This one out of the one and only Mass Gen. Apparently they have issues with people ordering PCR for Anaplasma when it is not needed (they state 3% of those PCRs ordered ended up being positive). As such, they did a 2 phase study to see if they could use CBC and LFTs to screen prior to sending off anaplasma PCR. In phase 1, they did a retrospective analysis to determine if CBC or LFTs could be used to rule out Anaplasma. They took anaplasma PCR and any associated CBC and LFTs they had within 2 days, if multiple, they only took the closest labs to the anaplasma. During this 41 month period, they had 2165 specimens with only 69 testing positive for anaplasma (3.2%). Only 66 had a CBC or LFTs:
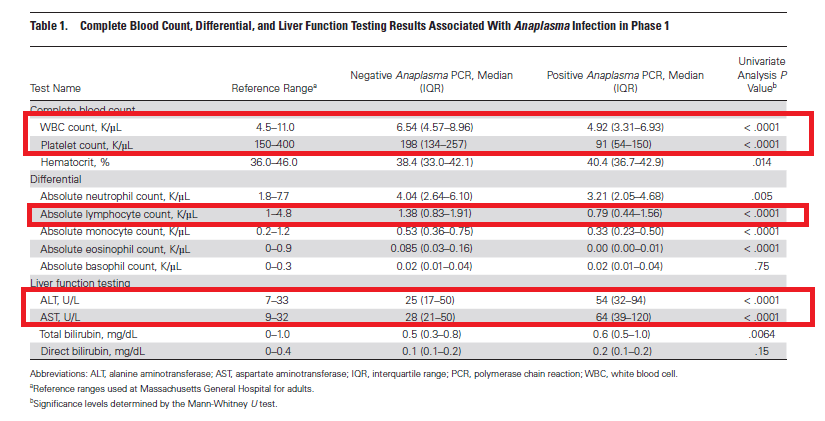
So those with Anaplasma had significantly lower WBC and Platelet count as well elevated LFTs. They deviced an algorithm to figure out who to screen and this involved looking at WBC and platelets (they threw out LFTs because this was not as commonly ordered test AND you can run the CBC w Diff from the same tube as the anaplasma PCR). They used a threshold of 11K for WBC and 300k for platelets (i.e. if you fell above, you did not get PCR) and found that this missed 3 cases. They further refined the algorithm to include those who were critically ill in the ICU OR had either SOT, SCT, leukemia/lymphoma, asplenia, immunosuppressive therapy (the patients that were missed had a combination of these).
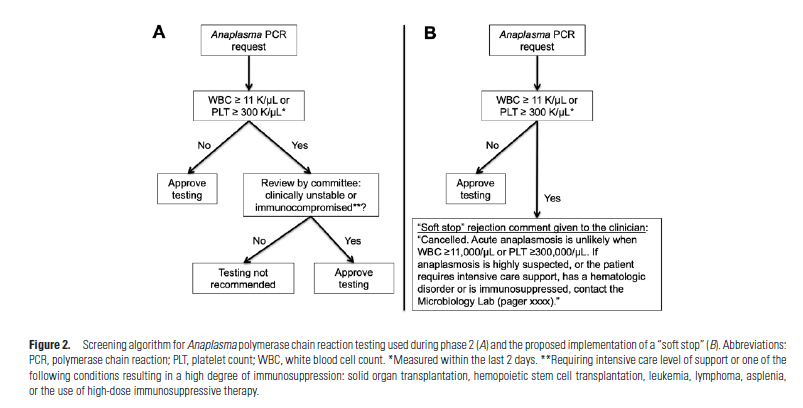
Phase 2 was a prospective “mock” implementation of the algorithm (They still approved all PCRs that were ordered, but a committee pretended to “reject” any test that did not meet the above criteria). They reviewed 663 cases during the mock period, during which the committee “Accepted” 96% of the true positive PCRs and only rejected 4% of those:

I think this is an interesting “proof of concept” study. This (Durham, NC) is not an endemic area for anaplasma, plus this is a single center study, so it is certainly not applicable. I wonder if something like this could be implemented for other tick borne illnesses. Either way, I included it here because I found it interesting.
