Hantaviruses are a thing. I am sure the vast majority of those who come across this post will not know or even see a case of hantavirus (as far as I am concerned, though keep reading).
Basics
Bunyaviridae is part of a larger family of viruses that cause hemorrhagic fevers (those other viruses include the arenaviruses, filoviruses, and flaviviruses). Within this family, there are four genera – bunyaviruses, phleboviruses, nairovirus, and hantavirus (1):

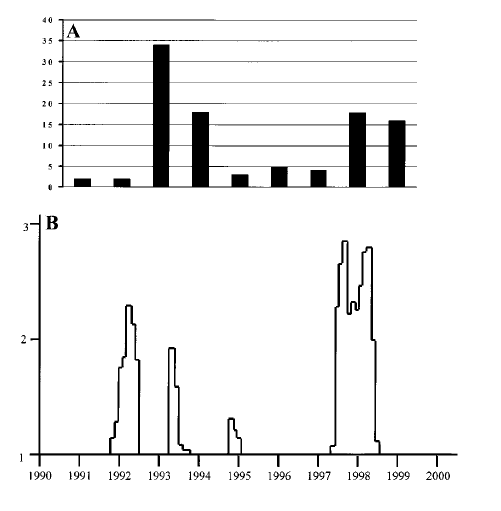
One of the characteristics that sets it apart from the other types of bunyaviridae is the fact there is no arthropod vector that is associated with human infection (2,3) with rodent hosts shedding viruses through saliva, urine, and feces with aerosols being the main route of transmission in humans (3). Other routes of transmission have been described, including transmission by bite with an infected animal as well as outbreaks being associated mites (4). For instance, a review found that outbreaks in China with viruses that caused hantavirus fever and renal syndrome (HFRS) was associated with increased mite density in the host rats, with lower rates associated with lower incidence of HFRS (4). Environmental influence on outbreaks was also seen in the 1997-1998 El Niño-Southern Oscillation (7). While the details about this phenomena is outside of my scope, the hypothesis is that increased precipitation increased rodent population densities. In this retrospective analysis, the ENSO event of 1997 lead to an increase in cases the following year:
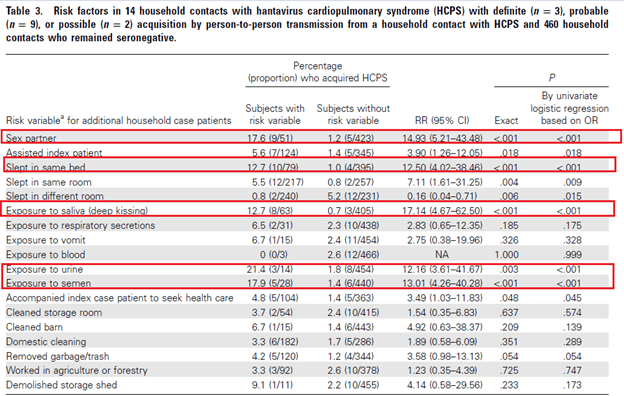
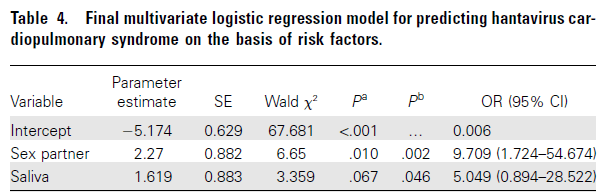
In general, hantaviruses have no evidence of person-to-person transmission however the exception to this seems to be the Andes virus. A retrospective analysis of 421 household contacts and 76 index cases found that risk factors associated with acquisition of Andes virus to be sleeping in the same bed or room, exposure to saliva, urine and semen:
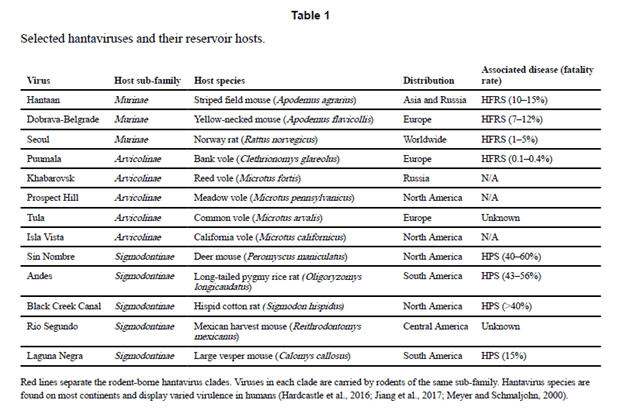
One of the issues that comes with learning about these viruses (or really, any virus such as Flaviviridae with several isolated types) is the staggering number of viruses and hosts there are. For instance, this review evaluated several selected hantaviruses:
This may make it a daunting task for learning them. But as it so happens, there is a way to divide them, at least the clinical syndromes (5):
- New world viruses – these usually cause hanta pulmonary syndrome and tend to be seen in the Americas
- Old world viruses – usually cause HFRS and are seen in Europe and Asia
- Non-virulent viruses – these are found in both the old and new world and are associated with either mild disease (as in the case of Puumala virus) or non-virulent.
