Leptospirosis is related to the other spirochetes such as T. pallidum and B. burgdorferi. As such, they are thin-coiled bacterium that are difficult to detect via traditional staining methods, and like B. burdorferi, it is a zoonotic disease. It is prevalent anywhere there is water, and has a worldwide distribution. Despite this, the prevalence varies from region to region, and is seen predominantly following rainfalls, lakes, in poor sanitary conditions, and following recreational water-related activities. For instance, a systematic review of 80 studies (1) found that the incidence ranged from 0.10 to 975 annual cases per 100,000 patients, with those with active surveillance having a higher morbidity compared to those with passive surveillance systems (12.09 vs 2.13 per 100,000 people). The most cases occurred in the tropical regions, usually in the Carribean, Oceania, South East Asia And East Sub-Saharan Africa. The estimated number of cases annually was 1,030,000 with over 58,000 deaths:
In a matched multivariate analysis of an outbreak of leptospirosis that occurred in Nicaragua in 1995 following a series of tropical storms (2) found that cases were more likely to have been reported walking through creeks or swimming in rivers, or have rodents in household food storage areas:
36% of household contacts of case patients had titers >400 for one or more Leptospira serovars, and had a higher OR for having high titers when compared to controls (OR 1.8, 95% CI 0.8-4.1). Notably, more canines from cases were likely to have leptospira titers >400:
The classification of any bacteria is difficult, however to keep things a bit simple, we can classify the genus leptospira into L. interrogans, which is the pathogenic strain, and L. biflexa, the non-pathogenic strain. There are a number of serotypes within each species, which vary depending on where in the world you happen to be at.
Above is an example of the broad number of species within each group (5), which is not incredibly important to remember. The reservoir tends to be mice and rats, where leptospires gain access to the kidneys and colonize the brush border of the proximal renal tubular epithelium (3). From here, the host can shed leptospira from the urine for a prolonged period of time (up to months) and at high concentrations (107 organisms/mL). Humans, who contract the bacteria via cuts and abrasions or mucous membranes and via indirect contact (water) or direct contact (animals), are the accidental host (3, 4). It is rare for leptospirosis to be contracted via oral route or inhalation.
Path and Presentation
Following inoculation, patients will have high levels of leptospira which leads to the activation of the innate immune system and subsequently to that of the adaptive immune system. The incubation period ranges from 7 to 12 days (3) but can be as long as a month. There are a broad range of symptoms, but these are generally classified into two stages:
- First stage: typically driven by the leptospiremia and are very non-specific
- Second stage: also known as Weil’s disease. Occurs after a brief cessation of the initial stage, or can follow it immediately. Here you see renal failure, pulmonary hemorrhage, and liver failure/hyperbilirubinemia.
In the previously cited study from Nicaragua (2), cases tended to have headache, chills, MSK pain, vomiting, and dyspnea when compared to negative leptospirosis controls:
A study of 93 patients from an outbreak in Chonbuk in 1987 (6) found that the most common presentation included severe and abrupt fever, chill, headache, and myalgia, with a biphasic nature seen in 18% of cases:
3 types of presentations were described, which were pneumonitis, hepatitis, and nephritis. 40% presented with hemoptysis, and imaging demonstrating multiple patterns, mostly diffuse patchy opacities.
