Staphylococcus aureus bacteremia is a cause of severe morbidity and mortality, especially when it is complicated by infective endocarditis. The mortality of Staphylococcus aureus bacteremia is 20% (1) and that of infective endocarditis due to Staph aureus is much higher. Guidelines for the treatment of MRSA SAB (2) recommends the use of echocardiography, with TEE being recommended over TTE due to its higher sensitivity. While this seems reasonable, given the fact that SAB tends to be one of the most common organisms causing infective endocarditis, there may be barriers to everyone being able to get a TEE and indeed, there may be an argument over the fact that not every patient needs one.
Modified Duke Criteria
Prior to proceeding, I think it may be reasonable to describe the modified Duke criteria for infective endocarditis, as this will give us some context going forward. This new criteria (3) was released in 2000 which found that TEE improved the utility of the modified Duke criteria in patients with negative TTE and intermediate pretest probability, but did not do much for those who had only one or 2 minor criteria. Possible IE definition was changed to have at least 3 minor or 1 major and 1 minor criteria.

A review (1) of 9 studies found that TEE had a higher sensitivity for IE (14-28%) when compared to TTE (2-15%), with TEE detecting IE in 15 out of 77 patients with negative TTE. In a cohort study of 103 patients (4) TEE was found to be more sensitive than TTE, the sensitivity of it plus Duke criteria being 32% (NPV 81%). Comparatively, the diagnosis of IE was established with TEE alone in 73%, yielding a sensitivity of 100% for TEE + Duke criteria. Another interesting study performed a Monter Carlo simulation on various echo strategies on 90 day survival amongst patients with SAB and <5% risk of IE (5). These strategies included TTE, TTE followed by TEE, TEE, or neither. Patients with high risk of TEE did not undergo the study and median 90 day survival benefit of any strategy compared to no echo was less than 0.5%:

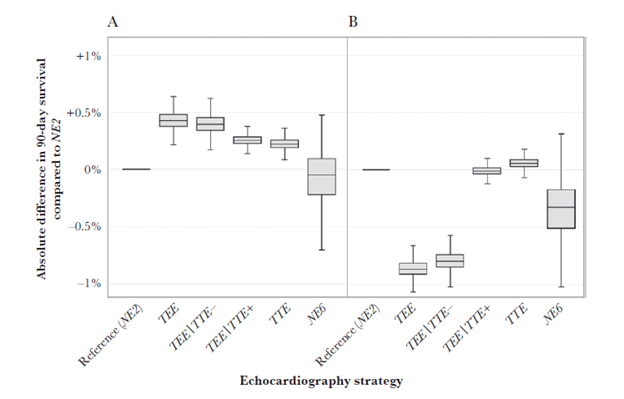
The TEE strategy still offered the best 90 day mortality but the absolute benefit compared to no echo and 2 weeks of therapy was only 0.12% (95% CI -0.02—0.3).

This suggests TEE did not have a significant influence in those who are at low risk of endocarditis. If this is the case, and if we can identify patients who are low risk, then we can probably avoid TEE in certain patients.
Risk Factors for Infective Endocarditis
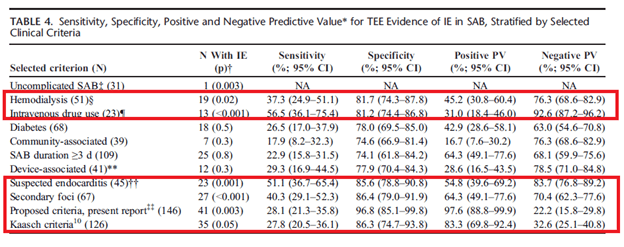
A prospective study (6) of 244 patients with SAB found that patients who were found to have an unknown source of SAB (38% vs 16%, p-value 0.001) and community-acquired infection (57% vs 34%, p-value 0.002) were more likely to have IE. Multivariate analysis found that embolic events (OR 5.73, 95% CI 1.66-19.79), having one or more pre-disposing conditions (IVDU, valvular disorder, prosthetic valves, cardiac device, OR 3.09, 95% CI 1.42-6.74, p-value 0.004), unknown source of SAB (OR 2.79, 95% CI 1.28-6.10, p-value 0.01) and heart murmur (OR 2.79, 95% CI 1.28-6.06) were independently associated with increased risk of IE. Further data (7) demonstrated that TTE and TEE found 7.8% and 23.7% of cases of IE respectively, in a cohort of 960 patients. Those who were IVDU, were on hemodialysis, had secondary foci of infection, and had complicated bacteremia were more likely to have a positive TEE:
How can we determine the low risk patients who do not require a TEE? Several criterial, similar to the NOVA and DENOVA criteria, have been proposed for this. Data analyzed post hoc form 2 prospective studies (8) with a total of 768 patients analyzed. Clinical prediction criteria evaluated included prolonged bacteremia (>4 days), presence of intracardiac device, hemodialysis dependency, spinal infection, and non-vertebral osteomyelitis. In one cohort, all patients with SAB IE fulfilled at least one criteria, and no cases with IE fulfill any criteria:
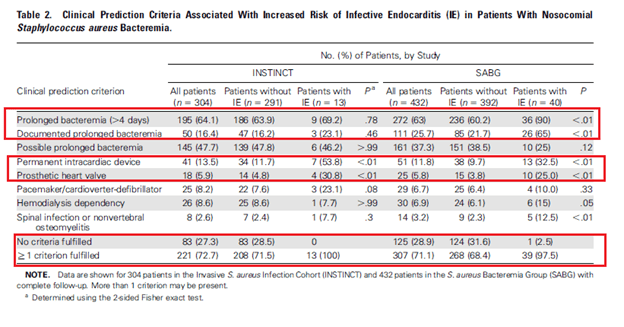
Notably, having just one risk factor was associated with nearly a 5% incidence of IE with having 2 or more risk factors increasing your risk to over 10%. Indeed, only using one risk yields a sensitivity of over 97% and has good NPV.

