Gram negative bacteremia tends to complicate a variety of infections, including urinary tract infections/pyelonephritis, and intra-abdominal infections. Further, a lot of these patients tend to have severe presentations leading to perhaps overtreatment with IV antibiotic therapy. Until recently, there had been little data to guide uncomplicated gram negative rod bacteremia but there has been a trend towards reducing the length of therapy as well early transitioning to PO antibiotic therapy. While this is a safe and effective strategy, it should be noted these strategies do not apply to:
- Immunosuppressed patients (i.e. SOT or HSCT)
- Prosthetic material (i.e. prosthetic joint, valves, lines)
- No source control
Which, I guess, should be pretty obvious.
Length of Therapy
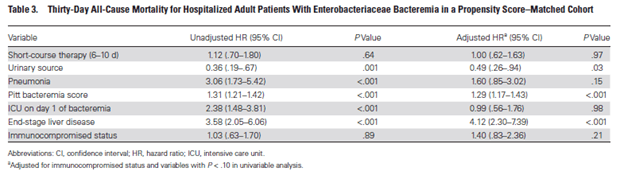
A 2018 retrospective study (1) evaluated short course (6-10 days) vs long course (11-16 days) of antibiotic therapy for gram negative bacteremia (Enterobacteriacae). Primary outcome was 30 day mortality. 1769 patients met eligibility criteria and 385 propensity score-matched patients were placed in each group. After propensity score matching, there were 37 (9.6%) deaths in the short-course group and 39 (10.1%) deaths in the long course group (HR 1.12, 95% CI 0.7-1.80), suggesting no difference in mortality between each treatment strategies. Further, there was no reduction in mortality for each additional antibiotic therapy for the 30 days after antibiotics were discontinued (HR 0.97, 95% CI 0.91-1.05). Factors associated with increased mortality included higher Pitt bacteremia score, ICU day admission, and ESLD (so, if you’re sick, you’re sick):
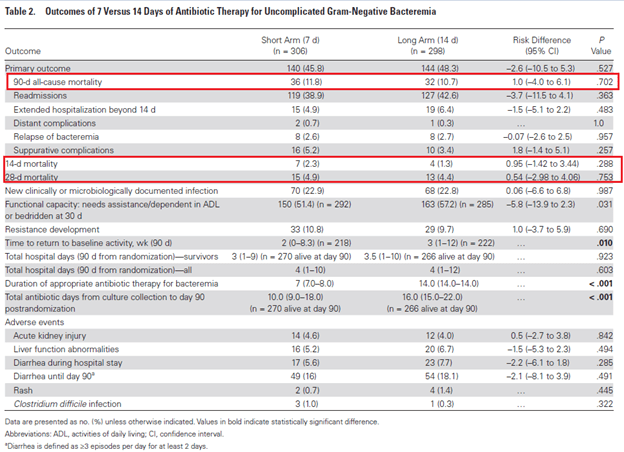
A non-inferiority open-label RCT (2) evaluated 7 days vs 14 days of antibiotic therapy if patients were hemodynamically stable and afebrile for 48 hours prior to randomization. Primary outcome was composite of all-caue mortality, clinical failure, local or distal complications, readmissions, and extended hospital stay at 90 days. 604 patients were included, and primary outcome occurred in 140/306 (45.8%) in the short-duration group vs 144/298 (48.3%) in the long-duration group (RD -2.6%, -10.5% to 5.3%). Further, there was no difference between each group in 14 day mortality of 28 day mortality:
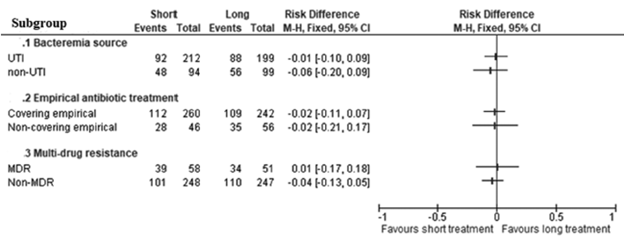
There were no differences in the primary outcomes in terms of the bacteremic source or MDR status:
Overall, it seems reasonable to proceed with a 7 day course of therapy overall for uncomplicated GNR bacteremia.
