HIV-associated Immune Reconstitution Inflammatory Syndrome (IRIS) is a known early complication of initiating ART therapy in those who are profoundly immunosuppressed. Here, immune recovery following ART initiation leads to an inflammatory response against an organism leading to a clinical deterioration a few weeks following ART initiation. You can think of it as follows; you have an opportunistic infection but due to your immunosuppression, it hangs out without repercussion. Once you get your CD4 cells up, the immune system “wakes” up and begins to attack this organism. This, in turn, leads to an inflammatory response as the immune system kicks into gear. While theoretically, IRIS can occur with any organism, most of the data comes from TB-associated IRIS (TB-IRIS) and cryptococcal IRIS (C-IRIS). When discussing any IRIS, there are 2 patterns to keep in mind.
- Paradoxical – you had an OI already diagnosed, for instance, tuberculosis, that was being treated adequately with good response. You get ART and a few weeks in, you get symptoms of TB. This is despite having responded to the initial therapy (1)
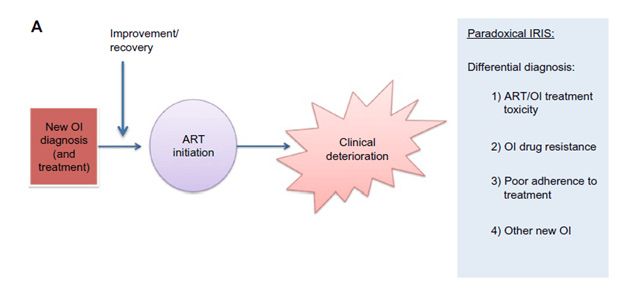
- Unmasked/ART-associated – you had no diagnosis of an OI/picked up a new OI that was not known prior to starting ART. Then you get symptoms of said OI. This is difficult to discern, since a lot of time, AIDS patients have lower sensitivities to screening tests for things such as TB/crypto (due to the nature of immunosuppression. How can you pick up antibodies OR IFN levels when you have an immune system that sucks at making both?).

The pathophysiology involves a combination of severe immunodeficiency that causes a high organism burden, which in turn, is followed by a pathological and dysregulated “immune awakening” following ART initiation that leads to the symptoms of IRIS:
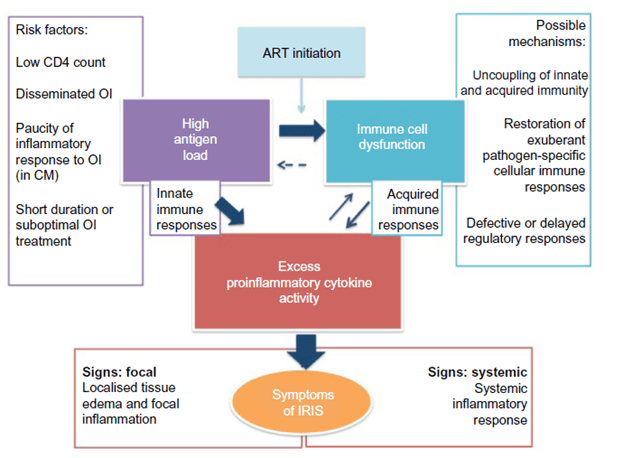
Risk Factors:
In general, lower CD4 counts, higher viral load, Integrase inhibitors, as well as certain pathogens (TB, cryptococcus) tend to be significant risk factors for IRIS. In a retrospective cohort (2) from Washington State, 196 patients who had started ART and had OI were evaluated for risk factors for the development of IRIS. 21 patients were deemed to have IRIS (11% of the entire cohort), with 2/3s occurring within the first 3 months:

Notable risk factors for IRIS were KS and multiple/widespread OIs, though not all OIs are created equal. In this cohort, Candida esophagitis and PCP were protective of IRIS:

Further, those patients with mucocutaneous KS IRIS and TB IRIS had significant increase in CD4, a pattern that was not seen in other OIs:

Notably, cryptococcus also had a significant rise in CD4 for those with IRIS compared to those who did not get IRIS, though it did not reach statistical significance. I chuck that to the low numbers of patients here, but the pattern seems to fit that of TB and KS. Speaking of KS, I am surprised that it is the mucocutaneous version rather than the visceral version of KS that is associated with IRIS. Go figure.
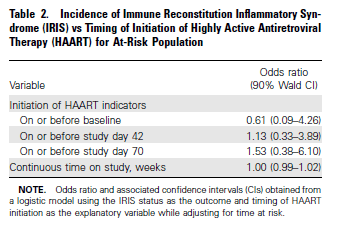
Another study (3) evaluated RF for IRIS in an RCT done to evaluate early vs deferred ART in setting of an OI. 262 patients were started on ART and were included in the analysis. Patients were randomized to receive ART at randomization (usually within 14d of OI diagnosis) or delayed at 4 weeks. 20 patients (7.6%) were diagnosed with IRIS, with a median time to diagnosis of 33 days, which did not differ between groups. Notably, while most cases were without complications, 4 of these were protracted and all were related to cryptococcal meningitis. There were no deaths attributed to IRIS in this cohort, though.
Overall, early or delayed ART did not impact the development of IRIS. In fact, the only factor associated with increased incidence of IRIS was non-PCP fungal infections (they did not include TB patients in this analysis).

Comparing those who had IRIS within 4 weeks or after 4 weeks, one noticeable trend is the significant rise in CD4 by week 4 as well as drop in viral load. This was not seen for CD8.