While the structure of the virus is not terribly interesting or useful clinically, I will bring a few things to your attention:
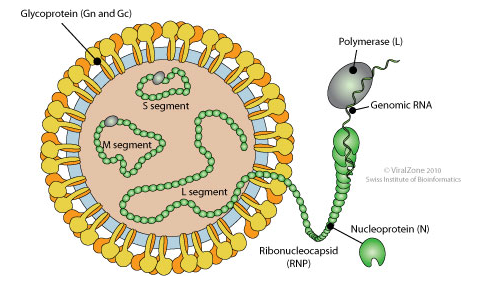
The two structures to consider here are the nucleoprotein (N) and the glycoprotein (Gn and Gc). This is because diagnostics involves antibody presence against N antigen and viral G antigen (the latter of which is highly specific; 6).
Geography:
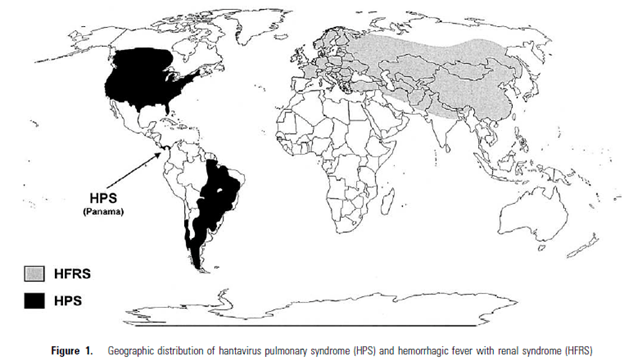
These viruses are found throughout Europe, Asia, and the Americas (2). As mentioned previously, clades of viruses of the Old World and New World cause different diseases. In general, any rodent of the family Muridae tends to harbor these viruses, however they have not been seen in rodents in Africa, Australia, or Antarctica for unclear reasons:
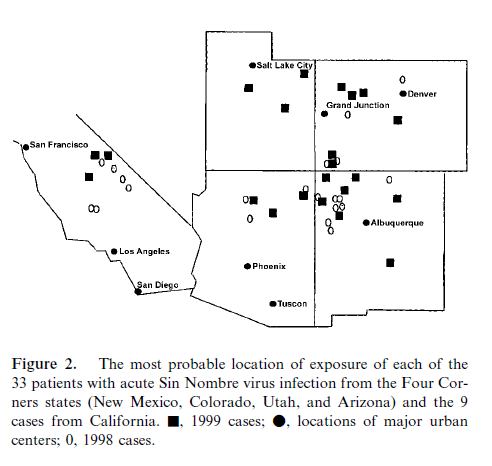
The most common virus within the US is the Sin Nombre Virus (yet to be named..) and it has been mostly found in the “Four Corner’s Area” and California. For instance, a case series of 5 patients were associated with exposure within this area of the country (8), and in the prior study evaluating the ELSO phenomena found the outbreak was concentrated in this area:
A review from NEJM (9) also found 18 patients, of which all lived within the Four Corners area.
Clinical Syndrome
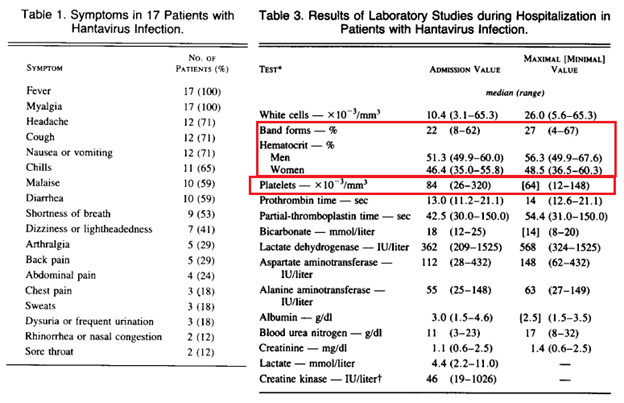
The incubation period for hantaviruses range from 0-33 days, with a median period of incubation being anywhere from 14-17 days (11). A report of 18 patients who were initially described from the four corners area (9) found the most common symptoms in the prodromal period to be fever, myalgia, headache, cough, nausea and vomiting, as well as GI symptoms. Laboratory studies associated with hantaviruses included elevated white count with atypical lymphocytes, elevated hematocrit, and thrombocytopenia:
Symptoms seem to be in stages. For instance, a retrospective analysis of 140 patients enrolled in a Ribavirin study were evaluated to look at differentiating characteristics of those who were ultimately diagnosed with HPS (12). Patients who had myalgias at onset of their illness, at initial outpatient visit and at admission to the hospital were more likely to have HPS. Further nausea became more common as the disease progressed. The presence of dyspnea and chest pain was associated with decreased OR for HPS in the early stages as well:

Notably, cough and coryza are absent in the initial stages. Upon admission, there seems to be a rapid decline in a significant proportion of patients. In a review (2) found that at around 48h after admission, all patients have evidence of interstitial edema and two-thirds of patients develop air-space disease. There is a rapid decompensation and 30-40% of patients die 24-48hrs after admission (2). By far, the most common symptoms are pulmonary and cardiac rather than renal. There are some subsequent reported renal issues. For instance, one retrospective study (31) found that 15/30 patients had proteinuria >150mg, however none had nephrotic range proteinuria. 7 patients had a CrCl <90 while 2 had CrCl <2 on follow up. This may not be evident until several weeks following initial illness.
