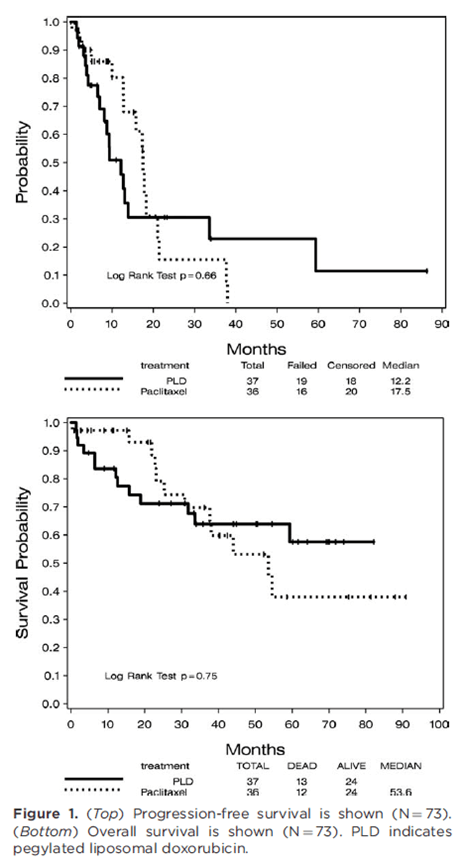
Aside from these agents, bleomycin/vincristine and PO etoposide have been used, albeit with less success. In a non-inferiority multicenter trial (25), PO etoposide and Bleomycin/vincristine were compared to paclitaxel in a low resource setting with primary outcome being progression-free survival at week 48. 334 patients were randomized, but the trial was closed early due to the inferiority of bleomycin/vincristine + ART as well as the PO etoposide + ART arms:
TL;DR:
- Risk factors for the development of AIDS-related KS include MSM, low CD4, high viral load and lack of response in terms of CD4 counts when on ART
- Any regimen can be used for KS (not just PI-based regimen, which interact with everything under the sun)
- Risk factors for KS-IRIS include higher CD4 count at the start of ART, visceral KS, only ART usage, higher viral load (typically >5 log 10), and corticosteroid use
- If KS is at an advanced stage (i.e. any visceral involvement) using ART + systemic chemotherapy (either pegylated liposomal doxorubicin, liposomal daunorubicin, or paclitaxel) can be used. Call your friendly neighborhood oncologist for this one.
References:
- Stebbing J, Portsmouth S, Gazzard B. How does HAART lead to the resolution of Kaposi’s sarcoma? J Antimicrob Chemother. 2003 May;51(5):1095-8. doi: 10.1093/jac/dkg199. Epub 2003 Mar 28. PMID: 12668573.
- Grabar S, Abraham B, Mahamat A, Del Giudice P, Rosenthal E, Costagliola D. Differential impact of combination antiretroviral therapy in preventing Kaposi’s sarcoma with and without visceral involvement. J Clin Oncol. 2006 Jul 20;24(21):3408-14. doi: 10.1200/JCO.2005.05.4072. PMID: 16849755.
- Mocroft A, Kirk O, Clumeck N, Gargalianos-Kakolyris P, Trocha H, Chentsova N, Antunes F, Stellbrink HJ, Phillips AN, Lundgren JD. The changing pattern of Kaposi sarcoma in patients with HIV, 1994-2003: the EuroSIDA Study. Cancer. 2004 Jun 15;100(12):2644-54. doi: 10.1002/cncr.20309. PMID: 15197808.
- Franceschi S, Maso LD, Rickenbach M, Polesel J, Hirschel B, Cavassini M, Bordoni A, Elzi L, Ess S, Jundt G, Mueller N, Clifford GM. Kaposi sarcoma incidence in the Swiss HIV Cohort Study before and after highly active antiretroviral therapy. Br J Cancer. 2008 Sep 2;99(5):800-4. doi: 10.1038/sj.bjc.6604520. Epub 2008 Jul 29. PMID: 18665172; PMCID: PMC2528138.
- Lodi S, Guiguet M, Costagliola D, Fisher M, de Luca A, Porter K; CASCADE Collaboration. Kaposi sarcoma incidence and survival among HIV-infected homosexual men after HIV seroconversion. J Natl Cancer Inst. 2010 Jun 2;102(11):784-92. doi: 10.1093/jnci/djq134. Epub 2010 May 4. PMID: 20442214; PMCID: PMC2879418.
- Vincent T, Moss K, Colaco B, Venables PJ. Kaposi’s sarcoma in two patients following low-dose corticosteroid treatment for rheumatological disease. Rheumatology (Oxford). 2000 Nov;39(11):1294-6. doi: 10.1093/rheumatology/39.11.1294. PMID: 11085817.
