The antibody of childhood is largely a response to the dominant antigen of the virus causing the first Type A influenza infection of the lifetime. As the group grows older and subsequent infections take place, antibodies to additional families of virus are acquired. But the striking feature is…the antibody-forming mechanisms have been highly conditioned by the first stimulus, so later infections with strains of the same type successively enhance the original antibody to maintain it at the highest level at all times in that age group. This we have called the doctrine of the original antigenic sin.”
Thomas Francis, 1960.
The observations by Thomas Francis that a person’s antibody titers were highest to the strain of influenza they were first exposed to when subsequently exposed to another different strain has been called the “original antigenic sin” (1). Another example of this came from Hoskin’s in 1973, where he evaluated 800 children after vaccinating them with an influenza vaccine containing A/Hong Kong/68 (2). Following an outbreak of A/England/42/72 strain of influenza, sera were obtained from the children. In a subset of children who were vaccinated with A/Hong Kong/68, the development of antibodies against A/England/42/72 was less compared to the vaccine strain. In other words, those who got the vaccine had boosted antibodies when infected by a different strain. Using a relatively recent example, the 2009 influenza pandemic was a strain of influenza A(H1N1). The circulating strain from 1918 to 1957 was influenza A(H1N1), and those patients who were born before 1957 were likely infected with this strain. Due to this, it is believed that during the 2009 pandemic, these patients had partial immunity from severe disease to having being exposed to this original strain. As such, some observations from USA, Mexico, and Australia show that patients lower than expected infection rates in patients aged between 50-60 yo (3,4).
While the detailed mechanism remains to be fully elucidated (and if it were, I wouldn’t be talking about it), a good diagram comes from a review of this phenomena in context of influenza (5):
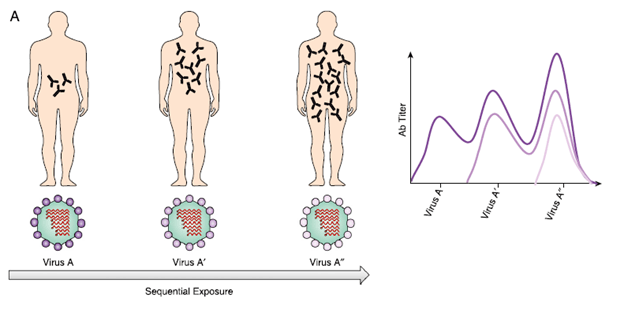
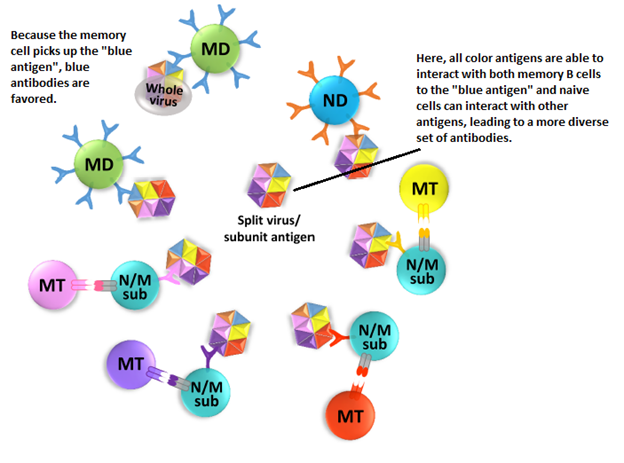
To keep it simple, the predominant antibody response tends to favor that of the first strain that was encountered. This does not mean antibodies will not be made towards other antigens, but rather that upon exposure to subsequent antigens, the original antibody will be “re-boosted.” A review proposes that memory B cells plays a major role in the mechanism (5). When a memory B cell captures an antigen that resembles a previously seen one (either a conserved or cross-reactive recognition site), these memory B cells kick start a mechanism that amplifies a previously antibody response. This is in comparison to naïve B cell, who may not mount an antibody against the already developed memory B cells. This has implications in vaccination. For instance, that same review proposed that a split virus/antigen may allow other native B cells to mount a memory response, rather than a “whole virus vaccine” which may favor an already memory B cell response that allows it to overtake the other antigens and favors the creating of more memory B cells and antibodies:
Dengue and the Antibody Dependent Enhancement:
Prior to discussing the data on influenza, I think it may be reasonable to discuss dengue and ADE as it pertains to the doctrine of the original antigenic sin. As it has been described in several case series, one of the risk factors for the development of severe dengue hemorrhagic fever is re-infection with a different strain of dengue virus. In one case series (6), 22 patients who had hemorrhagic shock all had secondary infection. Furthermore, a phase 3 vaccine trial (7) for the tetravalent dengue vaccine found that at year 3, there was a higher incidence of severe dengue in those who were aged <9yo when compared to the control group:
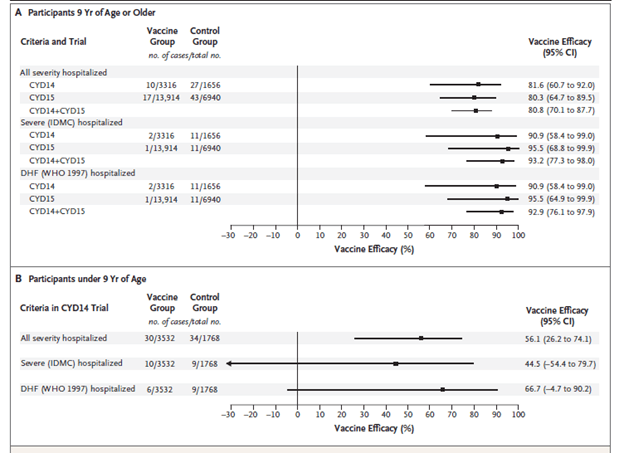
This observation was also noted in a RSV-immunization in children, with those who were immunized more likely to require subsequent hospitalization (8). So, what is going on here? There are 4 general strains that circulate, DENV 1-4. Before proceeding any further, here is the basic structure of IgG:
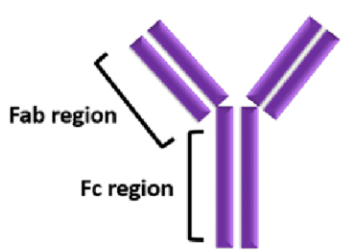
In general, virus elimination requires that an epitope (aka antigen) be recognized by the Fab region (9). The Fc region in turn binds to Fc gamma receptors in immune cells, leading to neutralization of the antigen:
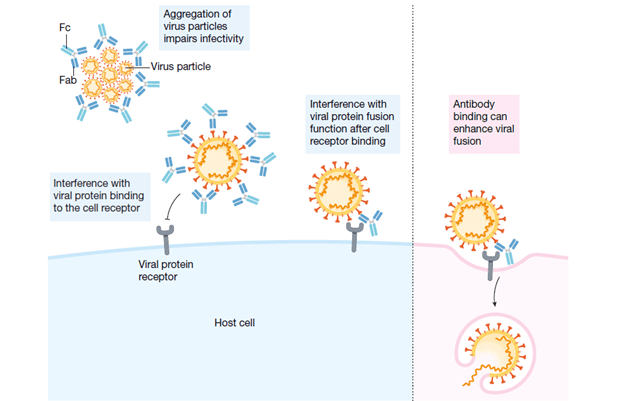
Cross-neutralization can occur if related viruses share the same epitope as that recognized by the original Fab of the IgG that was made by a previous virus. The Fc region in this relationship, in turn, may not have neutralizing activity and in fact, may facilitate the uptake, viral replication, and a detrimental cytokine response:
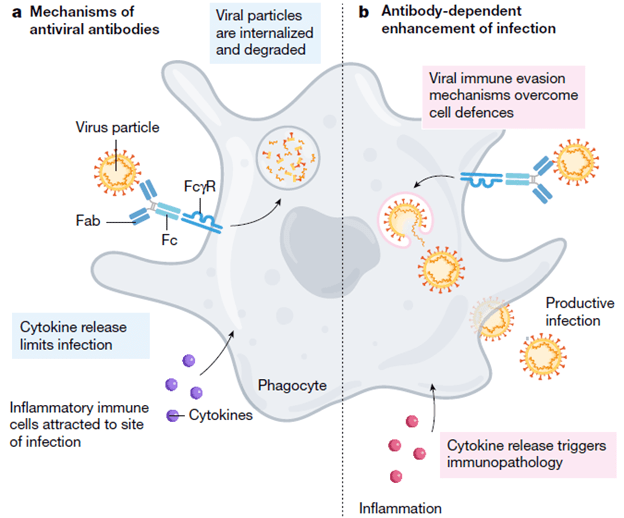
It should be noted that dengue virus actually targets monocytes, macrophages, and dendritic cells rather than neutrophils. Indeed, a review (10) notes that antibody dependent enhancement occurs in conditions of low antibody concentration or low antibody avidity, when the number of antibody molecules per virion is below the threshold necessary for neutralization of the virus (we will revisit this later). This, in turn, leads to activation of the complement pathway, leading to severe dengue infection.
There is some data suggesting how the original antigenic sin may play a role in pathophysiology for DHF. In one study (11) regarding serological responses to dengue infections, patients who had serum available from a primary and secondary dengue infection were evaluated. Patients who were exposed to another serotype the second time around had higher neutralizing antibodies to their original infection:

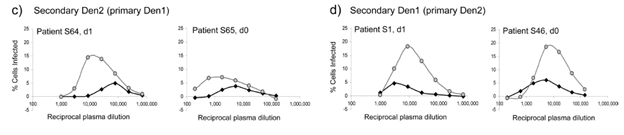
Furthermore, in another scenario of secondary infections, enhancement assays in 2 scenarios (Den1 followed by Den2 and Den2 followed by Den1) demonstrated that the concentration of immune serum required to enhance the second virus was greater than the concentration required to enhance the first virus:
The relationship of previous dengue antibodies and subsequent re-infection has been evaluated in several prospective studies. For instance, one prospective study of 153 Their children found that prior infection with a distinct serotype of Dengue was associated with increased incidence of hemorrhagic fever (12). Indeed, 17% of patients with a prior dengue infection were found to have hemorrhagic fever, compared to 0% of those who did not have a prior infection.