PO Stepdown
De-escalation is a common stewardship tool to avoid issues such as drug toxicity, cost, and the possibility of breeding multi-drug resistant organisms. A post-hoc analysis of a prospective study evaluated bacteremias due to enterobacteriacae (3). Patients were evaluated in terms of early de-escalation (within 4 days), late de-escalation (days 5-7) or no de-escalation and main outcome was 30 day all cause mortality. 516 patients were evaluated, with 241 receiving early de-escalation, 95 receiving late de-escalation, and 180 without any de-escalation. Those who did not receive de-escalation were more likely to have MDRO (including ESBL-producing organisms), have higher SOFA score, have nosocomial acquisition. Mortality rates were 4.1% in the early de-escalation group, 6.3% in the late de-escalation group, and 9.4% in the no de-escalation group. Notably, factors associated with de-escalation in this cohort included MDRO and nosocomial infection:
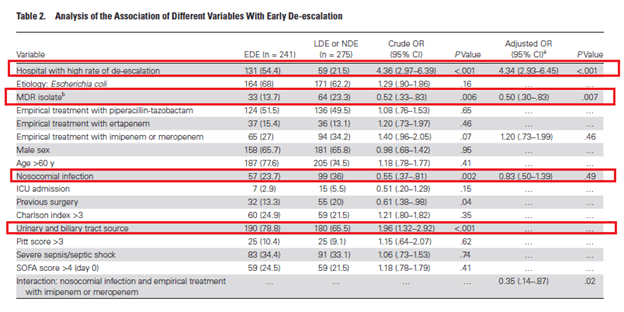
De-escalation was not associated with 30d mortality either in an adjusted multivariate analysis:
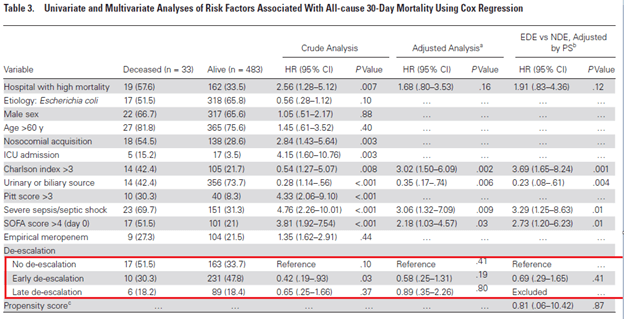
This suggest that in the right setting, de-escalation is a safe measure. In terms of oral stepdown for GNR, more data has been recently coming out in support of this strategy. The largest multicenter retrospective cohort (4) evaluated a PO stepdown strategy for Enterobacteriacae bloodstream infections in 2161 patients, with primary endpoint being 30d all cause mortality and 30d recurrent BSI. Patients that were included were non-complicated GNR bacteremias (i.e. removal of infected hardware, source control, resolution of biliary/urinary sources). PO stepdown were stratified based on bioavailability as well. Overall, there were 97 deaths (13.1%) in the PO stepdown group vs 99 (13.4%) in the IV group (HR 1.03, 95% CI 0.82-1.30), and 6 episodes of recurrent bacteremia in the PO group vs 4 in the IV group (HR 0.82 95% CI 0.33-2.01).