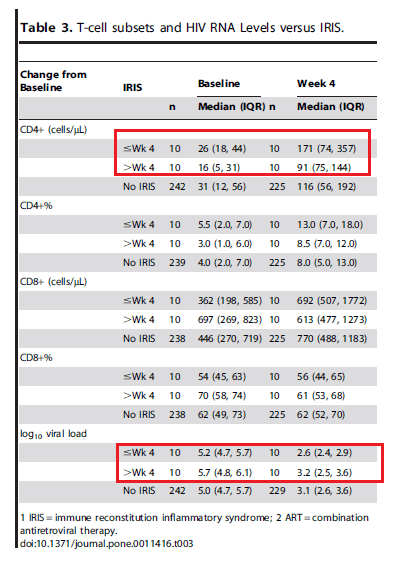
Further, lower CD4 count at baseline, higher CD4 on ART, and presence of non-PCP fungal infections were all associated with higher risk of IRIS:
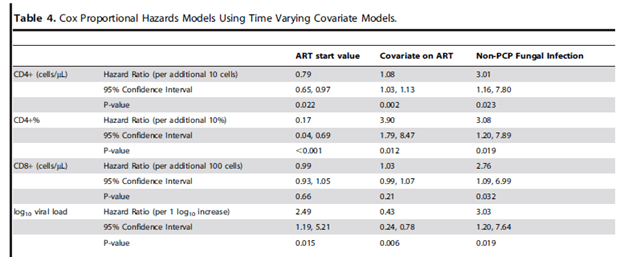
Overall, seems that non-PCP fungal infections are highly associated with the development of IRIS. Beyond that, it is difficult to predict who is going to get it.
In terms of medications, it seems that integrase inhibitor based regimens are associated with a higher risk of inducing IRIS. In a retrospective data analysis from the ATHENA trial (4) the incidence of IRIS in integrase containing regiments (INT) vs non-integrase regimens were compared in late presenting HIV Patients. 672 patients were included, 155 started no INT containing regimens, while the rest (517) were started on non-integrase based regimens. IRIS was found in 32.3% (50/155) in the INT group vs 18% (93/517) of the non-INT group (OR 2.17 95% CI 1.45-3.25, p-value 0.0003), with most being paradoxical IRIS (63.6% vs 25.2%). Most of the IRIS presentations were in those who had underlying TB, cryptococcal meningitis, and KS:
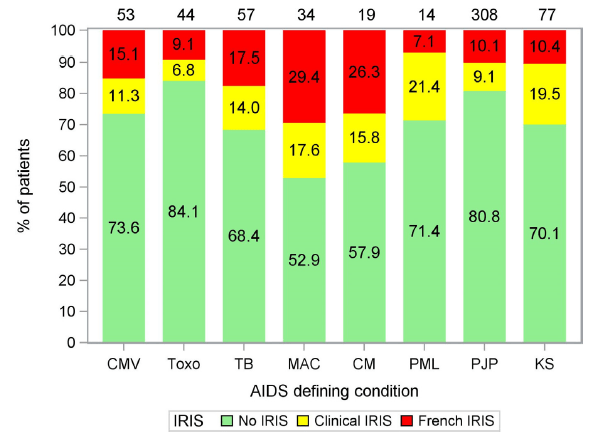
Multivariable logistic regression analysis demonstrated that the use of integrase inhibitors, diagnosis of cryptococcal meningitis, TB, MAC, and KS were associated with a higher likelihood of IRIS:

Of note, when evaluating each integrase inhibitor independent of one another, raltegravir was associated with a higher likelihood of IRIS when compared to elvitegravir and dolutegravir, though the incidence of DTG associated IRIS increased after 2015, when the use of RAL was phased out:
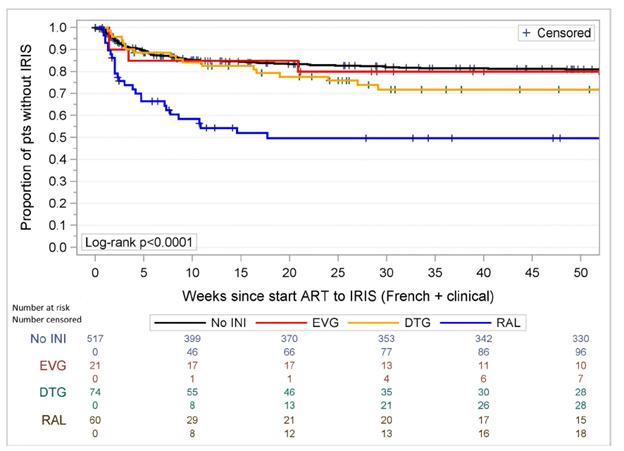
While the use of DTG may have not been associated with higher incidence of IRIS, at least statistically, there is some noise about its association. Overall, there was no increased in mortality rates in this cohort.
Summary of risk factors, adapted from Ref 1:

Tuberculosis:
A review (5) attempted to create case definitions to diagnose TB-IRIS. As most of the cases come from low-income areas, these definitions remove the CD4 or viral load requirement from the equation, since CD4 takes a while to increase and, assuming adherence, viral load drops quite rapidly after ART initiation.


