“A term that has been used in a number of systems of classification is variola vera. It is this idea of true or typical smallpox so often forming the subject of illustrations in text-books of medicine that has fixed in the minds of practitioners a single picture of what smallpox should look like. The literature on smallpox abounds with statements that a case, particularly the first in an outbreak, was” atypical” as it did not fit the clinician’s preconceived ideas on the subject. It must be emphasized that there is no such thing as atypical smallpox. The wide range of severity and variation in signs and symptoms is characteristic of the disease, and depends on the reaction of the host to the virus attack. It is therefore misleading to describe cases as typical or “classical” smallpox, sometimes calling it variola vera, and regard all other forms as unusual variations (1).”
A member of the Poxvaridea family of double-stranded DNA viruses and related to variola (smallpox), it seems Monkey pox is moving out of its endemic area to terrorize the world (2). At least it feels like it. Two clades have been identified; the Central African and the West African clade. While not terribly important, the Central African clade, also known as the Congo-Basin clade, seems to have a higher degree of transmissibility and mortality. One difference between monkeypox and smallpox is that the latter did not have an animal reservoir. Indeed, monkeypox requires transmission from an infected animal to a human via scratches or bites, and even handling of infected animal products:

In the past, most of the disease was limited to Central and West African countries (figures, given the clade names), with special notes on Cameroon, Central African Republic, the DRC, Nigeria, Sierra Leone, Gabon, and Cote d’Ivoire. Cases outside of these areas are usually imported (see later). The classic presentation of monkeypox is that of a diffuse rash that starts in the face and spreads to the rest of the body following a prodromal period of fevers and malaise. Typically, the rash evolves from a macule and then progresses towards a papule, vesicles, pustules, and scabs with all marks typically present in the same stage (2):

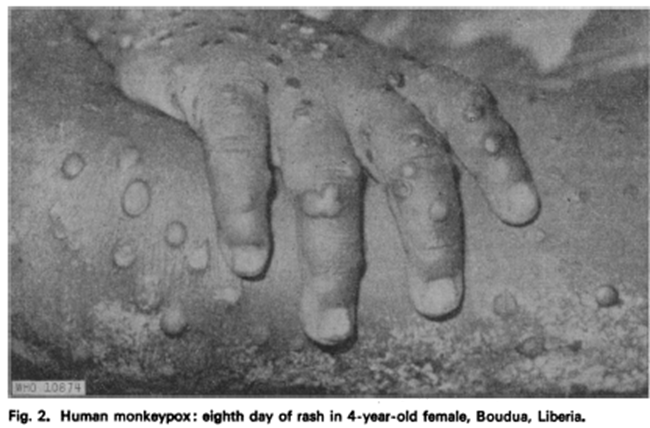
When one thinks of monkeypox, this is the picture they think of (3):

As you may know, the current outbreak of monkeypox looks nothing like that. As implied by the quote above, there may not be such thing as an “atypical” case of monkey pox. Similarly to monkeypox, there is no “typical” or “atypical” case of smallpox. Reading “Smallpox” by professor Dixon shows that the classic textbook presentation of the disease is quite unusual (1). Indeed, the presentation can range from a hyper-acute smallpox, reminiscent of sepsis with a maculopapular rash on the face, arms, and trunk, petechial rash that is similar to that of rubella, or a morbiliform rash.
The “Classics”:
Since the 1970s, the number of cases of monkeypox worldwide has increased, however the case fatality rate is not significantly high (4)


A case series of 6 patients described the lesions as discrete with rare coalescence, deep-seated, and present on the palms and soles of feet (5).
One of the first large, scale retrospective cohorts involved 47 patients reported from 1970 through 1979 (6). The authors described a period of 2-4 days of prodromal of fevers and prostration, prior to diffuse lesions that develop more or less simultaneously and evolve over a period of 2-4 weeks, favoring the periphery:

In this cohort, at least 4 patients may have been infected via secondary transmission, suggesting perhaps this mode of transmission has been around since its discovery, though the evidence wasn’t conclusive:

A retrospective study from Zaire that included 282 patients found what we would call the “classic” presentation of monkey pox, with most lesions being monomorphic, mostly in the face (7). These lesions ran through stages of macules, papules, vesicles, and pustules before umbilicating, drying, and desquamating.
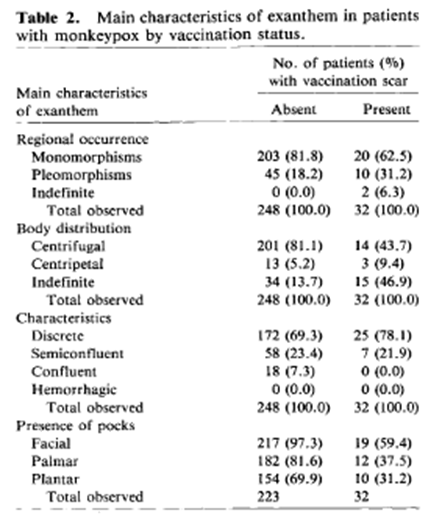
Course of illness was around two to four weeks, with scabs falling off by day 22-24 after onset of illness. Patients who were found to have a visible smallpox vaccination scar tended to have milder disease and no deaths compared to a mortality rate of 11% in non-vaccinated patients. More recent descriptions have been quite similar to these. For instance, an outbreak of 37 patients from the US (8) that was associated with direct contact with prairie dogs found that the median time from exposure to illness was around 12 days (range 11-18 days). 97% of patients had a rash, with fever (85%), chills (71%), adenopathy (71%), headache (65%), and myalgias (56%) with most patients presenting with fever and a rash:

Notably, 9.4% of patients had a rash in their groin/buttocks. Moreover, while most had a monomorphic rash, 29% had a pleomorphic rash:
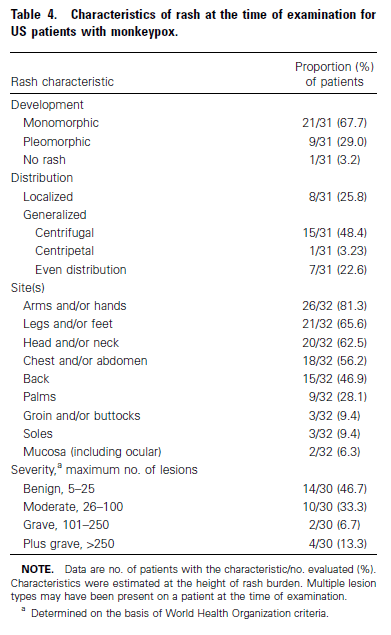
Notably, a fairly significant number of patients were found to have a rash in their groin, and not all rashes were in the same stage of evolution, suggesting monkeypox can present as a spectrum.
An epidemiological study of 276 suspected cases found that of the 122 confirmed cases, 36 had some contact with people with similar lesions (9). 12 of those were a confirmed case. Time from first contact to disease ranged from 3 days to 34 days. Of these confirmed cases, all had a vesiculopustular rash, and 88% had fever followed by headache (79%), pruritus (73%), lymphadenopathy (69%), myalgia (63%), and fever preceded rash only in 57% of cases. Rash was present in the face (96%), legs (91%), trunk (80%), arms (79%), palms (69%), soles (64%) and genitalia (68%). This suggests again the role of vertical transmission in monkeypox, reminiscent of the current outbreak. Moreover, a cohort of 419 patients from the DRC found that only 22% cases were primary, while the remainder was secondary, of which 53% reported having contact with another patient (10). One case describes a man who was diagnosed with monkey pox in 2018 following travel. One health-care worker who had exposure to the patient became ill, and subsequently 4 contacts of the healthcare worker also came down with monkey pox (11) Finally, a retrospective cohort from Nigeria found that 23/40 patients (65.7%) had a rash prior to fever, with 67.5% of rashes being observed in genitalia though 98% were in the face (12). Moreover, 63% of rashes were monomorphic while 37.5% were pleomorphic. One interesting bit was that all patients who had concomitant HIV infection had genital ulcers from monkey pox. The importance of this will be clear soon.
The New Outbreak:
The early descriptions of the new outbreak came from a handful of European reports. One Italian report described 4 cases of Monkey pox in 4 MSMs, who had localization of skin lesions in their genital and anal area, as well as an asynchronous evolution of symptoms (13). One of the earlier case reports came from London, UK, who reviewed 54 patients who presented to a sexual health center (14). All patients were male and identified themselves as MSM. A few things to note: the median time from symptom onset to testing was usually around 7 days, with median time from last sexual contact to symptom onset being around 2 days. All had skin lesions, which were described as pustular papules with a central umbilicated dip, fluid-filled vesicles, ulcerations, or eschars. 81% of patients had an associated symptom (fever, fatigue, lethargy, myalgia) prior to or at around the time of the assessment or the previous 2 weeks. Skin lesions had variable morphology even within one clinical site, however in 4 patients the rash was described as erythematous or confluent maculo-papular rash that spared the face.