Most of the patients received one of the high bioavailable antibiotics (83.5%), either a FQ or TMP-SMX
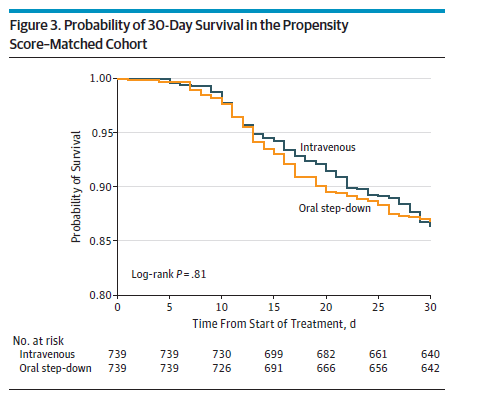
While this is a retrospective study, propensitiy matching was done so this lends some evidence towards use of PO antibiotics. The other issue, however, is the low percentage of PO beta-lactams that were used in this study (roughly 16%), which tends to be a poorly bioavailable antibiotic. Of course, the issue here is the fact that FQs tend to have significant side effects, especially in the elderly (AAA dissection and retinal detachment, anyone?). So how good are the different types of PO antibiotics? There are several studies on this issue. For instance, one retrospective study (5) evaluated the use of PO TMP-SMX, FQ, and Beta lactams as step down therapy for uncomplicated gram negative bacteremia. Primary endpoint was the composite rate of treatment failure (mortality within 30 days, recurrent bacteremia, or transition back to IV therapy). In 204 patients, composite endpoint occurred in 15 patients total (7.4%) with no difference between treatment arms (p-value 0.523). Notably, most patients received a FQ (66.7%), followed by a beta lactam (28.3%).
In another retrospective study evaluated PO FQ and beta-lactams as step-down therapy (6) in 224 patients. Primary outcome was clinical success, which was achieved in 86.9% of patients in the beta-lactam group vs 87.1% in patients in the FQ group, mean difference 0.2, 97.5% CI -10.3 to 10.7). All other clinical outcomes did not reach statistical significance for difference:
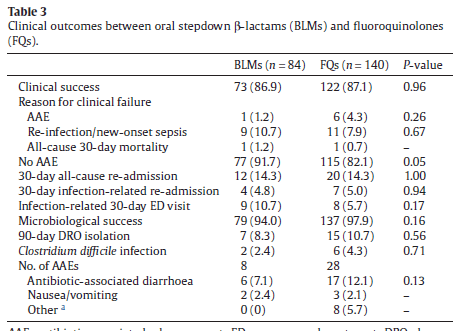
It should be said there were some imbalances between groups. For instance, those in the beta-lactam groups were more likely to have a transplanted organ, or have diabetes with complications. A meta-analysis evaluated patient outcomes in patients using either FQ/TMP-SMX or beta-lactams as step-down therapy (7). Eight studies were included, with most being single-center studies and all being retrospective cohorts. Of 2289 patients, 1489 (65%) received FQ, 177 (7.7%) received TMP-SMX, and 623 (27.2%) received beta-lactams. Duration of total antibiotics ranged from 13.6 to 16 days. 85 of 1666 (5.1%) that were transitioned to FQ or TMP-SMX and 22 of 623 (3.5%) of those transitioned to beta-lactam died from any cause, with no difference in odds of outcome in either group (OR 1.13, 95% CI 0.69-1.87). Notably, the beta-lactam group had higher recurrences (33 patients or 1.98% in the FQ/TMP-SMX group and 34 of 623 in the beta-lactam group, 5.46%, OR 2.06, 95% CI 1.18-3.61, p-value 0.01). There was no difference in recurrent bacteremia, however.
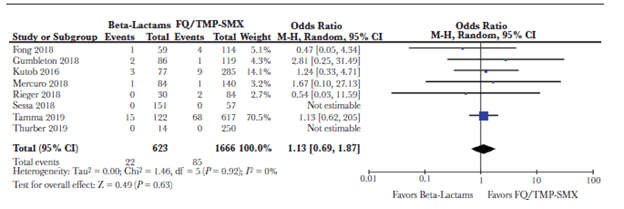
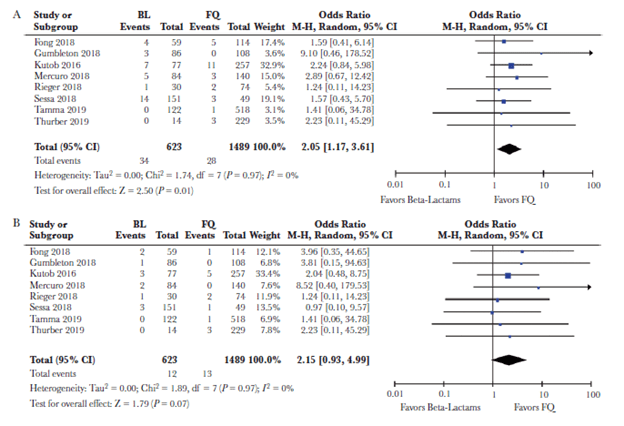
I do not think these data should be terribly surprising. In general, I do not think bacteremias (outside of Staph aureus and Candida) that are not complicated should change the course of therapy. As long as there is no source that needs to be drained (i.e. prosthetic joints or abscess), a short course of antibiotics should suffice. Look at the HOW-LONG study, which showed there was no difference in outcomes between short course and longer course of antibiotics as long as the source was taken care of (8). While the data is not strong, I think it is reasonable to pursue a PO beta-lactam course as well. Having said that, I would not blame anyone who would use FQ or TMP-SMX in that scenario, though their side effect profile leaves a lot to be desired.