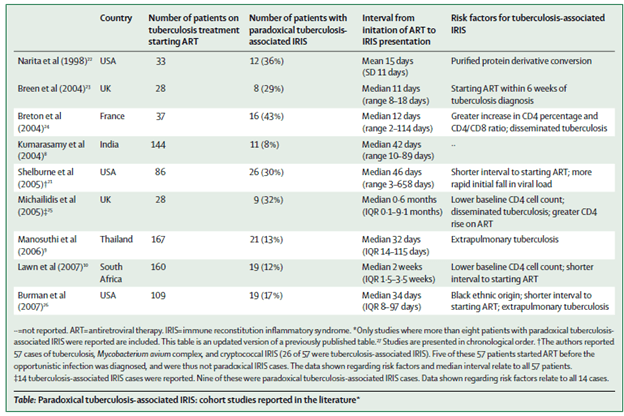
The overall prevalence of TB-related IRIS ranged form 8% to 43%:
Risk factors for TB-IRIS include early use of ART (within 2 weeks), extrapulmonary tuberculosis, and severe immunosuppression. Data from the CAMELIA trial (6), which was done to find out the optimal timing of ART initiation, found that IRIS was more likely in those who had ART started within 2 weeks of diagnosis of TB vs 8 weeks:
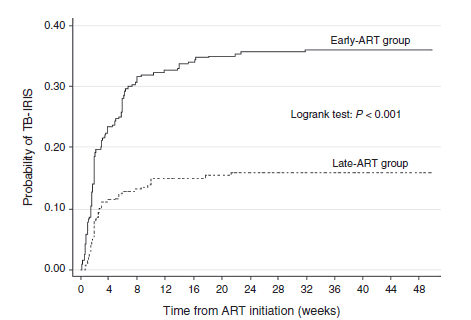
Most of the cases occurred within 3 months with a median time in early and late ART groups being 14 days. Most of the symptoms in both groups included lymphadenopathy and fever, and CXR changes were common well:
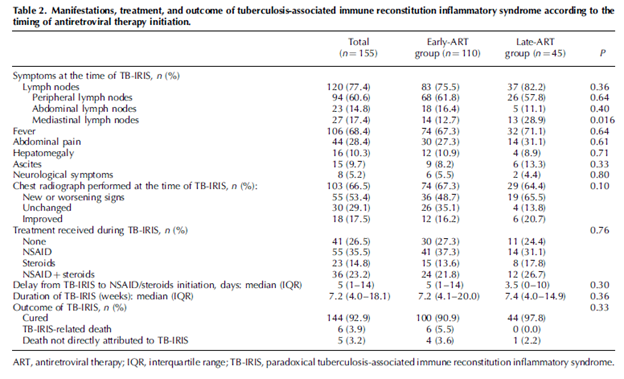
Risk factors associated with TB-IRIS included early initiation of ART, CD4 between 25-50, VL > 6 log10 copies/mL, hemoglobin <10, and extrapulmonary/disseminated TB:
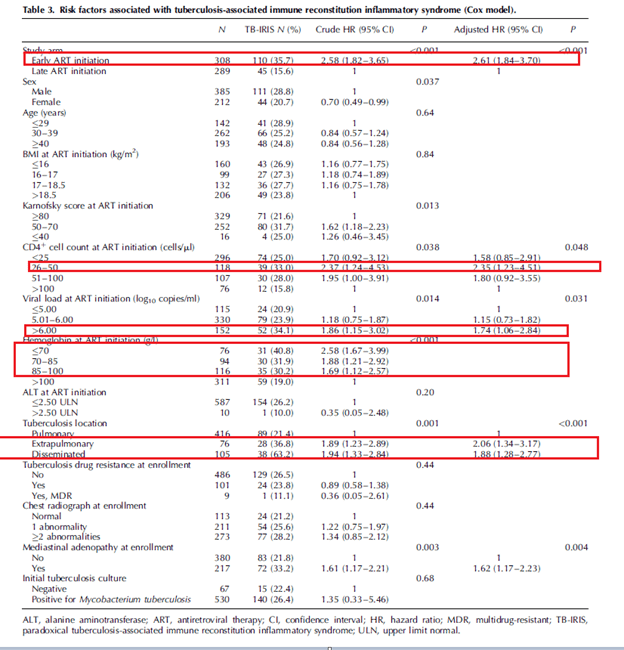
In a Cox proportional hazard model, there was no association between occurrence of TB-IRIS and mortality (HR 0.97 95% CI 0.6-1.57, p-value 0.91).
In another retrospective study, (7), found that predictors of TB-IRIS included Hbg <10, CD45 <5, extrapulmonary tuberculosis, and negative tuberculin skin test. While there was a trend towards early initiation of ART as a risk factor for TB-IRIS, this did not reach statistical significance:

Similarly, median time for symptoms was 14 days, with the highest proportion of patients experiencing symptoms within 60-90 days:
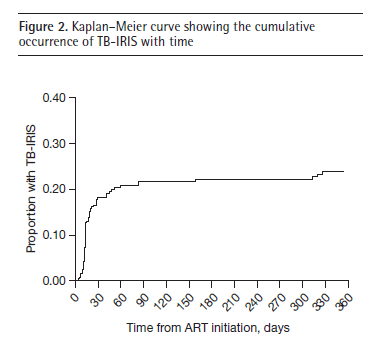
Clinical manifestations included worsening lymphadenopathy, fever, cough, and weight loss:
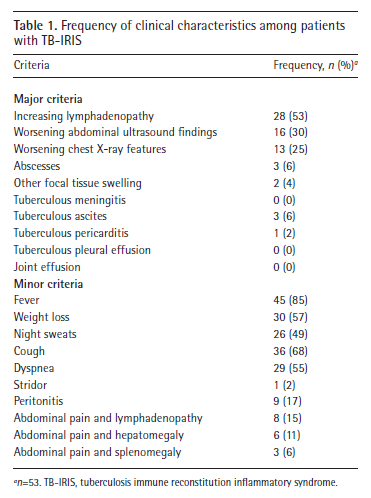
In another retrospective analysis of ART initiation of India (8), 627 patients were evaluated of which 237 were diagnosed with TB. Of those 237, 18 (7.5%) developed TB-IRIS while 12 of 390 patients (3%) developed ART-associated TB after ART initiation. Most of these occurred in the first 30 days, with the most common manifestations being new-onset fever or worsening cervical lymphadenopathy.