A larger outbreak from Germany involved 260 cases, of which 259 were identified as MSM. 79% of these cases did not have any international travel, and 58% reported having visited clubs, bars, or private parties during their assumed infection period (15). Given the above, it seems this outbreak is behaving more like an STI, similar to syphilis or HSV. This is highlighted by a descriptive case series of nearly 200 patients out of London, UK who were confirmed to have monkeypox via PCR (16). All had some degree of mucocutaneous manifestation, with lesions most commonly being found in the genitals (56%), and perianal area (41%), one or both occurring in 88% of all cases. Only 4.1% of these patients had more than 100 lesions. It should be noted that not many had diffuse body-rash and the characteristics were quite polymorphic:


As highlighted above, many other symptoms, including rectal pain or pain on defecation, were also fairly common. Fever, lymphadenopathy, and myalgias were also present however only 61% had the fever and adenopathy prior to the appearance of the rash. It was not uncommon for this cohort to have concomitant STIs, including HSV, syphilis, and gonorrhea. A total of 56 patients (31% of the cohort) had another STI.

A multicenter observational cohort from Spain assessed a total of 181 patients with monkey pox of which 40% were HIV-positive (17). Median incubation period was found to be 7 days and all were found to have skin lesions, with 78% having lesions in their anogenital region:
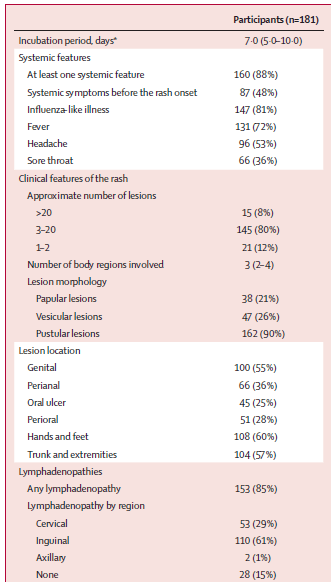
It should be noted that the vast majority of patients in this cohort had less than 20 lesions at the time of presentation. Moreover, 70% of patients who had throat swab and 78% of patients who had an anal swab were positive by PCR.

The largest multicenter observational study of 528 cases found that roughly 98% of cases were either gay or bisexual men (18). Of these, 41% were HIV-positive, with most of the skin lesions being in the anogenital area (73%), trunk, arms, or legs (55%), face (25%), and palms/soles (10%). Moreover, there was a wide spectrum of skin lesions ranging from macular rashes to crusted lesions, with multiple phases being present in patients. Interestingly 54 patients presented with a single genital ulcer and 26 had oropharyngeal symptoms.