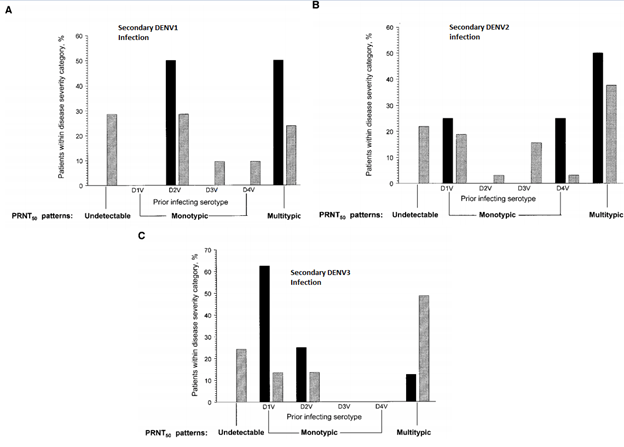
Notably, there was no statistical difference in incidence of dengue and dengue hemorrhagic fever in those who were subsequently infected with either DENV1 or DENV2 and had another infection with another strain in the past (though the correlation is there). The same study found that pre-exisiting levels of DENV3 antibodies were associated with lower viremia in DENV3 infections, a pattern not seen in DENV1 or DENV2. Despite this finding, not all antibody titers are equal. For instance, in one study of 6684 children (13) who had at least one DENV-Ab titer measured, those who pre-existing titers of 1:21 to 1:80 had a higher risk of dengue hemorrhagic fever (OR 7.64, 95% CI 3.19 to 18.28):
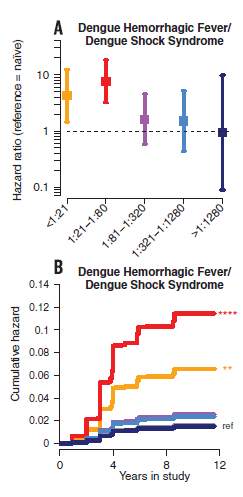
The OR of patients with severe hemorrhagic fever were higher when compared to controls:
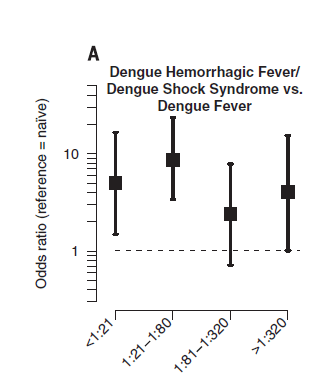
This suggests that high titers may actually have a neutralizing effect, but lower titers may harbor a deleterious effect, as mentioned previously. In a report of 604 patients with dengue hemorrhagic fever (14), 62% of patients had evidence of secondary dengue infection (by means of four-fold HI antibody response by day 4 of illness), suggesting this plays a role in DHF. Another study found that patients with secondary infection with a virus carrying a similar but distinct epitope can stimulate the proliferation of cross-reacting low affinity antibodies, leading to a higher degree of apoptosis in those who had severe DHF (15).
Influenza:
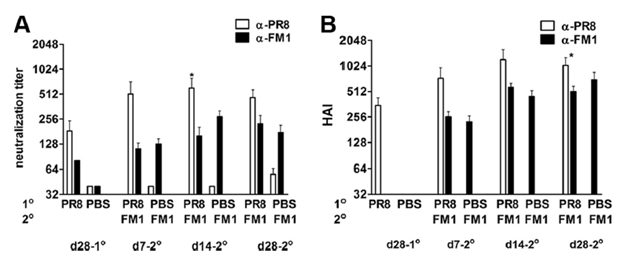
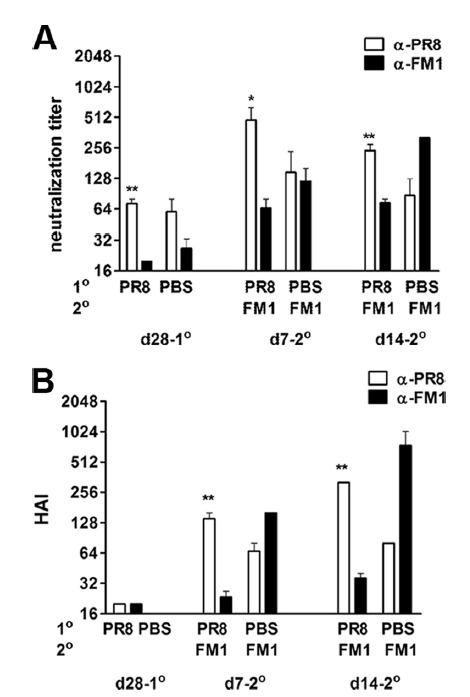
As mentioned previously, influenza has been the infection where many of the observations with regards to the antigenic sin have been made. For instance, in this animal study (16) , rats were immunized with H1N1 PR8 influenza (aka A/PR/8/34) followed by FM1 influenza (aka A/FM/1/47) a month later and antibodies against both were measured at day 28 post first shot, and days 7, 14, and 28 after the second shot. Neutralization titers against PR8 rose >2 fold at days 7 and 14 (p-value 0.27 and 0.12, respectively). At day 14 after immunization with FM1, the difference in titers between PR8 and FM1 was 4.5 fold (p-value 0.05), which dropped to insignificant value at day 28. This pattern of increase against HAI titers was also seen, with PR8 HAI titers being consistently higher than FM1 HAI titers following FM1 immunization:
When evaluating viral titers in mouse lung tissue, controls had significantly higher viral titers of FM1 virus. Notably, however, those who were immunized only with FM1 had significantly lower viral titers compared to those who were first immunized against PR8 followed by FM1:
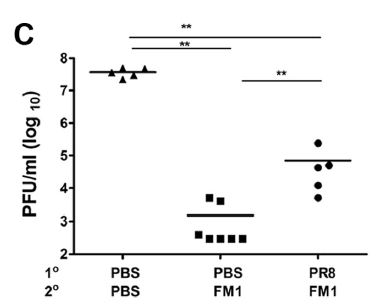
This suggest that the ability to mount a response against a live virus is impacted by the initial virus you are immunized against. Indeed, this was seen in PR8-HA primed mice, where FM1-HA vaccination induced a higher HAI against PR8:
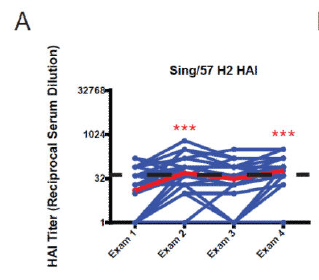
More compelling human data comes from a study evaluating the sera of 40 patients (17) from the Framingham Heart Study who were born between 1917 and 1952. Sera against H2N2, H3N2, H1N1 were evaluated. HAI titers encountered previously (i.e Jap/57 H2) increased over time:
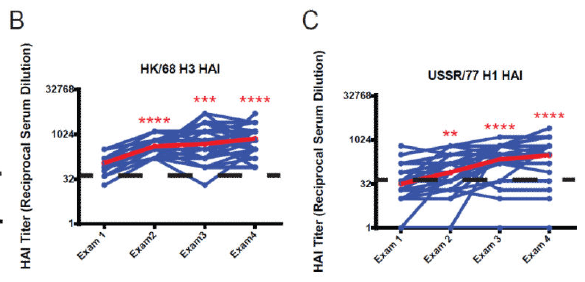
This pattern was also seen in HAI titers against HK/68 H3 and USSR/77 H1 (notably, these strains were seen earlier in 1968 and 1971, respectively):
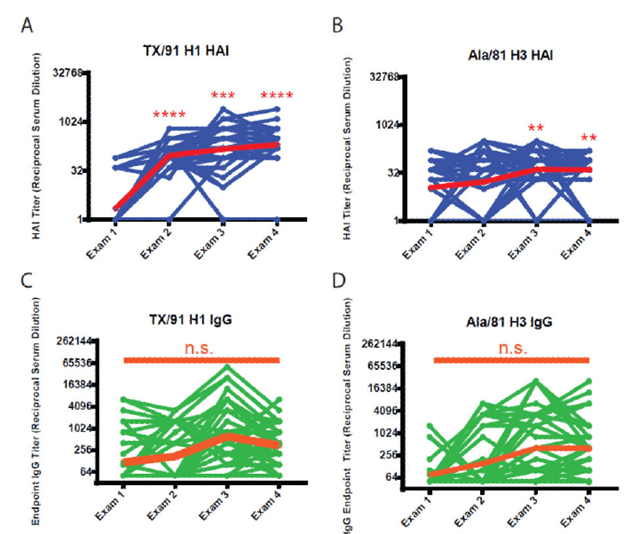
Interestingly, while the HAI increase was statistically significant, that for IgG was not. In another part of the study, the HAI titers against TX/91 and Ala/81 were evaluated. Not surprisingly, HAI titers against both increased over time, though IgG titers, while increasing, did not reach statistical significant:
Looking at the time of circulation of each strains, you can see that with each subsequent exposure to different strain leads to increase in previously seen influenza viruses rather than the one they were exposed to at that time:
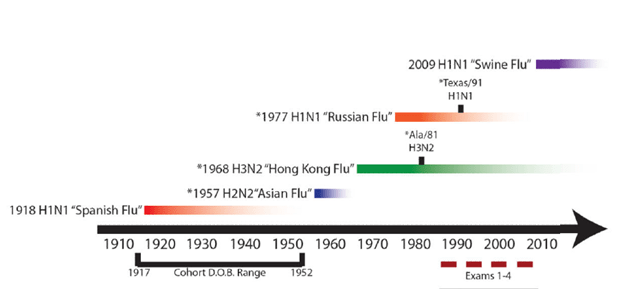
Another retrospective study (18) evaluated the serum of 151 patients for H3N2 antibodies and neutralization titers to strains circulating between 1968 and 2008. Neutralization titers were highest for H3N2 strains that circulated in a patient’s first decade of life with titers encountered later on in life being lower than the first strain:
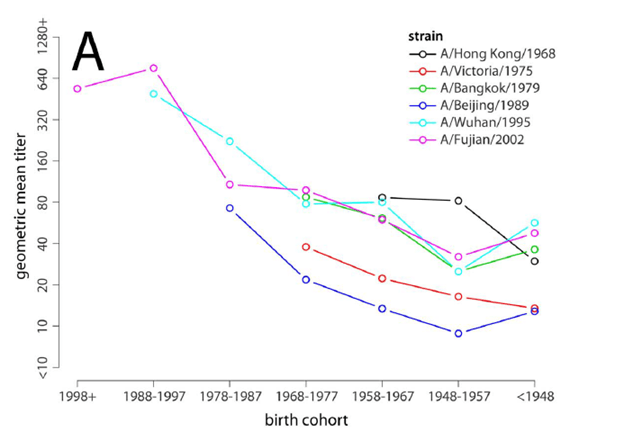
This is consistent with the original hypothesis of the original antigenic sin; the strains first encountered tend to be the highest overall. Notably, however, after age 60, all titers tended to increase, regardless of strain. The explanation for this, however, is unclear.