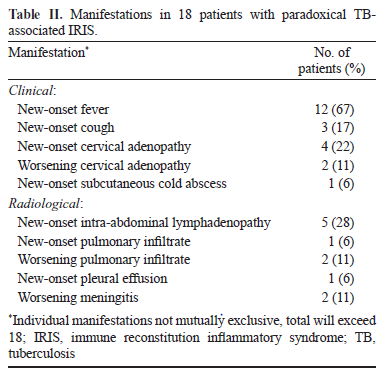
Risk factors in this cohort included low CD4 count as well as significant rise in CD4 after 6 months:
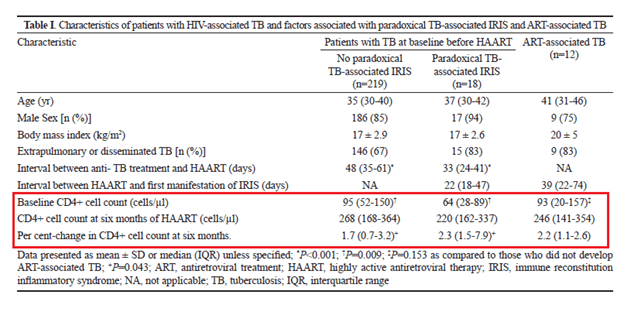
Despite this, mortality seems to not be correlated with the development of IRIS. For instance, Worodia et al (7) reported that 90% of all patients recovered, suggesting no impact of IRIS on mortality. The original data from the CAMILIA trial (9) suggested that early ART was associated with improved outcomes and Laureillard et al (6) found in a cox proportional hazard model there was no association between TB-IRIS and mortality (crude HR 0.97, 95% CI 0.6-1.57, p-value 0.91).
In another cohort study (10), 301 patients with TB-HIV coinfection were evaluated. At follow up of 2 years, 217 were alive while 53 had died. Mortality rate was 16 per 1000 person years overall, with rates being higher in the first 3 months (33.9 per 100 person years, 95% CI 22.7 to 50.5) and decreasing to 5.6 per 100 person years after 9 months (95% CI 2.8 to 11.1). 23 deaths (43%) occurred within 3 months and 36 (68%) occurred within 6 months. TB-IRIS developed in 54 (20.5%) of patients, of which 6 had died. Of these 6, five had a contributing cause of death unrelated to IRIS.
The factors associated with mortality included male sex, in-patient status, positive serum CrAg, negative tuberculin test, and no ART use. Notably, development of TB-IRIS was not associated with mortality:


So overall, seems that risk factors for TB-IRIS is early ART but this is not associated with mortality. This is not the same case for cryptococcus.
Cryptococcus:
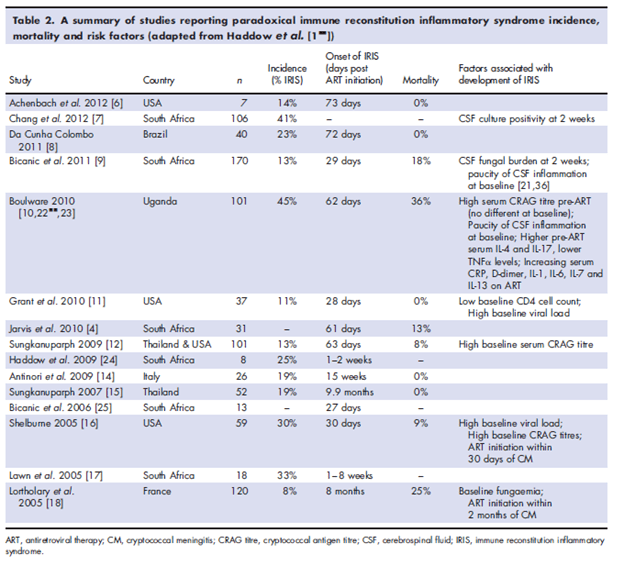
The incidence of C-IRIS ranged from 8% to 41%, with mortality ranging from 0% to 36% (11).