A case series of 353 laboratory confirmed cases of leptospirosis (7) found that the most common symptoms included fever (99%), myalgia (91%), headache (89%), chills (87%), anorexia (82%), and nausea/vomiting (77%). Conjunctival suffusion was found in 28%, while abdominal pain and backache was found in 51%. Elevated total bilirubin (>1mg/dl) was found in 70% of patients. A retrospective study of 40 patients evaluated several neurological abnormalities seen in leptospirosis (8) with most patients presenting with aseptic meningitis, followed by paraparesis, marked by initial flaccidity and subsequent hyperreflexia:
A review of 58 patients (9) found that 37 patients had some sort of pulmonary abnormality in imaging, usually appearing between 3rd and 7th day. The most common abnormalities were multiple nodular opacities (57%), consolidation (16%), and diffuse, ground-glass opacities (27%). Most lesions were peripheral (46%). To summarize:
As you can see, the most common signs and symtoms in multiple studies include:
- Headache (70-98%)
- Myalgia (40-97%)
- Fever
Risk Factors for Severe Leptospirosis
As mentioned previously, the disease tends to be biphasic, with the second one being the severe form aka Weil’s disease. This is largely driven by the cytokine storm (10). Furthermore, the serotype seems to play a role with a retrospective study finding that patients infected with icterohaemorrhagiae being more likely to have a severe presentation (2):
An observational, retrospective case-control study (11) evaluated patients with severe leptospirosis and mild leptospirosis. More patients with severe leptospirosis had jaundice, conjunctival suffusion, and oliguria compared to non-severe controls:
Factors associated with severe leptospirosis in multivariate model included tobacco use, L. interrogans infection, and delay of antibiotic therapy >2 days:
Multivariate analysis found that thrombocytopenia, elevated creatinine/lactate, and elevated amylase were also associated with severe leptospirosis:
Given that acute lung injury is another severe manifestation of leptospirosis, a prospective cohort study evaluated patients with leptospirosis who required ICU admission and mechanical ventilation to evaluate its risk factors (12). 690 patients were evaluated, of which 42 were transferred to the ICU for mechanical ventilation. Multivariate analysis found that shock, elevated creatinine, and hyperkalemia were independent risk factors for acute lung injury:
A retrospective study from Australia (13) evaluated 402 cases and found that patients with predictors of severe leptospirosis on multivariate analysis included abnormal ascultation on respiratory exam (OR 8.9, 95% CI 3.5-22.4), oliguria (8.2, 95% CI 3.2-21.1), hypotension (3.8, 95% CI 1.5-9.9), and creatinine >2mg/dL (7.0, 95% CI 2.7-18.1). These variables were put together to create the SPiRO score (SBP <100, resp auscultation abnormalities, oliguria), with each variable representing one point:
The utility of the score comes from a score of 0, with a NPV for severe disease of 97.2%:
One thing to note is this is a single center study, which may not be widely applicable.
In terms of risk factors for mortality, these tend to be fairly straight forward. One study of 468 suspected cases (14; case fatality rate 6.03%) found that that risk factors for mortality included low urine output, disorientation, jaundice, dyspnea, and meningismus:
Furthermore, hyperbilirubinemia and hyperkalemia were the labs more associated with mortality:
In a cohort of 68 patients with leptospirosis (mortality rate of 18%), multivariate regression analysis found that dyspnea, oliguria, alveolar infiltrates, and repolarization abnormalities were associated with mortality (15):
Furthermore, those with higher mean radiological score were more likely to have not survived:
Finally, a Brazilian retrospective study (16) evaluated 86 cases of leptospirosis and found an overall mortality of 22%. Univariate analysis found that the presence of rales, cardiac arrhythmia, older age, oliguria, and female gender were associated with higher mortality, but only oliguria was independently associated with mortality in multivariate analysis:
Furthermore, the frequency of dyspnea, pulmonary rales, and cardiac arrhythmia increased during the study period, though mortality decreased during the same time frame:
When controlled for age and gender, there was no difference and the frequency of therapy was significantly lower in 1994, during an outbreak, compared to the periods before and after. Again, worse labs = worse outcome, so not really surprising but I think it allows you to get an idea of risk of mortality.
Diagnosis:
The diagnosis of leptospirosis tends to be difficult for the following reasons:
- It looks like anything else! If you’re in malaria country, you look for malaria. If you have transaminitis, you look for hepatitis. If you are in the winter, you look for influenza and now COVID.
- A lot of the complications, especially in severe leptospirosis, looks like full blown septic shock. In particular, pulmonary hemorrhage. As a result, in endemic areas, people also look for hemorrhagic fevers
- Let’s be honest, we all are terrible at history taking, so we won’t even think to ask about water exposure and when we do, we think of legionella.
- Conjunctival suffusion, the thing that is “classic” for leptospirosis, is only seen in about 40% of cases.
