This is a complicated issue, but I think it makes antibiotic dosing (especially things like vancomycin and aminoglycoside) more easy to understand.
I’ll try to make it as easy and digestible as possible. I’ll discuss mostly pharmacodynamics (i.e how the drug exerts its antimicrobial effect).
I discussed MIC testing in a previous post, as well as the concept between bactericidal and bacteriostatic:
There are a few words to describe as they relate to both pharmacodynamics and microbiology to make sense of dosing:
- Maximal concentration – aka the “peak”
- Minimal concentration – aka “trough”
- Area under the curve – this is a bit more difficult. It relates to the total concentration over a specific period of time. Let’s illustrate this the only way I know how:
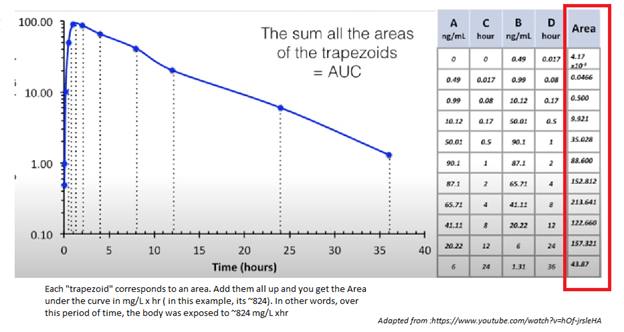
In other words, it tells us, at any point in time, how much drug is in the plasma. More importantly, it tells us how much drug is in the plasma OVER a given time (either 30min, 12hr, 24hr). Because it also takes into account the elimination and distribution, it makes it easier to translate what is going on in the serum to what is going on in the tissues. Because of this, AUC is usually represented as (mg x hr)/L
- MIC already defined
There are 3 PK-PD indices based on serum or plasma concentration that are associated with antimicrobial efficiency; perhaps you have seen this image:
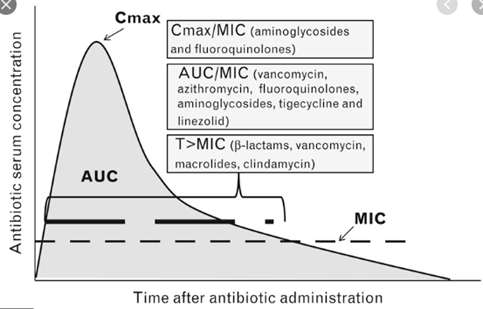
It essentially tells us how each antibiotic is able to efficiently kill off any organism, and how this plays a role into dosing. While this image is difficult to comprehend to a certain extent, it may be easier to define antibiotic by how they efficiently kill
- Concentration dependent – the “higher you go, the easier it is.” In other words, if you go beyond the concentration you need to inhibit growth, you get more efficient killing. So you can, theoretically, get just as efficient killing if you reach a super high concentration for a few hours before you go below the MIC.
- A more verbose way to say it: when the ratio of concentration at a specific site of infection is well above the MIC, greater killing occurs.
- Time dependent – “slow and steady” in other words, you want to keep the amount of antibiotic above the MIC for as long as you can.
This explains why some antibiotics are dosed the way they are (i.e. we usually dose aminoglycosides once or twice, but beta-lactams tend to be dosed every 4 hours or have continuous dosing such as nafcillin. You may also hear about extended release antibiotics with certain beta lactams in terms of certain bugs such as Pseudomonas).
Now, I lied when I said “peak” because in reality, by the time we start to measure the peak serum concentration, some of the drug may have been metabolized and/or eliminated. So the number may be lower. Plus, this only reflects the amount of drug in the plasma and not necessarily how much it gets into the bone/clot/abscess.
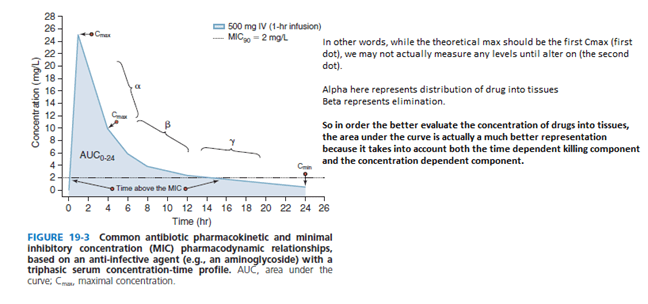
So you can see that, while there is a distinction between “concentration” dependent killing and “time” dependent killing, both of those variables are correlated with the area under the curve.
- If you shoot for a higher concentration, you can see that the AUC increases
- If you keep the concentration above MIC for a lot longer, the AUC increases
Which is why, on the first “AUC” table, there is a lot of overlap between Cmax/MIC, T >MIC and AUC/MIC and in reality, while the pharmacokinetics and dynamics (as if I know what that means) suggest that a certain antibiotic can be dosed a certain way to achieve results, it is all related.
Vancomycin
As highlighted above, the efficacy of vancomycin depends on both time and concentration dependent killing. In other words, the best way to figure out the pharmacokinetics of vancomycin is to take a look at the AUC. People smarter than me have figure out that success of vancomycin is predicted by the ratio of AUC24 OVER MIC (aka AUC24/MIC, or the overall concentration of drug per liter over 24hrs above the MIC). The target here is >400 (again, as defined by people smarter than me). But note this target is defined for a vancomycin MIC of <1 (see later as to why this is important).