Similar to TB-IRIS, case definitions have been proposed for C-IRIS:
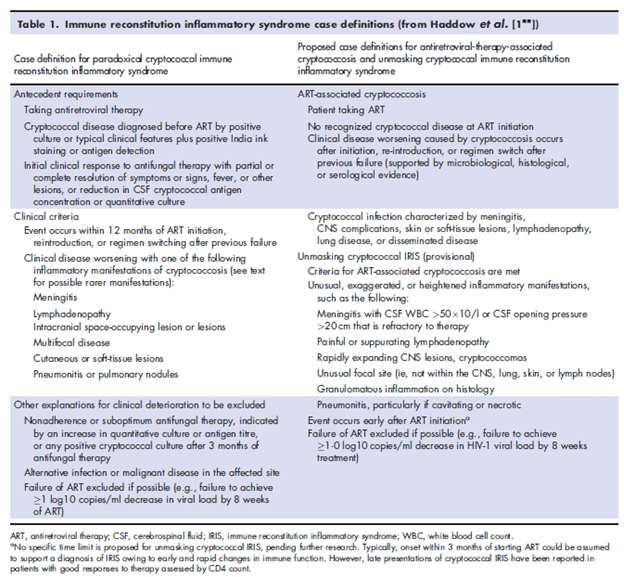
The early initiation of ART in setting of cryptococcal meningitis is associated with increased mortality. For instance, in a prospective, open labeled, RCT (12), early ART (within 72hrs of diagnosis of cryptococcal meningitis) was compared with delayed ART (10 weeks after cryptococcal meningitis). Notably, patients received fluconazole throughout this study. 93 patients were enrolled, 28 randomized to the early treatment arm and 26 to the delayed treatment arm. 3 year mortality rate was 88% in the delayed ART arm vs 54% in the early ART arm:
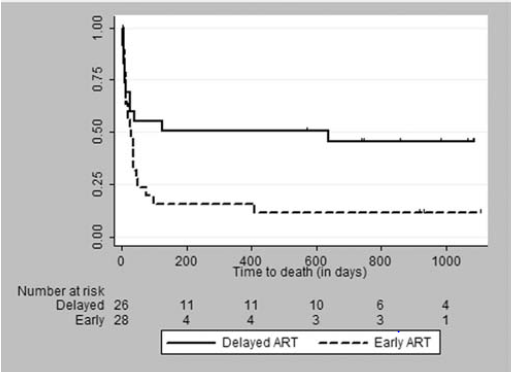
Most of the deaths occurred early on, with 10 deaths in the early ART arm and 7 in the delayed ART arm occurring within 2 weeks. The only predictor of mortality was early ART in this cohort. In another randomized trial (13), early ART (within 1-2 weeks of cryptococcal meningitis diagnosis) was compared to delayed ART (at week 5 of diagnosis) with primary end point being 26 week mortality. 177 patients were randomized, and at 26 weeks, more patients had died in the early ART group (40 of 88, 45%) compared to delayed ART (27 of 89, 30%), HR 1.73, 95% CI 1.06 to 2.82, p-value 0.03:
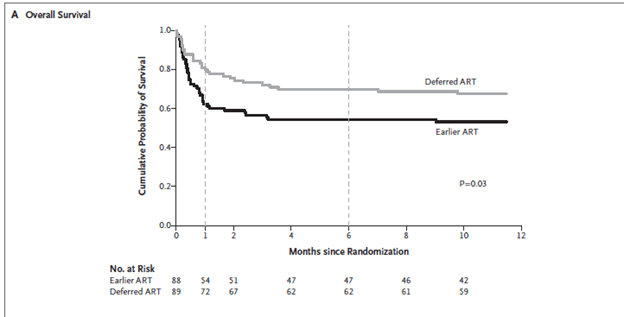
Notably, this occurred early on, within days 8 through 30, of therapy. Mortality rates did not differ beyond this time frame. Subgroup analysis revealed that mortality differed between groups in those whose CSF WBC was <5, while mortality was not different in those whose CSF WBC was >5:
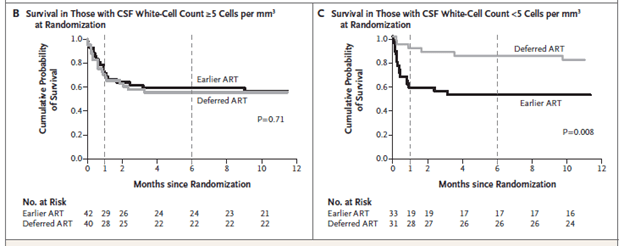
Other factors associated with increased mortality included higher cryptococcal fungal loads and GCS <15:
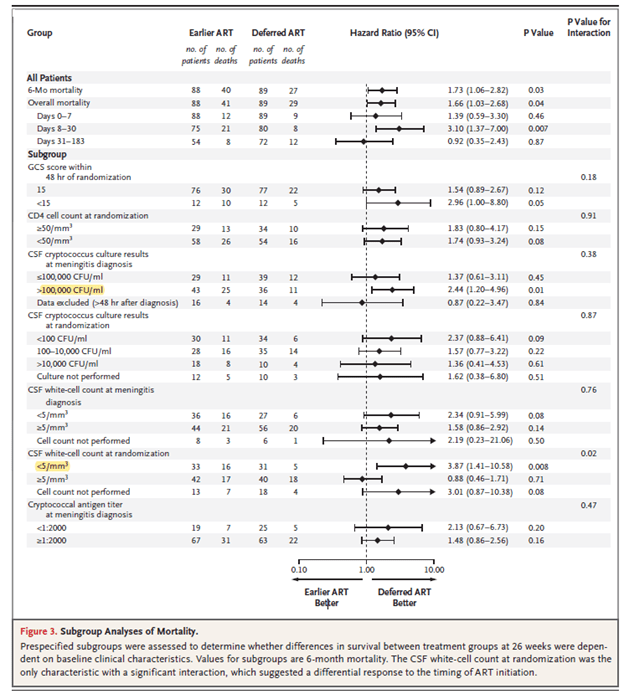
Notably, there was a trend towards higher rate of cryptococcal IRIS in those who had early ART, but this did not reach statistical significance.
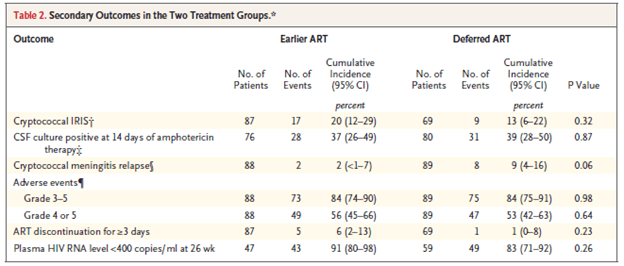
The CSF in the early therapy group had a higher proportion of patients with WBC >5 when compared to delayed ART (58% vs 40%, p-value 0.047).