- Manion M, Uldrick T, Polizzotto MN, Sheikh V, Roby G, Lurain K, Metzger D, Mican JM, Pau A, Lisco A, Laidlaw E, Hammoud DA, Whitby D, Yarchoan R, Sereti I. Emergence of Kaposi’s Sarcoma Herpesvirus-Associated Complications Following Corticosteroid Use in TB-IRIS. Open Forum Infect Dis. 2018 Oct 12;5(10):ofy217. doi: 10.1093/ofid/ofy217. PMID: 30568973; PMCID: PMC6182308.
- Bower M, Collins S, Cottrill C, Cwynarski K, Montoto S, Nelson M, Nwokolo N, Powles T, Stebbing J, Wales N, Webb A; AIDS Malignancy Subcommittee. British HIV Association guidelines for HIV-associated malignancies 2008. HIV Med. 2008 Jul;9(6):336-88. doi: 10.1111/j.1468-1293.2008.00608.x. PMID: 18705759.
- El Amari EB, Toutous-Trellu L, Gayet-Ageron A, Baumann M, Cathomas G, Steffen I, Erb P, Mueller NJ, Furrer H, Cavassini M, Vernazza P, Hirsch HH, Bernasconi E, Hirschel B; Swiss HIV Cohort Study. Predicting the evolution of Kaposi sarcoma, in the highly active antiretroviral therapy era. AIDS. 2008 May 31;22(9):1019-28. doi: 10.1097/QAD.0b013e3282fc9c03. PMID: 18520345.
- Gbabe OF, Okwundu CI, Dedicoat M, Freeman EE. Treatment of severe or progressive Kaposi’s sarcoma in HIV-infected adults. Cochrane Database Syst Rev. 2014 Aug 13;8(8):CD003256. doi: 10.1002/14651858.CD003256.pub2. PMID: 25221796; PMCID: PMC4174344.
- Grabar S, Abraham B, Mahamat A, Del Giudice P, Rosenthal E, Costagliola D. Differential impact of combination antiretroviral therapy in preventing Kaposi’s sarcoma with and without visceral involvement. J Clin Oncol. 2006 Jul 20;24(21):3408-14. doi: 10.1200/JCO.2005.05.4072. PMID: 16849755.
- Franceschi S, Maso LD, Rickenbach M, Polesel J, Hirschel B, Cavassini M, Bordoni A, Elzi L, Ess S, Jundt G, Mueller N, Clifford GM. Kaposi sarcoma incidence in the Swiss HIV Cohort Study before and after highly active antiretroviral therapy. Br J Cancer. 2008 Sep 2;99(5):800-4. doi: 10.1038/sj.bjc.6604520. Epub 2008 Jul 29. PMID: 18665172; PMCID: PMC2528138.
- Stebbing J, Portsmouth S, Gazzard B. How does HAART lead to the resolution of Kaposi’s sarcoma? J Antimicrob Chemother. 2003 May;51(5):1095-8. doi: 10.1093/jac/dkg199. Epub 2003 Mar 28. PMID: 12668573.
- Grabar S, Abraham B, Mahamat A, Del Giudice P, Rosenthal E, Costagliola D. Differential impact of combination antiretroviral therapy in preventing Kaposi’s sarcoma with and without visceral involvement. J Clin Oncol. 2006 Jul 20;24(21):3408-14. doi: 10.1200/JCO.2005.05.4072. PMID: 16849755.
- Martinez V, Caumes E, Gambotti L, Ittah H, Morini JP, Deleuze J, Gorin I, Katlama C, Bricaire F, Dupin N. Remission from Kaposi’s sarcoma on HAART is associated with suppression of HIV replication and is independent of protease inhibitor therapy. Br J Cancer. 2006 Apr 10;94(7):1000-6. doi: 10.1038/sj.bjc.6603056. PMID: 16570046; PMCID: PMC2361239.
- Mosam A, Shaik F, Uldrick TS, Esterhuizen T, Friedland GH, Scadden DT, Aboobaker J, Coovadia HM. A randomized controlled trial of highly active antiretroviral therapy versus highly active antiretroviral therapy and chemotherapy in therapy-naive patients with HIV-associated Kaposi sarcoma in South Africa. J Acquir Immune Defic Syndr. 2012 Jun 1;60(2):150-7. doi: 10.1097/QAI.0b013e318251aedd. PMID: 22395672; PMCID: PMC3360837.
- Martin-Carbonero L, Barrios A, Saballs P, Sirera G, Santos J, Palacios R, Valencia ME, Alegre M, Podzamczer D, González-Lahoz J; Caelyx/KS Spanish Group. Pegylated liposomal doxorubicin plus highly active antiretroviral therapy versus highly active antiretroviral therapy alone in HIV patients with Kaposi’s sarcoma. AIDS. 2004 Aug 20;18(12):1737-40. doi: 10.1097/01.aids.0000131385.60974.b9. PMID: 15280789.