How does this play a role in immunization? One review (19) posits that vaccine effectiveness may stem from the “antigenic distance hypothesis” which states that difference in vaccine efficacy are due to the relative antigenic relatedness of pass vaccine strains, current vaccine strains, and circulating epidemic strains. If the distance between all strains is close, then there should be a high vaccine efficacy. One Canadian study (20) found that vaccine effectiveness did not go above 40% in each of its 3 seasons, with findings suggesting that prior vaccination may modify current vaccine effectiveness in context of antigenic distance hypothesis. Other studies suggest, however, that antigenic seniority may play a role.
One study (21) noted that patients who received a prior season’s influenza vaccine and its current vaccine (with the prior season vaccine being antigenically distinct from the current season strain) had lower vaccine effectiveness when compared to those who got only the current season vaccine (41% vs 75%), though it was better than in those who did not get a vaccine in either season:
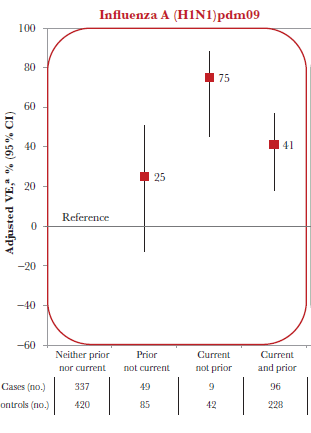
Furthermore, repeat vaccine recipients over 2 seasons had a 2 fold higher risk of H1N1 illness than those who were vaccinated for the current season only (OR 2.33, 95% CI 1.04-5.21). Finally, it was found that patients who were previously exposed to different H1N1 viruses had different vaccine efficacy, which varied by their birth cohort (22):
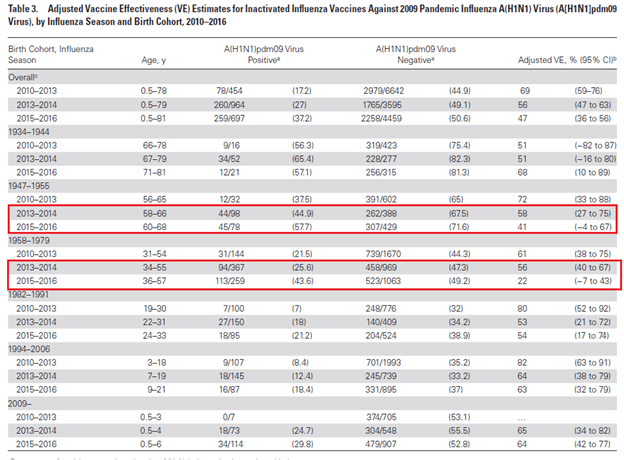
This suggest vaccine effectiveness within a cohort is influenced by the prior exposure to certain strains of H1N1, implying that perhaps, in a complex model, the OAN may play a role in this complicated issue. This, however, is far from the only explanation. Indeed, another review (30) evaluated the “Hoskins Paradox” where vaccinated subjects were more likely to be infected the following flu season. Despite this, and early concerns about annual immunization, the recommendation continues to be that of annual influenza immunization.
SARS-CoV2
This has been gaining some traction over the past few months. Some observations for the possible role of antigenic sin include the relative early seroconversion of IgG over IgM. For instance, one study found that 10 patients had seroconversion of IgG before IgM (23), however how this plays a role in pathophysiology is unclear. In one retrospective study (24), while IgM in general seroconverted first in most patients, those who were more critically ill were more likely to have higher levels of antibodies were more likely to have severe disease:
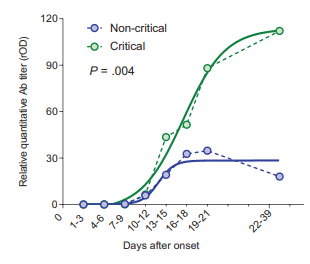
Another study found there was no cross-reactivity between SARS-CoV2 antibodies and MERS-CoV and SARS-CoV at least in the receptor binding domain (25), however they did cross react with other sections of the virus but they were not neutralizing antibodies. A review (26) proposes that previous infections with other human coronavirus do not predispose to more severe infections to viruses of the same lineage. Furthermore, neutralizing SARS-CoV2 antibodies have been used successfully, at least without worsening of the disease (27, 28).