The stages of HFRS tend to be a bit different (13). Incubation ranges from 2-3 weeks and varies from 5 to 42 days. In the prototypical disease, Korean hemorrhagic fever, the disease can be divided into the following phases:
- Febrile: last 3-8 days with fevers, chills, general malaise, myalgia, headache. During this phase, there is flushing on the face, neck, chest followed by conjunctival hemorrhage. Essentially this is flu + redness.
- Hypotensive – as it sounds, this is due to decreased cardiac output and increased capillary leakage.
- Oliguric – last 3 to 7 days. This occurs along with the typical renal injury symptoms.
- Diuretic – think of it as “ATN.” Diuresis occurs for days or weeks and typically 3 to 6L daily of diuresis is seen.
- Convalescent
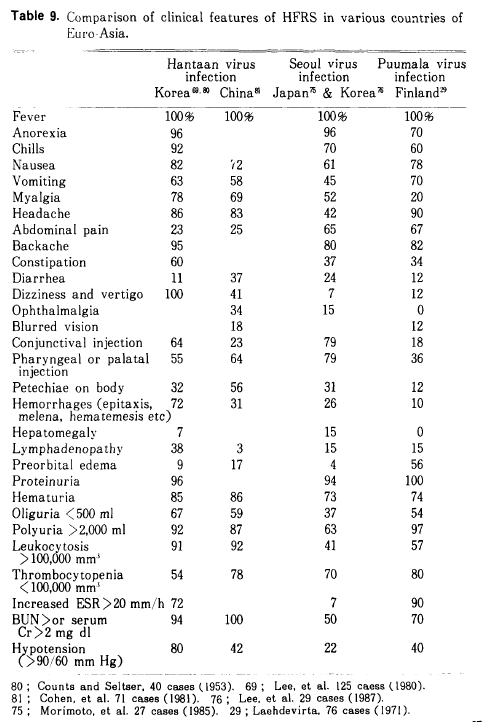
A review (3) also notes distinguishing characteristics between HPS and HFRS:

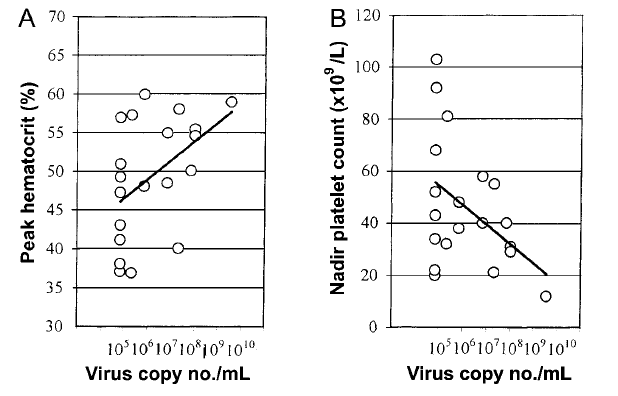
In terms of lab values, the 3 more commonly seen (and likely more useful) are early thrombocytopenia, elevated circulating immunoblasts (usually atypical lymphocytes), and elevated hematocrit. Logistic regression in one analysis (12) found that hematocrit level and platelet count were significant predictors of HPS status. On the initial stages of the illness, thrombocytopenia and high hematocrit level tend to be the rule:
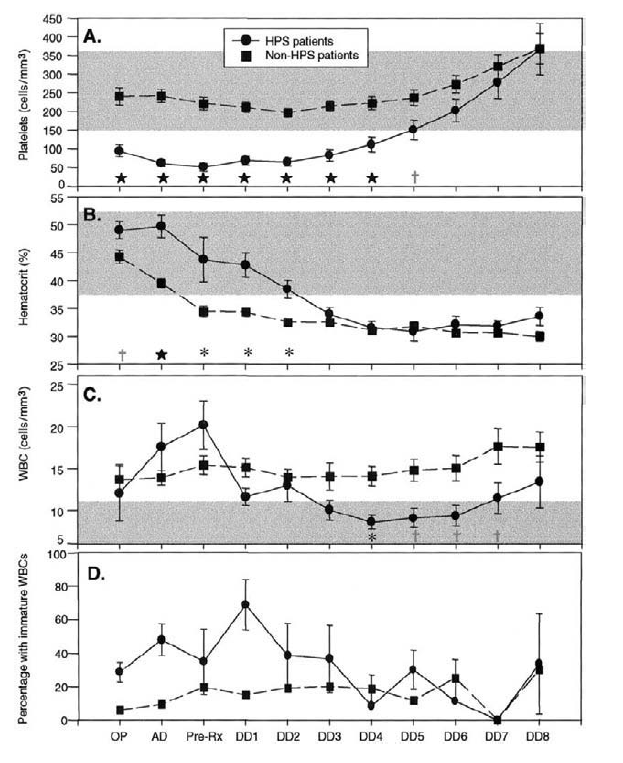
Similarly, lab results in a case series (8) of 5 patients found atypical lymphocytes, thrombocytopenia, and elevated hematocrit:
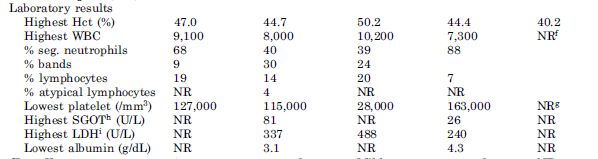
In general, mortality tends to be fairly high. For instance, in one initial series, the mortality was as high as 77% (9). One review quoted the mortality rates to be 12% for HFRS and 60% in other outbreaks of HPS (14). In the US, HPS has a mortality rate of roughly 35% (15), while for Puumala virus mortality tends to be roughly 0.4-0.5% (16). A small case series (27) found that cardiac index <2.5, lactate >4, PEA/VFib/VT, and refractory shock were associated with 100% mortality.