Exposure events included travel abroad (28%) and sex-on-site venues (32%), however the authors were unable to confirm sexual transmission.
So it is behaving like an STI, where MSM are at higher risk of obtaining it. Compared to prior outbreaks which were limited to Central and Western Africa and were mostly associated with animal exposure, there seems to be more of a human transmission component. Indeed, in prior outbreaks there were a small number of those being from human exposure. There may be an evolutionary component (19). A genome sequencing of monkey pox demonstrates the 2022 outbreak is part of a clade that has been previously sampled in Nigeria, or in those with a travel history to Nigeria. While outside of the scope of this post (and mostly because I do not want to relive genetics), the genome from the 2022 cases were compared with those obtained from outbreaks in Singapore, Israel, Nigeria, and UK (the closest genome) and found 47 single nucleotide difference. Given the estimated 1-2 nucleotide changes per year in the small pox virus, this is a lot of changes. Researchers have identified the deaminase APOBEC3 as being potentially responsible. Given the rise in human-to-human transmission, it may be this deaminase is facilitating this type of transmission i.e is this something seen in animals and is now in humans, or is the APOBEC3 a activity the virus adapting to humans, thus facilitating transmission. The conclusion of the authors:
“We therefore suggest that the pattern we see in these MPXV genomes since 2017 is indicative of replication in humans and the inheritance of the specific changes that occurred between 2017 and 2018 and then in the viruses from 2022 means that there has been sustained human to human transmission since at least 2017. The 10 mutations that do fit the APOBEC3 profile may represent an additional period of human to human transmission prior to the 2017 cases.”
Add monkeypox to the list of swabs for STI or rashes.
References:
- Dixon, C. W. (1962). Smallpox. London; J. & A. Churchill.
- Titanji BK, Tegomoh B, Nematollahi S, Konomos M, Kulkarni PA. Monkeypox: A Contemporary Review for Healthcare Professionals. Open Forum Infect Dis. 2022 Jun 23;9(7):ofac310. doi: 10.1093/ofid/ofac310. PMID: 35891689; PMCID: PMC9307103.
- Adebayo, Oladimeji & Owoeye, David. (2017). Monkeypox. Annals of Ibadan Postgraduate Medicine. 15. 145-148.
- Bunge EM, Hoet B, Chen L, Lienert F, Weidenthaler H, Baer LR, Steffen R. The changing epidemiology of human monkeypox-A potential threat? A systematic review. PLoS Negl Trop Dis. 2022 Feb 11;16(2):e0010141. doi: 10.1371/journal.pntd.0010141. PMID: 35148313; PMCID: PMC8870502.
- Foster SO, Brink EW, Hutchins DL, Pifer JM, Lourie B, Moser CR, Cummings EC, Kuteyi OE, Eke RE, Titus JB, Smith EA, Hicks JW, Foege WH. Human monkeypox. Bull World Health Organ. 1972;46(5):569-76. PMID: 4340216; PMCID: PMC2480784.
- Breman JG, Kalisa-Ruti, Steniowski MV, Zanotto E, Gromyko AI, Arita I. Human monkeypox, 1970-79. Bull World Health Organ. 1980;58(2):165-82. PMID: 6249508; PMCID: PMC2395797.
- Jezek Z, Szczeniowski M, Paluku KM, Mutombo M. Human monkeypox: clinical features of 282 patients. J Infect Dis. 1987 Aug;156(2):293-8. doi: 10.1093/infdis/156.2.293. PMID: 3036967.
- Huhn GD, Bauer AM, Yorita K, Graham MB, Sejvar J, Likos A, Damon IK, Reynolds MG, Kuehnert MJ. Clinical characteristics of human monkeypox, and risk factors for severe disease. Clin Infect Dis. 2005 Dec 15;41(12):1742-51. doi: 10.1086/498115. Epub 2005 Nov 11. PMID: 16288398.
- Yinka-Ogunleye A, Aruna O, Dalhat M, Ogoina D, McCollum A, Disu Y, Mamadu I, Akinpelu A, Ahmad A, Burga J, Ndoreraho A, Nkunzimana E, Manneh L, Mohammed A, Adeoye O, Tom-Aba D, Silenou B, Ipadeola O, Saleh M, Adeyemo A, Nwadiutor I, Aworabhi N, Uke P, John D, Wakama P, Reynolds M, Mauldin MR, Doty J, Wilkins K, Musa J, Khalakdina A, Adedeji A, Mba N, Ojo O, Krause G, Ihekweazu C; CDC Monkeypox Outbreak Team. Outbreak of human monkeypox in Nigeria in 2017-18: a clinical and epidemiological report. Lancet Infect Dis. 2019 Aug;19(8):872-879. doi: 10.1016/S1473-3099(19)30294-4. Epub 2019 Jul 5. PMID: 31285143.
- Centers for Disease Control and Prevention (CDC). Human monkeypox — Kasai Oriental, Democratic Republic of Congo, February 1996-October 1997. MMWR Morb Mortal Wkly Rep. 1997 Dec 12;46(49):1168-71. PMID: 9408046.