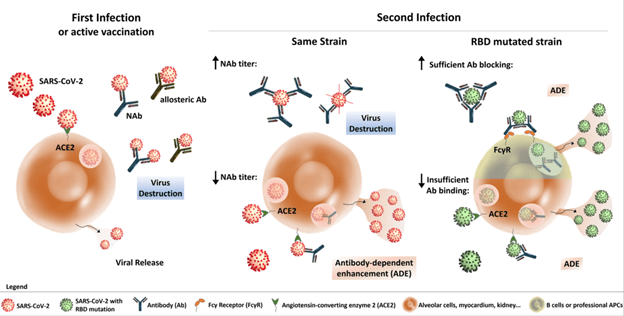
At this juncture, it seems that cross-reactivity between SARS-CoV2 and other human coronavirus antibodies may not play a role yet. In other words, prior infections with SARS-CoV may not convey any protective or deleterious effect for SARS-CoV2. Despite this, there is a concern about potential ADE with repeat infection or infection with a mutated strain. In terms of the former, as it was mentioned previously with Dengue, low neutralizing antibody titers following acute infection may lead to internalization of Ab-Ag complex and increase viral replication/increase cytokine release. In terms of the latter, RBD mutated strains may not allow for neutralization of the pathogen despite prior antibody and thus, antibody dependent enhancement may ensue (29).
TL;DR
- The Original Antigenic Sin refers to the immunological imprinting of the first viral infection encountered. If you encounter subsequent viral infections that differ in terms of antigens, the antibodies mounted in this second/third/fourth/etc infection will favor that of the first infection (in other words, the first antibodies will get “boosted” with subsequent infections, perhaps in favor of mounting antibodies against the subsequent infections). Simply put “the first infection in life orients immunological memory.”
- This may play a role in low vaccine efficacy in those who where exposed to different strains earlier in life or had repeat vaccinations
- The opposite may be true; infection with a different strain of a similar virus may lead to antibody dependent enhancement, where lack of significant neutralizing antibodies (either too low of titer or lack of cross-reactivity) causes increased viral load and subsequently worse outcomes (i.e. dengue hemorrhagic fever).
- This may play a role in the future of SARS-CoV2 (will immunization cause ADE, will infection with different strains lead to something similar seen in dengue, where the risk of mortality increases with infections of different strains).
References:
- Francis, Thomas. “On the Doctrine of Original Antigenic Sin.” Proceedings of the American Philosophical Society 104, no. 6 (1960): 572-78. Accessed November 11, 2020. http://www.jstor.org/stable/985534.
- Hoskins TW, Davies JR, Allchin A, Miller CL, Pollock TM. Controlled trial of inactivated influenza vaccine containing the a-Hong Kong strain during an outbreak of influenza due to the a-England-42-72 strain. Lancet. 1973 Jul 21;2(7821):116-20. doi: 10.1016/s0140-6736(73)93062-6. PMID: 4124042.
- Adalja AA, Henderson DA. Original antigenic sin and pandemic (H1N1) 2009. Emerg Infect Dis. 2010 Jun;16(6):1028-9. doi: 10.3201/eid1606.091653. PMID: 20507766; PMCID: PMC3086248.
- Krause R. The swine flu episode and the fog of epidemics. Emerg Infect Dis. 2006 Jan;12(1):40-3. doi: 10.3201/eid1201.051132. PMID: 16494715; PMCID: PMC3291407.
- Park MS, Kim JI, Park S, Lee I, Park MS. Original Antigenic Sin Response to RNA Viruses and Antiviral Immunity. Immune Netw. 2016 Oct;16(5):261-270. doi: 10.4110/in.2016.16.5.261. Epub 2016 Oct 25. PMID: 27799871; PMCID: PMC5086450.
- Sangkawibha N, Rojanasuphot S, Ahandrik S, Viriyapongse S, Jatanasen S, Salitul V, Phanthumachinda B, Halstead SB. Risk factors in dengue shock syndrome: a prospective epidemiologic study in Rayong, Thailand. I. The 1980 outbreak. Am J Epidemiol. 1984 Nov;120(5):653-69. doi: 10.1093/oxfordjournals.aje.a113932. PMID: 6496446.
- Hadinegoro SR, Arredondo-García JL, Capeding MR, Deseda C, Chotpitayasunondh T, Dietze R, Muhammad Ismail HI, Reynales H, Limkittikul K, Rivera-Medina DM, Tran HN, Bouckenooghe A, Chansinghakul D, Cortés M, Fanouillere K, Forrat R, Frago C, Gailhardou S, Jackson N, Noriega F, Plennevaux E, Wartel TA, Zambrano B, Saville M; CYD-TDV Dengue Vaccine Working Group. Efficacy and Long-Term Safety of a Dengue Vaccine in Regions of Endemic Disease. N Engl J Med. 2015 Sep 24;373(13):1195-206. doi: 10.1056/NEJMoa1506223. Epub 2015 Jul 27. PMID: 26214039.