Diagnosis
As mentioned previously, the nucleocapsid protein and glycoproteins are important in the diagnosis of Hantaviruses as they tend to be the major antigenic target in the early antibody response. Assays that are used are usually ELISA protocols with either native N protein antigen (aka a home-brew), baculovirus-expressed antigens, or E.-coli expressed antigens. Why is this the case? Because it is difficult to grow hantaviruses in the lab (they replicate slowly), have a variable yield in cell cultures, and it is hazardous to handle it. So we just stick DNA into a different organism, get the N antigen, and use that (obviously more complicated, but this suffices for now). In a study evaluating several assays for Puumala virus (17), the sensitivity of IgM assays was high when using the full N protein genome compared to a shorter N protein genome:

IgG assays also had fairly good sensitivity and specificities. This dependent on the method of capture (monoclonal antibody-capture ELISA having higher sensitivities compared to ELISA based on direct coating), as well as the length of the N protein genome:

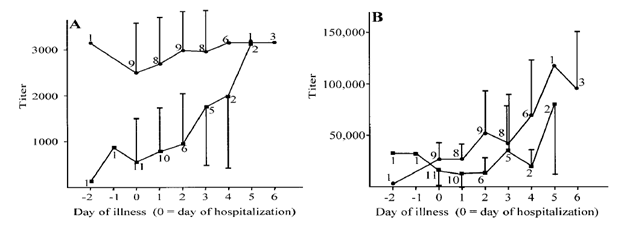
The antibody response seems to occur early on, including for neutralizing antibodies. In a retrospective study for 26 patients with several serum samples (18) obtained within 24 hours, all patients had a level of IgM N and IgG antibodies by day one of illness:
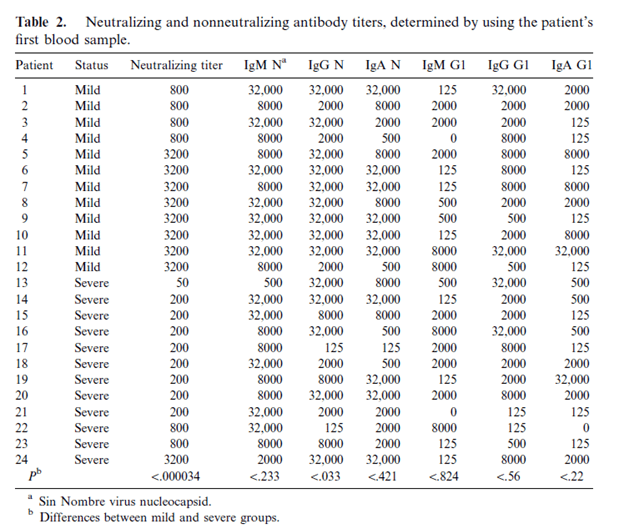
Notably, those who were less ill were more likely to have a titer <800 for neutralizing antibodies, suggesting a protective effect against illness severity. Furthermore, the levels of both titers increased over the hospitalization:
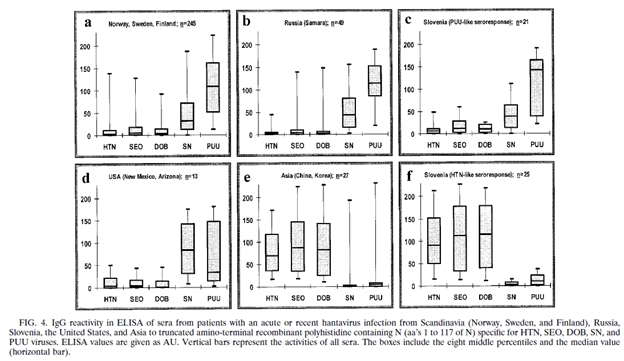
Another study (19) evaluated the efficacy of an ELISA for the serological diagnosis of several hantavirus infections based with the E.coli recombinant N antigen from 5 different hantaviruses (Hantaan, Seoul, Dobrava, Sin Nombre, Puumala). The sera from acute hantavirus infections were positive in those cases compared to healthy Swedish volunteers. Notably, depending on where the patient was from, IgG and IgM antibody varied. For instance, patients from Scandinavia and Russia reacted strongest to Puumala virus, while patients from Asia reacted to Hantaan, Seoul, and Dobravirus:
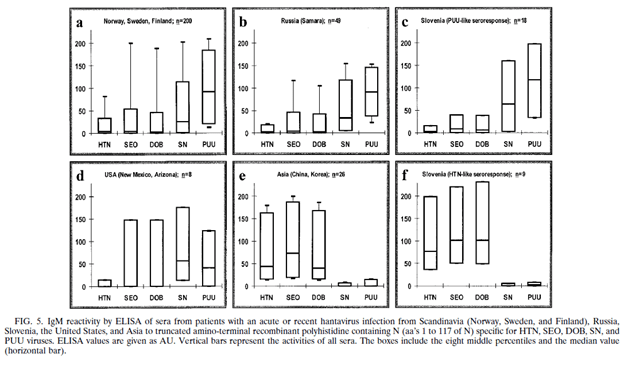
Immunoblot POC assays are also available with fairly good performance.