TL:DR
- A short course of antibiotics (i.e. 7 days) is reasonable if there is no complication (i.e. metastatic complications, no abscess, no prosthetic infections)
- Early de-escalation is a reasonable approach
- PO step-down is also reasonable as long as the above criteria are met. Also, as far as I am concerned, FQ = TMP-SMX = Beta-lactams
References:
- Chotiprasitsakul D, Han JH, Cosgrove SE, et al. Comparing the Outcomes of Adults With Enterobacteriaceae Bacteremia Receiving Short-Course Versus Prolonged-Course Antibiotic Therapy in a Multicenter, Propensity Score-Matched Cohort. Clin Infect Dis. 2018;66(2):172-177. doi:10.1093/cid/cix767
- Yahav D, Franceschini E, Koppel F, et al. Seven Versus 14 Days of Antibiotic Therapy for Uncomplicated Gram-negative Bacteremia: A Noninferiority Randomized Controlled Trial. Clin Infect Dis. 2019;69(7):1091-1098. doi:10.1093/cid/ciy1054
- Palacios-Baena ZR, Delgado-Valverde M, Valiente Méndez A, et al. Impact of De-escalation on Prognosis of Patients With Bacteremia due to Enterobacteriaceae: A Post Hoc Analysis From a Multicenter Prospective Cohort. Clin Infect Dis. 2019;69(6):956-962. doi:10.1093/cid/ciy1032
- Tamma PD, Conley AT, Cosgrove SE, et al. Association of 30-Day Mortality With Oral Step-Down vs Continued Intravenous Therapy in Patients Hospitalized With Enterobacteriaceae Bacteremia [published correction appears in JAMA Intern Med. 2019 Nov 1;179(11):1607]. JAMA Intern Med. 2019;179(3):316-323. doi:10.1001/jamainternmed.2018.6226
- Nisly SA, McClain DL, Fillius AG, Davis KA. Oral antibiotics for the treatment of Gram-negative bloodstream infections: A retrospective comparison of three antibiotic classes. J Glob Antimicrob Resist. 2020;20:74-77. doi:10.1016/j.jgar.2019.07.026
- Mercuro NJ, Stogsdill P, Wungwattana M. Retrospective analysis comparing oral stepdown therapy for enterobacteriaceae bloodstream infections: fluoroquinolones versus β-lactams. Int J Antimicrob Agents. 2018;51(5):687-692. doi:10.1016/j.ijantimicag.2017.12.007
- Punjabi C, Tien V, Meng L, Deresinski S, Holubar M. Oral Fluoroquinolone or Trimethoprim-sulfamethoxazole vs. ß-lactams as Step-Down Therapy for Enterobacteriaceae Bacteremia: Systematic Review and Meta-analysis [published online ahead of print, 2019 Aug 14]. Open Forum Infect Dis. 2019;6(10):ofz364. doi:10.1093/ofid/ofz364
- Sawyer RG, Claridge JA, Nathens AB, et al. Trial of short-course antimicrobial therapy for intraabdominal infection [published correction appears in N Engl J Med. 2018 Jan 25;:null]. N Engl J Med. 2015;372(21):1996-2005. doi:10.1056/NEJMoa1411162