Add to that the fact this is a spirochete (see my post on lyme disease and syphilis diagnostics), you can see the difficulty here. If you remember those posts, the spirochetes are very thin and difficult to culture and isolate. Leptospires are usually isolated from blood and CSF during the first 10 days of illness, when patients are bacteremic (4, 17), however they can take several weeks to grow and special media (the Ellinghausen-McCullough-Johnson Harris media) is required. These can also be isolated from urine at around the third week of illness (18). Along with their specific culture media, it seems that the specific type of culture bottle can also influence the longevity of the spirochetes. In one study evaluating 9 different commercial blood culture bottles at 2 different temperatures (30 C and 37C), the Southern Group blood culture system had the longest longevity at 30 C, though the more common BACTEC system was able to keep most of these serovars around for at least a week when cultured at the normal 37C (19).
Similar to Syphilis, direct detection by dark-field microscopy is another avenue to diagnose patients (using light and shadows to “stain” the still living spirochetes), however this requires experience with looking at dark-field microscopy and the actual apparatus (I’ll say this again, who has one of these in 2021?). As a result, the microagglutination test and the ELISA test have been the most common, indirect methods of diagnostic modality used.
What is the microagglutination test? This method allows you to get a patient’s serum and mix it with a culture of leptospires and then evaluate the degree of agglutination using a dark-field microscope, usually 50% agglutination (20). These usually take around 10-12 days to turn positive, and can turn positive as early as 5-7 days after illness, but there are several issues here. First, the boogeyman, the dark-field microscope. Second, many times, the culture of leptospires needs to be alive (the WHO mentions that killed antigens are more sensitive but less specific, but this is an issue) so there is some difficulty in handling them and keeping them alive to perform the test, so outside of referral centers, this test may not be widely available. Third, the test is only as good as however many serogroups you are able to get into it. In other words, if you live in the US and you have someone from South East Asia, there is a chance you’ll get a false negative as the US assay does not have a live serogroup that would agglutinate with the patient’s sera (2). Whether that last point is true or not is contested, but the first 2 are enough to mean this type of test is likely not going to be available everywhere. Lastly, you typically need paired sera (acute and convalescent) with a four-fold rise in titers, as one single titer is not diagnostic (4). The one exception to this is if the patient is presenting with acute symptoms and the titer is >1:800. While this may still be used in certain parts of the world and updated microtechnique that is as good but more reliable has been described before (21), given that you will not see this much, I won’t talk about it. I would encourage you to read the WHO document, as it gives a good overview as to how it is done.
ELISA is the more updated method of diagnosis. Still indirect, but ELISA tends to be more widely available and does not require the maintenance of a live set of leptospires to perform. In one study (22), blood samples were taken from patients at the time of admission, 4-7 days after admission (aka the acute phase) and then around 28 days (convalescent phase) after disease onset. MAT and IgM ELISA were compared. 321 cases of confirmed leptospirosis along with 317 non-leptospirosis cases were compared. The sensitivity of the ELISA test was higher during the first acute phase specimen and the second acute phase specimen compared to MAT, and all samples increased in sensitivity during the convalescent phase:
Another study (23) compared a rapid ELISA IgM with MAT and found that ELISA had a sensitivity of 100% when evaluating 41 specimens, with positive results seen as early as 3 days after the clinical onset of disease, with 12 positive ELISA tests detected before MAT antibodies, with 8/12 patients evaluated having positive IgM ELISA anywhere from 2 up to 48 months later. Furthermore, there was a false positive test in 17/292 patients who had an alternative diagnosis, including Brucella, EBV, Q fever, and CMV.
Two rapid test, an IgM-dipstick assay, and an indirect hemagglutination assay (IHA) have also been evaluated, with one study (24) finding that the IgM- dipstick had a sensitivity of 98% for acute leptospirosis with a specificity of 90.6% and a PPV of 98%. The IHA had a sensitivity, specificity, and PPV of 92.2%, 94.4%, and 95.9%, respectively. When looking at A1 samples, taken on the day of admission, 71% of the IgM-dipstick were positive compared to 49% of IHA samples. The sensitivity for the A1 sample was 35% for MAT, 67% for IgM-ELISA, 71% for IgM-dipstick and 49% for IHA, with the earliest positive sample seen in both IgM assays:
A larger, multinational study (25) evaluated the LEPTO dipstick assay with MAT and ELISA IgM and found fairly good sensitivity/specificity overall that increased when the test was drawn more than 10 days after the onset of illness:
Notably, the countries had different ranges of prevalence, suggesting this is a good overall diagnostic test.