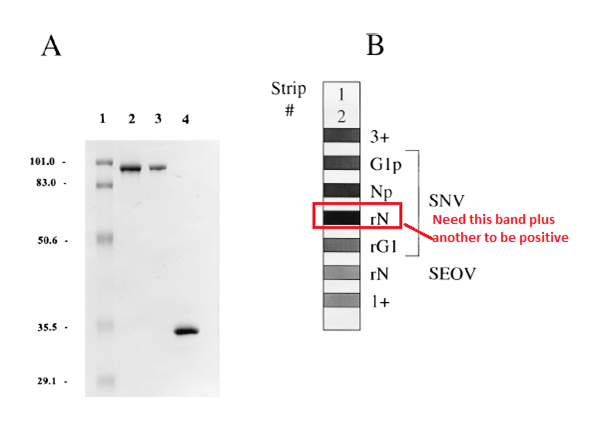
In one study (20) evaluating reactivities (using reactivity to rN antigen and another antigen) found 58/59 of samples with acute HPS. Four out of four other samples tested positive for Sin Nombre virus when they had another Hantavirus.
What about PCR? While it can be used, it is not widely available in many labs. The sensitivity of RT-PCR has been evaluated to be as low as 103 copies per reaction (30). In the same study, 20 out of 26 samples obtained on the day of admission were RT-PCR positive.
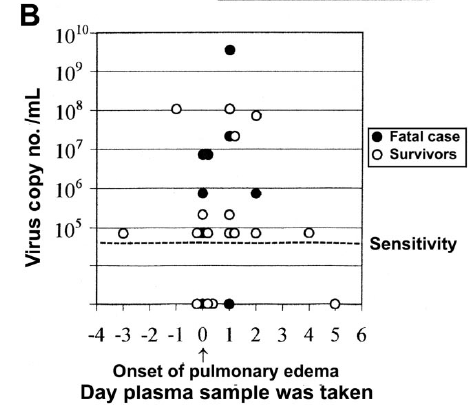
There was notably, a small correlation between peak hematocrit, nadir platelets, and viral load (r=0.46 for hematocrit, p<0.05, r=0.44, p=0.05).
Therapy
In general, as this looks like ARDS and shock, supportive therapy is the go-to along with early recognition. Ribavirin, a broad-spectrum antiviral, has been shown in vitro to have activity against Hantaviruses (21). Initial clinical data came from a prospective, double-blind, placebo-controlled trial (22) from China, which evaluated 242 patients with HFRS. Mortality was significantly reduced in the ribavirin group seven-fold. In those who HCP, the data is less clear. An open label study (23) of 30 patients with Hantavirus infection who got ribavirin for 7 days compared to those who did not get ribavirin did not show mortality improvement:
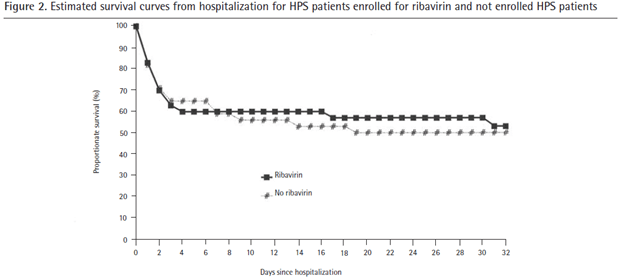
There were notable differences between these groups, as the “control” group were identified from 18 other states not part of the study group. For instance, 18% of the patients were infected with different variants of hantaviruses and 41% were identified in states where pre-test probability was quite low. Furthermore, as the therapy had to be shipped, there were significant delays in implementation, which may have affected the results. All in all, there is little to suggest any benefit but there are issues with the study itself. Furthermore, only 26% of patients were able to complete the therapy. Another smaller RCT (24) of 23 patients did not find mortality benefit at 28 days after enrollment, even when administering the drug in the prodromal phase:
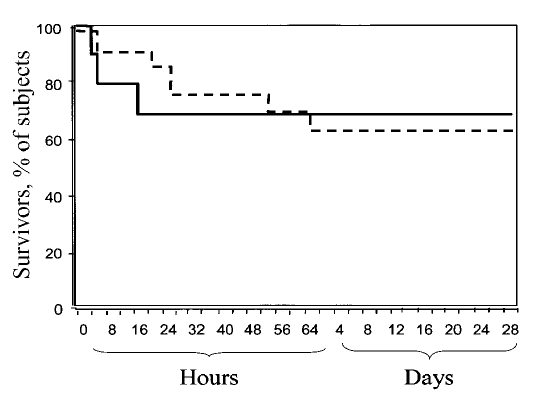
In another prospective study (25) found that ribavirin improved the development of oliguria and dialysis compared to historical controls, however these were not adjusted. All in all, it is difficult to suggest the benefit of ribavirin for hantavirus infection, given the lack of overwhelming data.