SO the best way to dose the vancomycin would be to calculate the AUC, right? But that is difficult to do, under the best of circumstances. For instance, you need to calculate several samples and apply the linear-trapezoidal rule to determine the AUC in a patient. Too much work! So what did people smarter than me recommend?
What if we use a surrogate marker for the AUC that is easy to measure? Thus, we use the trough, which is the concentration of the drug prior to the next dose of the drug. In evaluating the trough as a surrogate, simulations have found that shooting for a target trough of 15-20 will yield an AUC of >400:
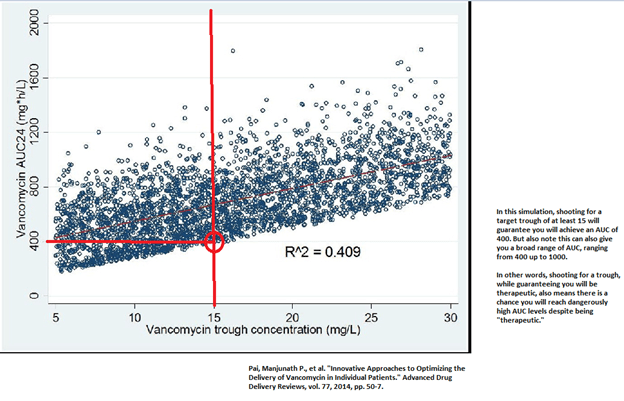
If you want to verify this by math, then the trough concentration that we shoot for is
- 17mg/L x24 = 408mg/Lxhr
- Again, keep in mind that the trough is only the lowest concentration before you give another dose. In other words, it does not account for what is going on before you measure it (again, too much work to stick people and calculate the AUC). SO even when you target troughs of 15, it is very likely that you will achieve that AUC of 400mg/L xhr
- Keep in mind, this AUC for effective antimicrobial activity only is true if the vanc MIC is < or equal to 1! (see later).
- Again, notice the variability of AUC when your trough is at least 15. IT can go up to >1200! Not only does this apply to simulations, but similar observations have been seen in clinical data:
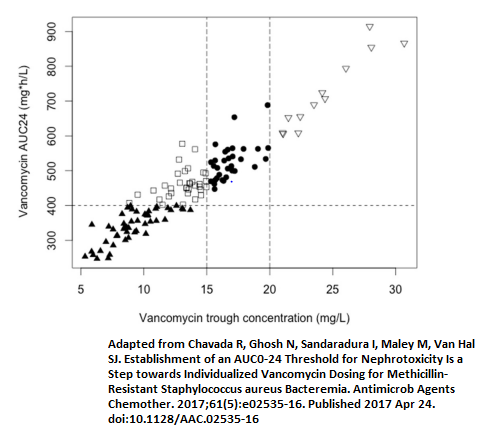
Now notice that between a trough of 15-20 gives you a AUC of 400-700 on this study. Notice also what goes up in this range:

In other words, the proportion of patients with AKI with an AUC of ~500 is nearly 20%. This proportion is correlated to the AUC, not the trough. Imagine a situation (as we saw above) where the trough is 15 but AUC is way higher than that of your “therapeutic range.”
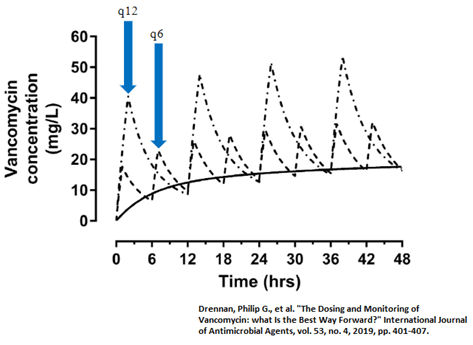
To further illustrate the point here, below is an example of how frequency of dosing can influence your AUC, without changing your “trough goal.”
The last thing to keep in mind is that the AUC of 400mg/L x hr being therapeutic only applies to an MIC to vancomycin of </= 1.
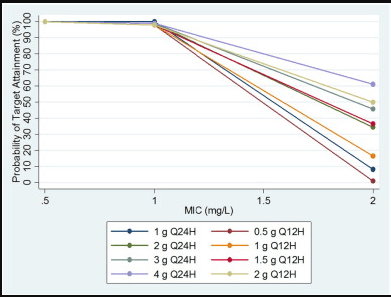
Above this, outcomes get worse. CLSI will flag a vancomycin MIC of 2 as susceptible, but for all purposes, MIC >1 = Resistant (especially with Staph aureus bacteremia!).
References:
Pai, Manjunath P., et al. “Innovative Approaches to Optimizing the Delivery of Vancomycin in Individual Patients.” Advanced Drug Delivery Reviews, vol. 77, 2014, pp. 50-7.
Chavada, R, et al. “Establishment of an AUC0-24 Threshold for Nephrotoxicity Is a Step Towards Individualized Vancomycin Dosing for Methicillin-Resistant Staphylococcus Aureus Bacteremia.” Antimicrobial Agents and Chemotherapy, vol. 61, no. 5, 2017.
Drennan, Philip G., et al. “The Dosing and Monitoring of Vancomycin: what Is the Best Way Forward?” International Journal of Antimicrobial Agents, vol. 53, no. 4, 2019, pp. 401-407.

1 comments on “Pharmacokinetics and Vancomycin!”