Recently, PCR has emerged as an alternative option for diagnosis. In a study of 185 patients with leptospirosis (26) tested on admission, MAT was able to detect 26% of cases compared to 38% by PCR. Here, two targets, lipL32 by qPCR/16s and rrs by nested PCR, were used. Another study (27) using similar targets in 400 Slovenian patients found that conventional and RT-PCR results were concordant in 94% of the samples. Notably, these used both blood and urine suggesting the utility of a less invasive mode of diagnosis:
Therapy:
The utility of antibiotics has been difficult to demonstrate, with one small open label trial of 41 patients not finding any benefit of IV penicillin on disease outcomes (28). A meta-analysis (29) evaluated 5 randomized trials comparing placebo with IV penicillin and found no difference in outcomes when it comes to mortality (OR 1.59, 95% 0.59-4.29), fever days, oliguria (OR 1.795, 95% CI 0.325-9.929) and those who underwent renal replacement therapy (OR 1.59, 95% CI 0.92 to 2.731). Despite this, it is recommended that antibiotics are initiated as soon as possible (for obvious reasons). In one study, duration of illness was significantly shortened when treatment was initiated within 6 days (7):
While penicillin is the treatment of choice, other beta-lactams such as ceftriaxone and cefotaxime, and even doxycycline, have been compared with penicillin and seem to be equally as (in)effective. A randomized controlled trial (30) evaluated ceftriaxone 1g daily and penicillin G 1.5 million units q6h for 7 days. Primary endpoint time to resolution of fever. 173 patients were randomized, with no difference between groups in terms of time to resolution of fever (absolute difference, 0, 95% CI -0.2 to 0.2):
There was also no difference in terms of organ dysfunction. Another trial (31) compared cefotaxime, doxycycline, and penicillin G in patients with severe leptospirosis. Amoxicillin was used as an oral stepdown for the cefotaxime and penicillin groups, while the doxycycline IV group got doxycycline PO. otal duration was 7 days. 264 patients were enrolled, with similar outcomes in terms of death, clinical treatment failure, and duration of treatment between the 3 groups.
There was no difference in death or duration of hospitalization:
There was no difference in the duration of renal/hepatic dysfunction between groups, though one notable thing was that duration of fever after therapy was associated with multiorgan dysfunction: 60h fever for patients with one organ dysfunction and 84h for those with 2 organ dysfunction.
A multicenter, open label, randomized trial (32) evaluated therapy with doxycycline and azithromycin in both leptospirosis and scrub typhus. Doxycycline loaded with 200mg followed by 100mg q12h for seven days, while azithromycin was loaded with 1g and then 500mg daily for 3 days. 296 patients were enrolled overall, of which 69 were diagnosed with leptospirosis. There was no difference in treatment outcome between groups (96.5% treatment success in the doxycycline group vs 97.4% in the azithromycin group), with similar time to defervescence within the leptospirosis subgroup:
It seems that, at best, therapy with either a beta-lactam or atypical coverage does not worsen outcomes, but the verdict is still out as to whether there is any benefit. Despite this, the recommendation is to initiate some sort of antibiotic therapy, especially in severe cases.
As I noted before, it seems that cytokine storm may be the underlying pathogenic principle behind the icteric form of the disease and due to this, one would expect that steroids would be of benefit. A meta-analysis of 4 studies does not show this to be the case unfortunately (33), with the exception of one study where methylprednisone was given to pulmonary leptospirosis (34). Here, 30 cases of severe pulmonary leptospirosis requiring ICU care were evaluated. The first 13 cases did not receive steroids, while the next 17 did (methylpred 1gm per day followed by prednisolone 1mg/kg for 7 days). The mortality in the steroid group was significantly lower than in the non-steroid group, even when evaluating the sickest patients by APACHE II scores:
Unfortunately, the single-center nature of it, the lack of randomization, and the introduction of the steroid regimen 13 patients into the outbreak means not a lot of conclusions could be drawn here.