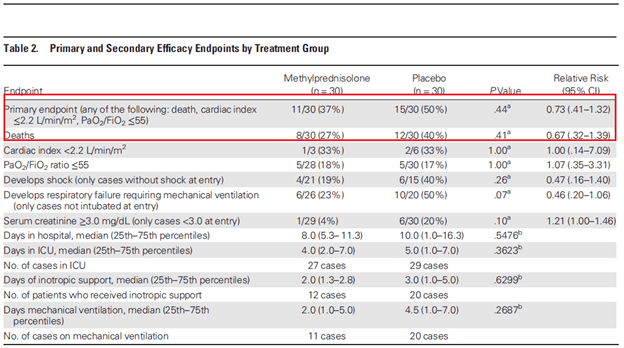
What about steroids? There is little data here as well. While they have been used in case reports, the only study was a phase , double-blind, RCT that evaluated methylprednisolone and placebo in 60 patients (26) with Andes virus. Primary endpoint was a composite of 28 day mortality, P/F ratio <55, CI <2.2, PEA, VT or Vfib. While patients in the placebo group were more likely to have shock (50% vs 30%), be intubated at enrollment (33.3% vs 13.3%) and have higher SOFA scores (7.9 vs 6.1), there were no differences in the composite primary endpoint:
The University of New Mexico has had significant experience with ECMO. A retrospective study of 38 patients (28) who were treated with VA ECMO found that in-hospital mortality was 39.5% compared to the expected 100% of those who had significant illness. Pre-ECMO cardiac arrest, pre-ECMO MAP, and pre-ECMO lactate were associated with increased mortality:
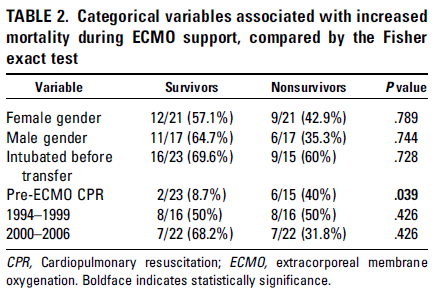
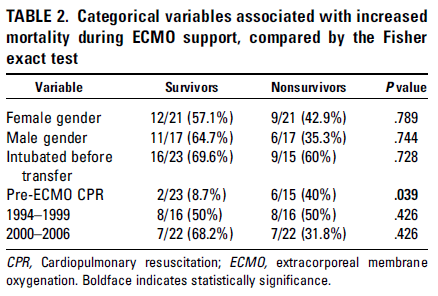
Another retrospective study of 51 patients (29) compared different cannulation strategies (not cannulated until unstable vs elective cannulation after arrival to the hospital). In-hospital mortality here was 33.3%, with no difference between cannulation strategies. The only variable associated with increased mortality was elevated serum lactate (p-value 0.001). Again, these results were compared to the 100% mortality risk factors that were previously described. While I do not anticipate a well controlled trial or at least a prospective trial to answer this question, ECMO seems to be a reasonable option if there is a suspicion for hantavirus infection, as these patients tend to decompensate quite quickly.
References:
- Shope RE. Bunyaviruses. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 56. Available from: https://www.ncbi.nlm.nih.gov/books/NBK8004/
- C. J. Peters, Ali S. Khan, Hantavirus Pulmonary Syndrome: The New American Hemorrhagic Fever, Clinical Infectious Diseases, Volume 34, Issue 9, 1 May 2002, Pages 1224–1231,
- Schmaljohn, C., & Hjelle, B. (1997). Hantaviruses: a global disease problem. Emerging infectious diseases, 3(2), 95–104. https://doi.org/10.3201/eid0302.970202
- Yu XJ, Tesh RB. The role of mites in the transmission and maintenance of Hantaan virus (Hantavirus: Bunyaviridae). J Infect Dis. 2014 Dec 1;210(11):1693-9. doi: 10.1093/infdis/jiu336. Epub 2014 Jun 23. PMID: 24958909; PMCID: PMC4296190.
- Mittler E, Dieterle ME, Kleinfelter LM, Slough MM, Chandran K, Jangra RK. Hantavirus entry: Perspectives and recent advances. Adv Virus Res. 2019;104:185-224. doi: 10.1016/bs.aivir.2019.07.002. Epub 2019 Aug 7. PMID: 31439149; PMCID: PMC6881143.
- Bharadwaj M, Nofchissey R, Goade D, Koster F, Hjelle B. Humoral immune responses in the hantavirus cardiopulmonary syndrome. J Infect Dis. 2000 Jul;182(1):43-8. doi: 10.1086/315657. Epub 2000 Jun 27. PMID: 10882580.
- Hjelle B, Glass GE. Outbreak of hantavirus infection in the Four Corners region of the United States in the wake of the 1997-1998 El Nino-southern oscillation. J Infect Dis. 2000 May;181(5):1569-73. doi: 10.1086/315467. Epub 2000 May 15. PMID: 10823755.