- Busakhala N, Kigen G, Waako P, Strother RM, Chite F, Loehrer P Sr. Three year survival among patients with aids-related Kaposi sarcoma treated with chemotherapy and combination antiretroviral therapy at Moi teaching and referral hospital, Kenya. Infect Agent Cancer. 2019 Sep 10;14:24. doi: 10.1186/s13027-019-0242-9. PMID: 31516547; PMCID: PMC6734447.
- Achenbach CJ, Harrington RD, Dhanireddy S, Crane HM, Casper C, Kitahata MM. Paradoxical immune reconstitution inflammatory syndrome in HIV-infected patients treated with combination antiretroviral therapy after AIDS-defining opportunistic infection. Clin Infect Dis. 2012 Feb 1;54(3):424-33. doi: 10.1093/cid/cir802. Epub 2011 Nov 17. PMID: 22095568; PMCID: PMC3258272.
- Bower M, Nelson M, Young AM, Thirlwell C, Newsom-Davis T, Mandalia S, Dhillon T, Holmes P, Gazzard BG, Stebbing J. Immune reconstitution inflammatory syndrome associated with Kaposi’s sarcoma. J Clin Oncol. 2005 Aug 1;23(22):5224-8. doi: 10.1200/JCO.2005.14.597. PMID: 16051964.
- Letang E, Lewis JJ, Bower M, Mosam A, Borok M, Campbell TB, Naniche D, Newsom-Davis T, Shaik F, Fiorillo S, Miro JM, Schellenberg D, Easterbrook PJ. Immune reconstitution inflammatory syndrome associated with Kaposi sarcoma: higher incidence and mortality in Africa than in the UK. AIDS. 2013 Jun 19;27(10):1603-13. doi: 10.1097/QAD.0b013e328360a5a1. PMID: 23462220.
- Fernández-Sánchez M, Iglesias MC, Ablanedo-Terrazas Y, Ormsby CE, Alvarado-de la Barrera C, Reyes-Terán G. Steroids are a risk factor for Kaposi’s sarcoma-immune reconstitution inflammatory syndrome and mortality in HIV infection. AIDS. 2016 Mar 27;30(6):909-14. doi: 10.1097/QAD.0000000000000993. PMID: 26636923; PMCID: PMC4794188.
- Cooley T, Henry D, Tonda M, Sun S, O’Connell M, Rackoff W. A randomized, double-blind study of pegylated liposomal doxorubicin for the treatment of AIDS-related Kaposi’s sarcoma. Oncologist. 2007 Jan;12(1):114-23. doi: 10.1634/theoncologist.12-1-114. PMID: 17227906.
- Cianfrocca M, Lee S, Von Roenn J, Tulpule A, Dezube BJ, Aboulafia DM, Ambinder RF, Lee JY, Krown SE, Sparano JA. Randomized trial of paclitaxel versus pegylated liposomal doxorubicin for advanced human immunodeficiency virus-associated Kaposi sarcoma: evidence of symptom palliation from chemotherapy. Cancer. 2010 Aug 15;116(16):3969-77. doi: 10.1002/cncr.25362. PMID: 20564162; PMCID: PMC3157242.
- Krown SE, Moser CB, MacPhail P, Matining RM, Godfrey C, Caruso SR, Hosseinipour MC, Samaneka W, Nyirenda M, Busakhala NW, Okuku FM, Kosgei J, Hoagland B, Mwelase N, Oliver VO, Burger H, Mngqibisa R, Nokta M, Campbell TB, Borok MZ; A5263/AMC066 protocol team. Treatment of advanced AIDS-associated Kaposi sarcoma in resource-limited settings: a three-arm, open-label, randomised, non-inferiority trial. Lancet. 2020 Apr 11;395(10231):1195-1207. doi: 10.1016/S0140-6736(19)33222-2. Epub 2020 Mar 5. PMID: 32145827; PMCID: PMC7236082.c

Hi, what about use of antiviral (aciclovir, ganciclovir, cidofovir) on outcomes ?
Is there a minimal threshold of DNA hhv6 in the blood to develop kaposi?
LikeLike
good questions. from my experience, antivirals are not typically used in practice (though I could be wrong; a cursory pubmed search reveals these have been used but with mixed results) as KS tends to be quite end-stage. honestly, i’ve seen it to the point that you need chemo to do anything and even then, outcomes are not great. i think this would make a great topic for a post.
LikeLike
also, i do not know a thresold HHV6 DNA; i never thought of that question to be honest.
LikeLike