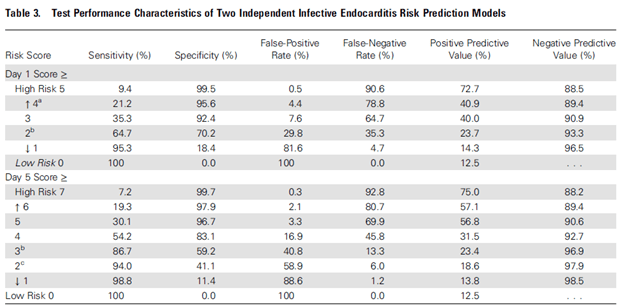
The PREDICT study (8) evaluated an algorithm that was internally validating using a bootstrap method. 678 patients with SAB were evaluated, and risk factors for IE included presence of a cardiac implantable device, and community onset of SAB, both at days 1 and 5:
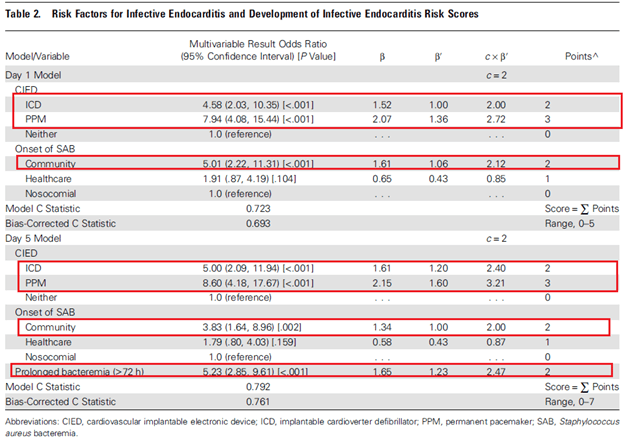
The authors proposed a cut-off score of 4 points on day 1 and 2 on day 5 to get a TEE to maximize NPV. Per the authors, this allows day 1 to identify high risk patients at the highest risk of IE:
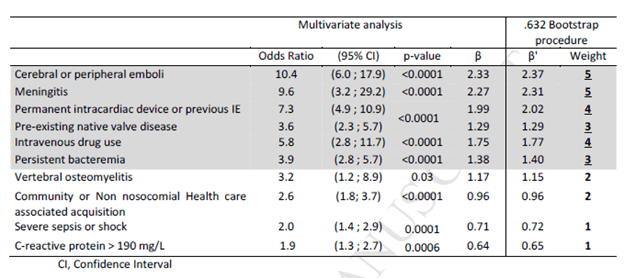
The most robust of these scoring systems has been the VRISTA scoring system (9). This was a multicenter observational prospective cohort study that evaluated predictive factors for IE in SAB. The model was validated using a bootstrap procedure with median beta coefficients rounded to the nearest half and multiplied by 2 to arrive to a point system. 1348 patients underwent TTE and 605 underwent TEE, with a rate of definitive endocarditis being 15.6% (n=211). Here, 10 predictive factors were found to be associated with definitive IE:
From here, a theoretical score from 0 to 30 was calculated, though in this cohort, the score ranged from 0 to 20. Not surprisingly, the rate of IE increased from 1.1% when the score was <2 to 17.4% when the score was >3 and 70.8% when the score was >10:
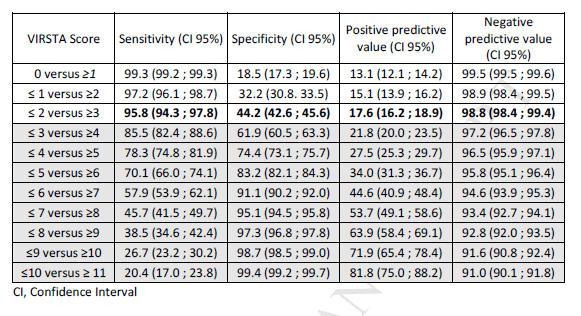
The TTE performance rate rose from 54.2% with a score <2 to 89.9% with a score of >10. Of course, these scores have not been externally validated, so their utility is still up in the air. A study evaluated the preferred echo modality in several hypothetical situations (10) and used the VRISTA score to guide these scenarios. Not surprisingly, those who had a VRISTA score >3 were more likely to have a strategy that included TEE:

What can we do this these data? While we cannot use a specific score (at least as of yet) we can actually risk stratify patients based on certain characteristics. First, onset of bacteremia. Outpatient onset is a risk factors, as is the presence of any implantable cardiac devices and to a certain extent, hemodialysis dependency. Further, prolonged bacteremia is also a risk factor for IE. If these criteria are met, it seems TEE can be safely avoided. A review (1) found patients who lacked these risk factors to have a low risk of IE. The NPV for these criteria ranged from 93 to 100%.
- Absence of permanent intracardiac device
- Sterile follow up blood cultures within 4 days after the initial set
- No Hemodialysis dependence
- Nosocomial acquisition
- Absence of secondary foci of infection
- No clinical signs of infective endocarditis (though you can also add initial TTE to this)
References:
- Holland TL, Arnold C, Fowler VG Jr. Clinical management of Staphylococcus aureus bacteremia: a review. JAMA. 2014 Oct 1;312(13):1330-41. doi: 10.1001/jama.2014.9743. PMID: 25268440; PMCID: PMC4263314.
- Liu C, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, Kaplan SL, Karchmer AW, Levine DP, Murray BE, J Rybak M, Talan DA, Chambers HF; Infectious Diseases Society of America. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011 Feb 1;52(3):e18-55. doi: 10.1093/cid/ciq146. Epub 2011 Jan 4. Erratum in: Clin Infect Dis. 2011 Aug 1;53(3):319. PMID: 21208910.
- Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG Jr, Ryan T, Bashore T, Corey GR. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000 Apr;30(4):633-8. doi: 10.1086/313753. Epub 2000 Apr 3. PMID: 10770721.
- Fowler VG Jr, Li J, Corey GR, Boley J, Marr KA, Gopal AK, Kong LK, Gottlieb G, Donovan CL, Sexton DJ, Ryan T. Role of echocardiography in evaluation of patients with Staphylococcus aureus bacteremia: experience in 103 patients. J Am Coll Cardiol. 1997 Oct;30(4):1072-8. doi: 10.1016/s0735-1097(97)00250-7. PMID: 9316542.
- Heriot GS, Tong SYC, Cheng AC, Liew D. Benefit of Echocardiography in Patients With Staphylococcus aureus Bacteremia at Low Risk of Endocarditis. Open Forum Infect Dis. 2018 Dec 11;5(12):ofy303. doi: 10.1093/ofid/ofy303. PMID: 30555848; PMCID: PMC6288770.
- Rasmussen RV, Høst U, Arpi M, Hassager C, Johansen HK, Korup E, Schønheyder HC, Berning J, Gill S, Rosenvinge FS, Fowler VG Jr, Møller JE, Skov RL, Larsen CT, Hansen TF, Mard S, Smit J, Andersen PS, Bruun NE. Prevalence of infective endocarditis in patients with Staphylococcus aureus bacteraemia: the value of screening with echocardiography. Eur J Echocardiogr. 2011 Jun;12(6):414-20. doi: 10.1093/ejechocard/jer023. PMID: 21685200; PMCID: PMC3117467.
- Kaasch AJ, Fowler VG Jr, Rieg S, Peyerl-Hoffmann G, Birkholz H, Hellmich M, Kern WV, Seifert H. Use of a simple criteria set for guiding echocardiography in nosocomial Staphylococcus aureus bacteremia. Clin Infect Dis. 2011 Jul 1;53(1):1-9. doi: 10.1093/cid/cir320. PMID: 21653295; PMCID: PMC3149212.
- Palraj BR, Baddour LM, Hess EP, Steckelberg JM, Wilson WR, Lahr BD, Sohail MR. Predicting Risk of Endocarditis Using a Clinical Tool (PREDICT): Scoring System to Guide Use of Echocardiography in the Management of Staphylococcus aureus Bacteremia. Clin Infect Dis. 2015 Jul 1;61(1):18-28. doi: 10.1093/cid/civ235. Epub 2015 Mar 25. PMID: 25810284; PMCID: PMC4542912.
- Tubiana S, Duval X, Alla F, Selton-Suty C, Tattevin P, Delahaye F, Piroth L, Chirouze C, Lavigne JP, Erpelding ML, Hoen B, Vandenesch F, Iung B, Le Moing V; VIRSTA/AEPEI Study Group. The VIRSTA score, a prediction score to estimate risk of infective endocarditis and determine priority for echocardiography in patients with Staphylococcus aureus bacteremia. J Infect. 2016 May;72(5):544-53. doi: 10.1016/j.jinf.2016.02.003. Epub 2016 Feb 22. PMID: 26916042.
- Heriot, G. S., Tong, S., Cheng, A. C., & Liew, D. (2020). A Scenario-Based Survey of Expert Echocardiography Recommendations for Patients With Staphylococcus aureus Bacteremia at Varying Risk for Endocarditis. JAMA network open, 3(4), e202401. https://doi.org/10.1001/jamanetworkopen.2020.2401