- Kitsutani PT, Denton RW, Fritz CL, Murray RA, Todd RL, Pape WJ, Wyatt Frampton J, Young JC, Khan AS, Peters CJ, Ksiazek TG. Acute Sin Nombre hantavirus infection without pulmonary syndrome, United States. Emerg Infect Dis. 1999 Sep-Oct;5(5):701-5. doi: 10.3201/eid0505.990512. PMID: 10511527; PMCID: PMC2627725.
- Duchin JS, Koster FT, Peters CJ, Simpson GL, Tempest B, Zaki SR, Ksiazek TG, Rollin PE, Nichol S, Umland ET, et al. Hantavirus pulmonary syndrome: a clinical description of 17 patients with a newly recognized disease. The Hantavirus Study Group. N Engl J Med. 1994 Apr 7;330(14):949-55. doi: 10.1056/NEJM199404073301401. PMID: 8121458.
- Ferres M, Vial P, Marco C, Yanez L, Godoy P, Castillo C, Hjelle B, Delgado I, Lee SJ, Mertz GJ; Andes Virus Household Contacts Study Group. Prospective evaluation of household contacts of persons with hantavirus cardiopulmonary syndrome in chile. J Infect Dis. 2007 Jun 1;195(11):1563-71. doi: 10.1086/516786. Epub 2007 Apr 16. PMID: 17471425.
- Young JC, Hansen GR, Graves TK, Deasy MP, Humphreys JG, Fritz CL, Gorham KL, Khan AS, Ksiazek TG, Metzger KB, Peters CJ. The incubation period of hantavirus pulmonary syndrome. Am J Trop Med Hyg. 2000 Jun;62(6):714-7. doi: 10.4269/ajtmh.2000.62.714. PMID: 11304061.
- Chapman LE, Ellis BA, Koster FT, Sotir M, Ksiazek TG, Mertz GJ, Rollin PE, Baum KF, Pavia AT, Christenson JC, Rubin PJ, Jolson HM, Behrman RE, Khan AS, Bell LJ, Simpson GL, Hawk J, Holman RC, Peters CJ; Ribavirin Study Group. Discriminators between hantavirus-infected and -uninfected persons enrolled in a trial of intravenous ribavirin for presumptive hantavirus pulmonary syndrome. Clin Infect Dis. 2002 Feb 1;34(3):293-304. doi: 10.1086/324619. Epub 2001 Dec 13. PMID: 11774075.
- Lee, PW. Immunofluorescent antibody technique. In: Lee, HW, Dalrymple, JM, eds. Manual of hemorrhagic fever with renal syndrome. WHO Collaborating Centre for Virus Reference and Research, Institute for Virus Diseases, Korea University, 1989: 77–82.
- Jonsson, C. B., Figueiredo, L. T., & Vapalahti, O. (2010). A global perspective on hantavirus ecology, epidemiology, and disease. Clinical microbiology reviews, 23(2), 412–441. https://doi.org/10.1128/CMR.00062-09
- Hantavirus Disease, by State of Reporting. (2017, September 13). Retrieved November 04, 2020, from https://www.cdc.gov/hantavirus/surveillance/reporting-state.html
- Hjertqvist, M., Klein, S. L., Ahlm, C., & Klingstrom, J. (2010). Mortality rate patterns for hemorrhagic fever with renal syndrome caused by Puumala virus. Emerging infectious diseases, 16(10), 1584–1586. https://doi.org/10.3201/eid1610.100242
- Sjölander, K. B., Elgh, F., Kallio-Kokko, H., Vapalahti, O., Hägglund, M., Palmcrantz, V., Juto, P., Vaheri, A., Niklasson, B., & Lundkvist, A. (1997). Evaluation of serological methods for diagnosis of Puumala hantavirus infection (nephropathia epidemica). Journal of clinical microbiology, 35(12), 3264–3268. https://doi.org/10.1128/JCM.35.12.3264-3268.1997
- Bharadwaj M, Nofchissey R, Goade D, Koster F, Hjelle B. Humoral immune responses in the hantavirus cardiopulmonary syndrome. J Infect Dis. 2000 Jul;182(1):43-8. doi: 10.1086/315657. Epub 2000 Jun 27. PMID: 10882580.
- Elgh F, Lundkvist A, Alexeyev OA, Stenlund H, Avsic-Zupanc T, Hjelle B, Lee HW, Smith KJ, Vainionpää R, Wiger D, Wadell G, Juto P. Serological diagnosis of hantavirus infections by an enzyme-linked immunosorbent assay based on detection of immunoglobulin G and M responses to recombinant nucleocapsid proteins of five viral serotypes. J Clin Microbiol. 1997 May;35(5):1122-30. doi: 10.1128/JCM.35.5.1122-1130.1997. PMID: 9114393; PMCID: PMC232715.
- Hjelle B, Jenison S, Torrez-Martinez N, Herring B, Quan S, Polito A, Pichuantes S, Yamada T, Morris C, Elgh F, Lee HW, Artsob H, Dinello R. Rapid and specific detection of Sin Nombre virus antibodies in patients with hantavirus pulmonary syndrome by a strip immunoblot assay suitable for field diagnosis. J Clin Microbiol. 1997 Mar;35(3):600-8. doi: 10.1128/JCM.35.3.600-608.1997. PMID: 9041397; PMCID: PMC229635.