- Fulginiti VA, Eller JJ, Sieber OF, Joyner JW, Minamitani M, Meiklejohn G. Respiratory virus immunization. I. A field trial of two inactivated respiratory virus vaccines; an aqueous trivalent parainfluenza virus vaccine and an alum-precipitated respiratory syncytial virus vaccine. Am J Epidemiol. 1969 Apr;89(4):435-48. doi: 10.1093/oxfordjournals.aje.a120956. PMID: 4305199
- Arvin AM, Fink K, Schmid MA, Cathcart A, Spreafico R, Havenar-Daughton C, Lanzavecchia A, Corti D, Virgin HW. A perspective on potential antibody-dependent enhancement of SARS-CoV-2. Nature. 2020 Aug;584(7821):353-363. doi: 10.1038/s41586-020-2538-8. Epub 2020 Jul 13. PMID: 32659783.
- Rothman AL. Immunity to dengue virus: a tale of original antigenic sin and tropical cytokine storms. Nat Rev Immunol. 2011 Jul 15;11(8):532-43. doi: 10.1038/nri3014. PMID: 21760609.
- Midgley CM, Bajwa-Joseph M, Vasanawathana S, Limpitikul W, Wills B, Flanagan A, Waiyaiya E, Tran HB, Cowper AE, Chotiyarnwong P, Grimes JM, Yoksan S, Malasit P, Simmons CP, Mongkolsapaya J, Screaton GR. An in-depth analysis of original antigenic sin in dengue virus infection. J Virol. 2011 Jan;85(1):410-21. doi: 10.1128/JVI.01826-10. Epub 2010 Oct 27. Erratum in: J Virol. 2011 Nov;85(22):12100. Chotiyarnwon, Pojchong [corrected to Chotiyarnwong, Pojchong]. PMID: 20980526; PMCID: PMC3014204.
- Endy TP, Nisalak A, Chunsuttitwat S, Vaughn DW, Green S, Ennis FA, Rothman AL, Libraty DH. Relationship of preexisting dengue virus (DV) neutralizing antibody levels to viremia and severity of disease in a prospective cohort study of DV infection in Thailand. J Infect Dis. 2004 Mar 15;189(6):990-1000. doi: 10.1086/382280. Epub 2004 Mar 1. PMID: 14999601.
- Katzelnick LC, Gresh L, Halloran ME, Mercado JC, Kuan G, Gordon A, Balmaseda A, Harris E. Antibody-dependent enhancement of severe dengue disease in humans. Science. 2017 Nov 17;358(6365):929-932. doi: 10.1126/science.aan6836. Epub 2017 Nov 2. PMID: 29097492; PMCID: PMC5858873.
- Halstead SB, Nimmannitya S, Cohen SN. Observations related to pathogenesis of dengue hemorrhagic fever. IV. Relation of disease severity to antibody response and virus recovered. Yale J Biol Med. 1970 Apr;42(5):311-28. PMID: 5419206; PMCID: PMC2591704.
- Mongkolsapaya J, Dejnirattisai W, Xu XN, Vasanawathana S, Tangthawornchaikul N, Chairunsri A, Sawasdivorn S, Duangchinda T, Dong T, Rowland-Jones S, Yenchitsomanus PT, McMichael A, Malasit P, Screaton G. Original antigenic sin and apoptosis in the pathogenesis of dengue hemorrhagic fever. Nat Med. 2003 Jul;9(7):921-7. doi: 10.1038/nm887. PMID: 12808447.
- Kim JH, Skountzou I, Compans R, Jacob J. Original antigenic sin responses to influenza viruses. J Immunol. 2009 Sep 1;183(5):3294-301. doi: 10.4049/jimmunol.0900398. Epub 2009 Jul 31. PMID: 19648276; PMCID: PMC4460008.
- Miller MS, Gardner TJ, Krammer F, Aguado LC, Tortorella D, Basler CF, Palese P. Neutralizing antibodies against previously encountered influenza virus strains increase over time: a longitudinal analysis. Sci Transl Med. 2013 Aug 14;5(198):198ra107. doi: 10.1126/scitranslmed.3006637. PMID: 23946196; PMCID: PMC4091683.
- Lessler J, Riley S, Read JM, Wang S, Zhu H, Smith GJ, Guan Y, Jiang CQ, Cummings DA. Evidence for antigenic seniority in influenza A (H3N2) antibody responses in southern China. PLoS Pathog. 2012;8(7):e1002802. doi: 10.1371/journal.ppat.1002802. Epub 2012 Jul 19. PMID: 22829765; PMCID: PMC3400560.
- Zhang A, Stacey HD, Mullarkey CE, Miller MS. Original Antigenic Sin: How First Exposure Shapes Lifelong Anti-Influenza Virus Immune Responses. J Immunol. 2019 Jan 15;202(2):335-340. doi: 10.4049/jimmunol.1801149. PMID: 30617114.
- Skowronski DM, Chambers C, De Serres G, Sabaiduc S, Winter AL, Dickinson JA, Gubbay JB, Fonseca K, Drews SJ, Charest H, Martineau C, Krajden M, Petric M, Bastien N, Li Y, Smith DJ. Serial Vaccination and the Antigenic Distance Hypothesis: Effects on Influenza Vaccine Effectiveness During A(H3N2) Epidemics in Canada, 2010-2011 to 2014-2015. J Infect Dis. 2017 Apr 1;215(7):1059-1099. doi: 10.1093/infdis/jix074. PMID: 28180277; PMCID: PMC5853783.