- Ogoina D, Iroezindu M, James HI, Oladokun R, Yinka-Ogunleye A, Wakama P, Otike-Odibi B, Usman LM, Obazee E, Aruna O, Ihekweazu C. Clinical Course and Outcome of Human Monkeypox in Nigeria. Clin Infect Dis. 2020 Nov 5;71(8):e210-e214. doi: 10.1093/cid/ciaa143. PMID: 32052029.
- Vaughan A, Aarons E, Astbury J, Brooks T, Chand M, Flegg P, Hardman A, Harper N, Jarvis R, Mawdsley S, McGivern M, Morgan D, Morris G, Nixon G, O’Connor C, Palmer R, Phin N, Price DA, Russell K, Said B, Schmid ML, Vivancos R, Walsh A, Welfare W, Wilburn J, Dunning J. Human-to-Human Transmission of Monkeypox Virus, United Kingdom, October 2018. Emerg Infect Dis. 2020 Apr;26(4):782-785. doi: 10.3201/eid2604.191164. Epub 2020 Apr 17. PMID: 32023204; PMCID: PMC7101111.
- Antinori A, Mazzotta V, Vita S, Carletti F, Tacconi D, Lapini LE, D’Abramo A, Cicalini S, Lapa D, Pittalis S, Puro V, Rivano Capparuccia M, Giombini E, Gruber CEM, Garbuglia AR, Marani A, Vairo F, Girardi E, Vaia F, Nicastri E; INMI Monkeypox Group. Epidemiological, clinical and virological characteristics of four cases of monkeypox support transmission through sexual contact, Italy, May 2022. Euro Surveill. 2022 Jun;27(22):2200421. doi: 10.2807/1560-7917.ES.2022.27.22.2200421. PMID: 35656836; PMCID: PMC9164671.
- Girometti N, Byrne R, Bracchi M, Heskin J, McOwan A, Tittle V, Gedela K, Scott C, Patel S, Gohil J, Nugent D, Suchak T, Dickinson M, Feeney M, Mora-Peris B, Stegmann K, Plaha K, Davies G, Moore LSP, Mughal N, Asboe D, Boffito M, Jones R, Whitlock G. Demographic and clinical characteristics of confirmed human monkeypox virus cases in individuals attending a sexual health centre in London, UK: an observational analysis. Lancet Infect Dis. 2022 Sep;22(9):1321-1328. doi: 10.1016/S1473-3099(22)00411-X. Epub 2022 Jul 1. PMID: 35785793.
- Selb R, Werber D, Falkenhorst G, Steffen G, Lachmann R, Ruscher C, McFarland S, Bartel A, Hemmers L, Koppe U, Stark K, Bremer V, Jansen K; Berlin MPX study group. A shift from travel-associated cases to autochthonous transmission with Berlin as epicentre of the monkeypox outbreak in Germany, May to June 2022. Euro Surveill. 2022 Jul;27(27):2200499. doi: 10.2807/1560-7917.ES.2022.27.27.2200499. PMID: 35801518; PMCID: PMC9264732.
- Patel A, Bilinska J, Tam JCH, Da Silva Fontoura D, Mason CY, Daunt A, Snell LB, Murphy J, Potter J, Tuudah C, Sundramoorthi R, Abeywickrema M, Pley C, Naidu V, Nebbia G, Aarons E, Botgros A, Douthwaite ST, van Nispen Tot Pannerden C, Winslow H, Brown A, Chilton D, Nori A. Clinical features and novel presentations of human monkeypox in a central London centre during the 2022 outbreak: descriptive case series. BMJ. 2022 Jul 28;378:e072410. doi: 10.1136/bmj-2022-072410. PMID: 35902115; PMCID: PMC9331915.
- Tarín-Vicente EJ, Alemany A, Agud-Dios M, Ubals M, Suñer C, Antón A, Arando M, Arroyo-Andrés J, Calderón-Lozano L, Casañ C, Cabrera JM, Coll P, Descalzo V, Folgueira MD, García-Pérez JN, Gil-Cruz E, González-Rodríguez B, Gutiérrez-Collar C, Hernández-Rodríguez Á, López-Roa P, de Los Ángeles Meléndez M, Montero-Menárguez J, Muñoz-Gallego I, Palencia-Pérez SI, Paredes R, Pérez-Rivilla A, Piñana M, Prat N, Ramirez A, Rivero Á, Rubio-Muñiz CA, Vall M, Acosta-Velásquez KS, Wang A, Galván-Casas C, Marks M, Ortiz-Romero PL, Mitjà O. Clinical presentation and virological assessment of confirmed human monkeypox virus cases in Spain: a prospective observational cohort study. Lancet. 2022 Aug 27;400(10353):661-669. doi: 10.1016/S0140-6736(22)01436-2. Epub 2022 Aug 8. PMID: 35952705.
- Thornhill JP, Barkati S, Walmsley S, Rockstroh J, Antinori A, Harrison LB, Palich R, Nori A, Reeves I, Habibi MS, Apea V, Boesecke C, Vandekerckhove L, Yakubovsky M, Sendagorta E, Blanco JL, Florence E, Moschese D, Maltez FM, Goorhuis A, Pourcher V, Migaud P, Noe S, Pintado C, Maggi F, Hansen AE, Hoffmann C, Lezama JI, Mussini C, Cattelan A, Makofane K, Tan D, Nozza S, Nemeth J, Klein MB, Orkin CM; SHARE-net Clinical Group. Monkeypox Virus Infection in Humans across 16 Countries – April-June 2022. N Engl J Med. 2022 Aug 25;387(8):679-691. doi: 10.1056/NEJMoa2207323. Epub 2022 Jul 21. PMID: 35866746.
- O’Toole A, Rambaut A. Initial observations about putative APOBEC3 deaminase editing driving short-term evolution of MPXV since 2017. 2022 (https://virological.org/t/initial-observations-about-putative-apobec3-deaminase-editing-driving-short-term-evolution-of-mpxv-since-2017/830)