- Sun Y, Chung DH, Chu YK, Jonsson CB, Parker WB. Activity of ribavirin against Hantaan virus correlates with production of ribavirin-5′-triphosphate, not with inhibition of IMP dehydrogenase. Antimicrob Agents Chemother. 2007 Jan;51(1):84-8. doi: 10.1128/AAC.00790-06. Epub 2006 Oct 23. PMID: 17060520; PMCID: PMC1797647.
- Huggins JW, Hsiang CM, Cosgriff TM, Guang MY, Smith JI, Wu ZO, LeDuc JW, Zheng ZM, Meegan JM, Wang QN, et al. Prospective, double-blind, concurrent, placebo-controlled clinical trial of intravenous ribavirin therapy of hemorrhagic fever with renal syndrome. J Infect Dis. 1991 Dec;164(6):1119-27. doi: 10.1093/infdis/164.6.1119. PMID: 1683355.
- Chapman LE, Mertz GJ, Peters CJ, Jolson HM, Khan AS, Ksiazek TG, Koster FT, Baum KF, Rollin PE, Pavia AT, Holman RC, Christenson JC, Rubin PJ, Behrman RE, Bell LJ, Simpson GL, Sadek RF. Intravenous ribavirin for hantavirus pulmonary syndrome: safety and tolerance during 1 year of open-label experience. Ribavirin Study Group. Antivir Ther. 1999;4(4):211-9. PMID: 10723500.
- Mertz GJ, Miedzinski L, Goade D, Pavia AT, Hjelle B, Hansbarger CO, Levy H, Koster FT, Baum K, Lindemulder A, Wang W, Riser L, Fernandez H, Whitley RJ; Collaborative Antiviral Study Group. Placebo-controlled, double-blind trial of intravenous ribavirin for the treatment of hantavirus cardiopulmonary syndrome in North America. Clin Infect Dis. 2004 Nov 1;39(9):1307-13. doi: 10.1086/425007. Epub 2004 Oct 11. PMID: 15494907.
- Rusnak JM, Byrne WR, Chung KN, Gibbs PH, Kim TT, Boudreau EF, Cosgriff T, Pittman P, Kim KY, Erlichman MS, Rezvani DF, Huggins JW. Experience with intravenous ribavirin in the treatment of hemorrhagic fever with renal syndrome in Korea. Antiviral Res. 2009 Jan;81(1):68-76. doi: 10.1016/j.antiviral.2008.09.007. Epub 2008 Oct 31. Erratum in: Antiviral Res. 2009 Jul;83(1):99-100. PMID: 18977392; PMCID: PMC7127354.
- Vial PA, Valdivieso F, Ferres M, Riquelme R, Rioseco ML, Calvo M, Castillo C, Díaz R, Scholz L, Cuiza A, Belmar E, Hernandez C, Martinez J, Lee SJ, Mertz GJ; Hantavirus Study Group in Chile. High-dose intravenous methylprednisolone for hantavirus cardiopulmonary syndrome in Chile: a double-blind, randomized controlled clinical trial. Clin Infect Dis. 2013 Oct;57(7):943-51. doi: 10.1093/cid/cit394. Epub 2013 Jun 19. PMID: 23784924; PMCID: PMC3765009.
- Crowley MR, Katz RW, Kessler R, Simpson SQ, Levy H, Hallin GW, Cappon J, Krahling JB, Wernly J. Successful treatment of adults with severe Hantavirus pulmonary syndrome with extracorporeal membrane oxygenation. Crit Care Med. 1998 Feb;26(2):409-14. doi: 10.1097/00003246-199802000-00047. Erratum in: Crit Care Med 1998 Apr;26(4):806. PMID: 9468181.
- Jorge A. Wernly, Charles A. Dietl, Cyril Etta Tabe, Stuart B. Pett, Cameron Crandall, Karen Milligan, Mark R. Crowley, Extracorporeal membrane oxygenation support improves survival of patients with Hantavirus cardiopulmonary syndrome refractory to medical treatment, European Journal of Cardio-Thoracic Surgery, Volume 40, Issue 6, December 2011, Pages 1334–1340, https://doi.org/10.1016/j.ejcts.2011.01.089
- Wernly JA, Dietl CA, Tabe CE, Pett SB, Crandall C, Milligan K, Crowley MR. Extracorporeal membrane oxygenation support improves survival of patients with Hantavirus cardiopulmonary syndrome refractory to medical treatment. Eur J Cardiothorac Surg. 2011 Dec;40(6):1334-40. doi: 10.1016/j.ejcts.2011.01.089. Epub 2011 Sep 6. PMID: 21900022.
- Terajima M, Hendershot JD 3rd, Kariwa H, Koster FT, Hjelle B, Goade D, DeFronzo MC, Ennis FA. High levels of viremia in patients with the Hantavirus pulmonary syndrome. J Infect Dis. 1999 Dec;180(6):2030-4. doi: 10.1086/315153. PMID: 10558964.
- Pergam SA, Schmidt DW, Nofchissey RA, Hunt WC, Harford AH, Goade DE. Potential renal sequelae in survivors of hantavirus cardiopulmonary syndrome. Am J Trop Med Hyg. 2009 Feb;80(2):279-85. PMID: 19190227; PMCID: PMC2706524.