In a Cochrane review (14) of early vs late ART in context of cryptococcal meningitis, all-cause mortality favored delayed ART (by at least a month):
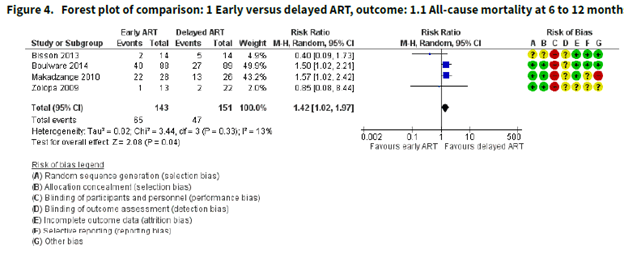
Notice the bias. However data did not support the notion that early ART impacted the incidence of IRIS, though the authors note “the certainty of the evidence for this outcome was very low due to marked imprecision of the estimate, risk of bias, and indirectness related to the IRIS assessment.” Go figure.

Early ART is associated with increased mortality for cryptococcus, but seems that early ART is not necessarily associated with IRIS. Smaller studies seems to suggest that IRIS is not associated with increased mortality. For instance, a small retrospective study (15) of 101 patients identified 13 cases of IRIS. Most common symptoms included headache and focal neurological deficits. Overall, there was no statistical difference in the incidence of IRIS that was dependent on the time to initiation of ART, however the numbers are small enough that it is difficult to draw a definitive conclusion:
Having said that, those with IRIS had higher opening pressure, higher percentage of neutrophils and lymphocytes as well as lower CrAg titers at the time of the first culture negative visit:
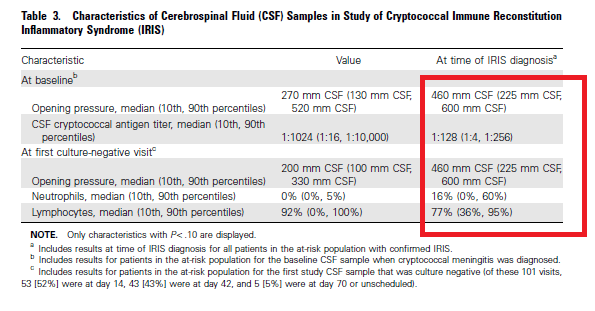
Interestingly, mortality rate in IRIS was not statistically different when compared to the rest of the cohort, 7.7% in the IRIS group vs 14.8% in the non-IRIS group (p-value 0.69). Only initial serum CrAg and BMI were associated with increased risk of CM-IRIS:

In a prospective study (16), patients with cryptococcal meningitis were started on ART within 4 weeks following diagnosis of meningitis. 65 patients were included and 11 of these (17%) developed IRIS with a median of 29 days after starting ART. 3 of the 11 patients died, though the authors note there is difficulty in ascertain if IRIS played a role and note that the remaining but one had complete recovery. Overall, there was no difference in mortality (statistically) between the non-IRIS group and the IRIS group:
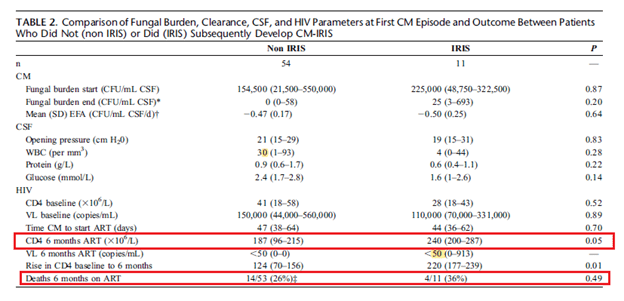
Further, beyond higher PMNs in the CSF and lower crypto Ag, this study did not find any differences in the CSF in those who developed IRIS from the first LP to the LP following IRIS diagnosis:
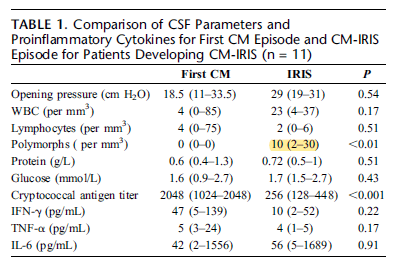
This contrasts with another prospective study (17) of 122 ART patients with a recent diagnosis of cryptococcal meningitis. There were 57 episodes of IRIS in 45 patients with mortality being higher in those with IRIS than the rest of the cohort (HR 2.3, 95% CI 1.1-5.1, p-value 0.04).