- Skowronski DM, Chambers C, Sabaiduc S, De Serres G, Winter AL, Dickinson JA, Gubbay JB, Drews SJ, Martineau C, Charest H, Krajden M, Bastien N, Li Y. Beyond Antigenic Match: Possible Agent-Host and Immuno-epidemiological Influences on Influenza Vaccine Effectiveness During the 2015-2016 Season in Canada. J Infect Dis. 2017 Dec 19;216(12):1487-1500. doi: 10.1093/infdis/jix526. PMID: 29029166; PMCID: PMC5853508.
- Flannery B, Smith C, Garten RJ, Levine MZ, Chung JR, Jackson ML, Jackson LA, Monto AS, Martin ET, Belongia EA, McLean HQ, Gaglani M, Murthy K, Zimmerman R, Nowalk MP, Griffin MR, Keipp Talbot H, Treanor JJ, Wentworth DE, Fry AM. Influence of Birth Cohort on Effectiveness of 2015-2016 Influenza Vaccine Against Medically Attended Illness Due to 2009 Pandemic Influenza A(H1N1) Virus in the United States. J Infect Dis. 2018 Jun 20;218(2):189-196. doi: 10.1093/infdis/jix634. PMID: 29361005; PMCID: PMC6009604.
- Long Q, Deng H, Chen J, Hu J, Liu B, Liao P, et al. Antibody responses to SARS-CoV-2 in COVID-19 patients: the perspective application of serological tests in clinical practice. medRxiv. (2020). doi: 10.1101/2020.03.18.20038018
- Zhao J, Yuan Q, Wang H, Liu W, Liao X, Su Y, Wang X, Yuan J, Li T, Li J, Qian S, Hong C, Wang F, Liu Y, Wang Z, He Q, Li Z, He B, Zhang T, Fu Y, Ge S, Liu L, Zhang J, Xia N, Zhang Z. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019. Clin Infect Dis. 2020 Mar 28:ciaa344. doi: 10.1093/cid/ciaa344. Epub ahead of print. PMID: 32221519; PMCID: PMC7184337.
- Ju, B., Zhang, Q., Ge, J. et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature 584, 115–119 (2020). https://doi.org/10.1038/s41586-020-2380-z
- Arvin AM, Fink K, Schmid MA, Cathcart A, Spreafico R, Havenar-Daughton C, Lanzavecchia A, Corti D, Virgin HW. A perspective on potential antibody-dependent enhancement of SARS-CoV-2. Nature. 2020 Aug;584(7821):353-363. doi: 10.1038/s41586-020-2538-8. Epub 2020 Jul 13. PMID: 32659783.
- Duan K, Liu B, Li C, Zhang H, Yu T, Qu J, Zhou M, Chen L, Meng S, Hu Y, Peng C, Yuan M, Huang J, Wang Z, Yu J, Gao X, Wang D, Yu X, Li L, Zhang J, Wu X, Li B, Xu Y, Chen W, Peng Y, Hu Y, Lin L, Liu X, Huang S, Zhou Z, Zhang L, Wang Y, Zhang Z, Deng K, Xia Z, Gong Q, Zhang W, Zheng X, Liu Y, Yang H, Zhou D, Yu D, Hou J, Shi Z, Chen S, Chen Z, Zhang X, Yang X. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Natl Acad Sci U S A. 2020 Apr 28;117(17):9490-9496. doi: 10.1073/pnas.2004168117. Epub 2020 Apr 6. PMID: 32253318; PMCID: PMC7196837.
- Li L, Zhang W, Hu Y, Tong X, Zheng S, Yang J, Kong Y, Ren L, Wei Q, Mei H, Hu C, Tao C, Yang R, Wang J, Yu Y, Guo Y, Wu X, Xu Z, Zeng L, Xiong N, Chen L, Wang J, Man N, Liu Y, Xu H, Deng E, Zhang X, Li C, Wang C, Su S, Zhang L, Wang J, Wu Y, Liu Z. Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial. JAMA. 2020 Aug 4;324(5):460-470. doi: 10.1001/jama.2020.10044. Erratum in: JAMA. 2020 Aug 4;324(5):519. PMID: 32492084; PMCID: PMC7270883.
- Ulrich H, Pillat MM, Tárnok A. Dengue Fever, COVID-19 (SARS-CoV-2), and Antibody-Dependent Enhancement (ADE): A Perspective. Cytometry A. 2020 Jul;97(7):662-667. doi: 10.1002/cyto.a.24047. Epub 2020 Jun 7. PMID: 32506725; PMCID: PMC7300451.
- Monto AS, Malosh RE, Petrie JG, Martin ET. The Doctrine of Original Antigenic Sin: Separating Good From Evil. J Infect Dis. 2017 Jun 15;215(12):1782-1788. doi: 10.1093/infdis/jix173. PMID: 28398521; PMCID: PMC5853211.