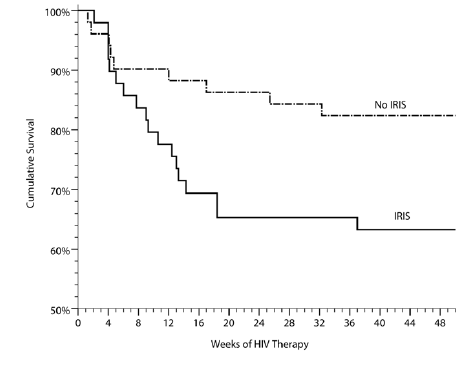
Those with IRIS were likely to have a significant elevation in CD4 cells by week 48 as well as higher serum CrAg titer prior to starting ART:
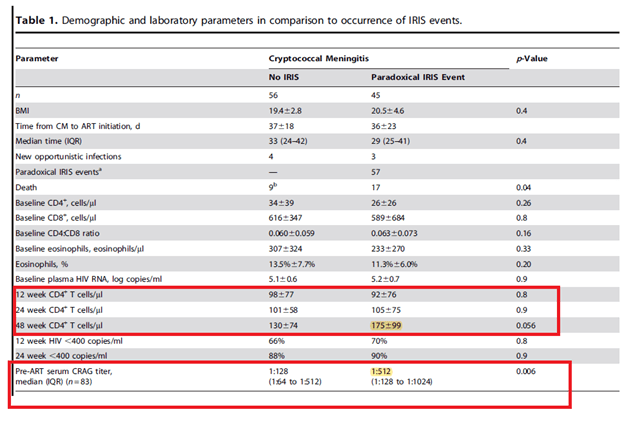
Multiple biomarkers were evaluated to see if these could predict mortality in this patient population. While most are not terribly helpful in the clinical setting (yeah, when was the last time you ordered an IL-6 before COVID, or IL-7, IL 13 or GSCF? Exactly), CRP seems to be fairly helpful in this scenario. For instance, those with CRP >32 had a higher incidence of IRIS (OR 3.9, 95% CI 1.3-11.3 p-value 0.01) and shorter time to event (HR 3.4, 95% CI 1.8-6.6, p-value <0.001) and had a higher mortality rate than those with CPR <32:
In another retrospective study (18) from Texas, 84 HIV patients with cryptococcal meningitis were evaluated. 18 patients (21.4%), were diagnosed with IRIS with most of them (11 out of 18) developing it within 30 days after starting ART. The risk factors evaluated in this cohort were higher HIV viral load (5.54 vs 5.24 log 10 copies/ml, p-value 0.013), more likely to be ART naïve (100% vs 61%, p-value 0.004), and had higher CSF CrAg titer (1:4096 vs 1:1023, p-value 0.02). Further, those who started ART within 30d of meningitis diagnosis were more likely to develop IRIS (RR 1.73, 95% CI 1.03-2.29). In another study of 116 patients (19), 27 patients (25.5% of the cohort were classified as IRIS). Those who had IRIS tended to have lower CD4 levels prior to ART, as well as lower CSF protein levels, lower CSF white count, and higher cryptococcal burden when compared to the non-IRIS cohort.
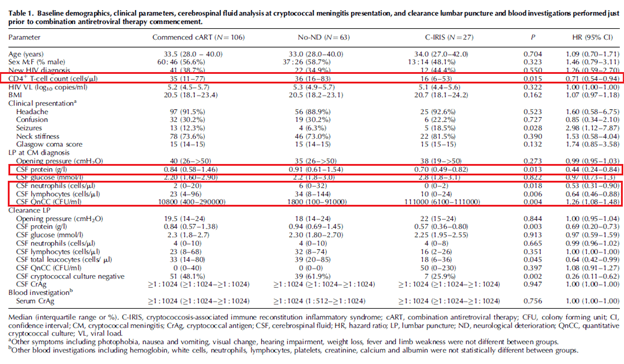
Further, a higher CD4 T-cell count over 24 weeks (HR 0.99, p-value 0.026), and negative cryptococcal culture (HR 0.27, p-value 0.027) were associated with lower incidence of IRIS. Notably, clearance of cryptococcal CSF cultures was associated with decreased incidence of neurological deterioration, IRIS, deaths, and relapse.
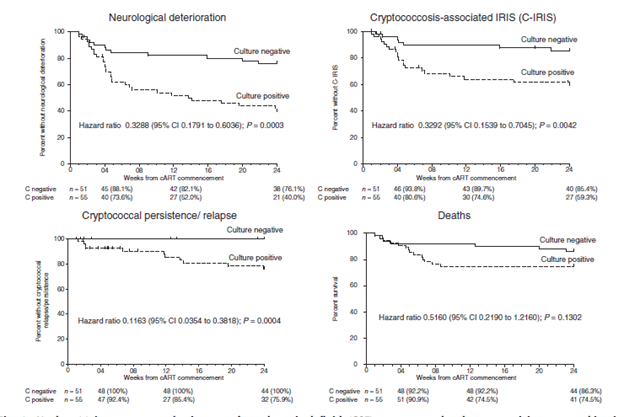
This would suggest that higher cryptococcal burden in setting of severe immunosuppression (i.e. low CD4) followed by significant rise in CD4 and subsequent ongoing presence of cryptococcus are risk factors for developing IRIS. Which is not surprising, given the proposed pathophysiology of IRIS.
Overall, while early ART is strongly associated with TB-IRIS, the same cannot be said for C-IRIS (at least, with the same degree of confidence). Data does suggest that staring ART in cryptococcal meningitis is associated with increased mortality, though not necessarily with increased incidence of IRIS, though some of the studies that did not find this association had either low numbers or were retrospective cohorts. It may be helpful to check cryptococcal antigen and T-spot/other IGRA prior to starting ART. If there is any concern for cryptococcal meningitis, delaying ART may be reasonable, though you should not delay ART for TB. This may not apply for tuberculous meningitis, however. In an RCT (19) comparing early (within 7d) vs delayed (after 2 month) ART after TB-meningitis therapy did not find any difference in mortality (HR 1.12; 95% confidence interval [CI], .81–1.55; P = .50) though the early ART had more grade 3 and 4 adverse events, suggesting that in TB meningitis, delaying ART is probably best.
Take Homes
- Any infection can lead to IRIS in HIV patients
- Any fungal infections (except PCP, candida), TB, KS, and Cryptococcus are pathogens that tend to confer risk for IRIS
- Low CD4 count, high viral load (>5 log 5 usually), integrase-inhibitor based therapy are other patient specific risk factors for IRIS. Significant decrease in VL, as well as rise in CD4 count are also risk factors.
- In terms of TB, while early ART (within 2 weeks) confers risk for IRIS, the mortality from TB-IRIS is not significant enough to warrant delay in therapy. In fact, in TB it is better to get patients started on ART early
- The same is not true for cryptococcus. While the data is not particularly compelling here, early ART may be associated with IRIS (again, data goes back and forth), but it is associated with increased mortality.
References:
- Walker, Naomi F et al. “Immune reconstitution inflammatory syndrome in HIV-infected patients.” HIV/AIDS (Auckland, N.Z.) vol. 7 49-64. 12 Feb. 2015, doi:10.2147/HIV.S42328
- Achenbach CJ, Harrington RD, Dhanireddy S, Crane HM, Casper C, Kitahata MM. Paradoxical immune reconstitution inflammatory syndrome in HIV-infected patients treated with combination antiretroviral therapy after AIDS-defining opportunistic infection. Clin Infect Dis. 2012;54(3):424-433. doi:10.1093/cid/cir802
- Grant PM, Komarow L, Andersen J, et al. Risk factor analyses for immune reconstitution inflammatory syndrome in a randomized study of early vs. deferred ART during an opportunistic infection. PLoS One. 2010;5(7):e11416. Published 2010 Jul 1. doi:10.1371/journal.pone.0011416c
- Wijting IEA, Wit FWNM, Rokx C, et al. Immune reconstitution inflammatory syndrome in HIV infected late presenters starting integrase inhibitor containing antiretroviral therapy. EClinicalMedicine. 2019;17:100210. Published 2019 Dec 13. doi:10.1016/j.eclinm.2019.11.003
- Meintjes G, Lawn SD, Scano F, et al. Tuberculosis-associated immune reconstitution inflammatory syndrome: case definitions for use in resource-limited settings. Lancet Infect Dis. 2008;8(8):516-523. doi:10.1016/S1473-3099(08)70184-1
- Laureillard D, Marcy O, Madec Y, et al. Paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome after early initiation of antiretroviral therapy in a randomized clinical trial. AIDS. 2013;27(16):2577-2586. doi:10.1097/01.aids.0000432456.14099.c7
- Worodria W, Menten J, Massinga-Loembe M, et al. Clinical spectrum, risk factors and outcome of immune reconstitution inflammatory syndrome in patients with tuberculosis-HIV coinfection. Antivir Ther. 2012;17(5):841-848. doi:10.3851/IMP2108
- Sharma SK, Dhooria S, Barwad P, et al. A study of TB-associated immune reconstitution inflammatory syndrome using the consensus case-definition. Indian J Med Res. 2010;131:804-808.
- Blanc FX, Sok T, Laureillard D, et al. Earlier versus later start of antiretroviral therapy in HIV-infected adults with tuberculosis. N Engl J Med. 2011;365(16):1471-1481. doi:10.1056/NEJMoa1013911
- Worodria W, Massinga-Loembe M, Mazakpwe D, et al. Incidence and predictors of mortality and the effect of tuberculosis immune reconstitution inflammatory syndrome in a cohort of TB/HIV patients commencing antiretroviral therapy. J Acquir Immune Defic Syndr. 2011;58(1):32-37. doi:10.1097/QAI.0b013e3182255dc2
- Longley N, Harrison TS, Jarvis JN. Cryptococcal immune reconstitution inflammatory syndrome. Curr Opin Infect Dis. 2013;26(1):26-34. doi:10.1097/QCO.0b013e32835c21d1
- Makadzange AT, Ndhlovu CE, Takarinda K, et al. Early versus delayed initiation of antiretroviral therapy for concurrent HIV infection and cryptococcal meningitis in sub-saharan Africa. Clin Infect Dis. 2010;50(11):1532-1538. doi:10.1086/652652
- Boulware DR, Meya DB, Muzoora C, et al. Timing of antiretroviral therapy after diagnosis of cryptococcal meningitis. N Engl J Med. 2014;370(26):2487-2498. doi:10.1056/NEJMoa1312884
- Eshun-Wilson I, Okwen MP, Richardson M, Bicanic T. Early versus delayed antiretroviral treatment in HIV-positive people with cryptococcal meningitis. Cochrane Database Syst Rev. 2018;7(7):CD009012. Published 2018 Jul 24. doi:10.1002/14651858.CD009012.pub3
- Sungkanuparph S, Filler SG, Chetchotisakd P, et al. Cryptococcal immune reconstitution inflammatory syndrome after antiretroviral therapy in AIDS patients with cryptococcal meningitis: a prospective multicenter study. Clin Infect Dis. 2009;49(6):931-934. doi:10.1086/605497
- Bicanic T, Meintjes G, Rebe K, et al. Immune reconstitution inflammatory syndrome in HIV-associated cryptococcal meningitis: a prospective study. J Acquir Immune Defic Syndr. 2009;51(2):130-134. doi:10.1097/QAI.0b013e3181a56f2e
- Boulware DR, Meya DB, Bergemann TL, et al. Clinical features and serum biomarkers in HIV immune reconstitution inflammatory syndrome after cryptococcal meningitis: a prospective cohort study. PLoS Med. 2010;7(12):e1000384. Published 2010 Dec 21. doi:10.1371/journal.pmed.1000384
- Shelburne SA 3rd, Darcourt J, White AC Jr, et al. The role of immune reconstitution inflammatory syndrome in AIDS-related Cryptococcus neoformans disease in the era of highly active antiretroviral therapy. Clin Infect Dis. 2005;40(7):1049-1052. doi:10.1086/428618
- Török, M Estee et al. “Timing of initiation of antiretroviral therapy in human immunodeficiency virus (HIV)–associated tuberculous meningitis.” Clinical infectious diseases : an official publication of the Infectious Diseases Society of America vol. 52,11 (2011): 1374-83. doi:10.1093/cid/cir230