TL;DR
- Leptospirosis is a spirochete, meaning it will be difficult to detect off blood cultures
- Presentation is similar to other diseases such as malaria, hepatitis, influenza
- Look for elevated bilirubin, hypokalemia, conjunctival suffusion, and myalgia (especially lower back and calves) in context of any water exposure or presence of mice/rats. This is a tip off for leptospirosis. Another tip off is hemoptysis with an ARDS-like picture
- Another clue is a patient from Hawaii, the Caribbean, South East Asia, though keep in mind this is something it may be underreported.
- Diagnosis is based on microagglutination (usually in send-off labs) requiring either a four-fold rise in acute to convalescent titers or one single titer >800, ELISA test, or PCR.
- Penicillin, first gen cephalosporins, or doxycycline are recommended, but data for its efficacy is lacking.
- Use of the ‘roids is not widely recommended.
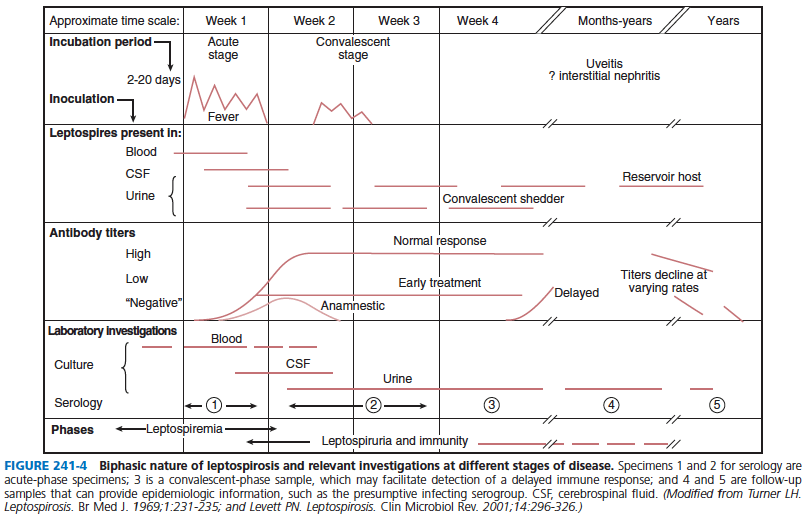
Summary table for presentation/diagnostics (4):
References:
- Costa F, Hagan JE, Calcagno J, Kane M, Torgerson P, Martinez-Silveira MS, Stein C, Abela-Ridder B, Ko AI. Global Morbidity and Mortality of Leptospirosis: A Systematic Review. PLoS Negl Trop Dis. 2015 Sep 17;9(9):e0003898. doi: 10.1371/journal.pntd.0003898. PMID: 26379143; PMCID: PMC4574773.
- Trevejo RT, Rigau-Pérez JG, Ashford DA, McClure EM, Jarquín-González C, Amador JJ, de los Reyes JO, Gonzalez A, Zaki SR, Shieh WJ, McLean RG, Nasci RS, Weyant RS, Bolin CA, Bragg SL, Perkins BA, Spiegel RA. Epidemic leptospirosis associated with pulmonary hemorrhage-Nicaragua, 1995. J Infect Dis. 1998 Nov;178(5):1457-63. doi: 10.1086/314424. PMID: 9780268.
- Haake DA, Levett PN. Leptospirosis in humans. Curr Top Microbiol Immunol. 2015;387:65-97. doi: 10.1007/978-3-662-45059-8_5. PMID: 25388133; PMCID: PMC4442676.
- Bennett, Raphael Dolin, Martin J. Blaser. Mandell, Douglas, And Bennett’s Principles and Practice of Infectious Diseases. Philadelphia, PA :Elsevier/Saunders, 2015.
- Miotto BA, da Hora AS, Taniwaki SA, Brandão PE, Heinemann MB, Hagiwara MK. Development and validation of a modified TaqMan based real-time PCR assay targeting the lipl32 gene for detection of pathogenic Leptospira in canine urine samples. Braz J Microbiol. 2018 Jul-Sep;49(3):584-590. doi: 10.1016/j.bjm.2017.09.004. Epub 2017 Dec 2. PMID: 29233483; PMCID: PMC6066729.
- Park SK, Lee SH, Rhee YK, Kang SK, Kim KJ, Kim MC, Kim KW, Chang WH. Leptospirosis in Chonbuk Province of Korea in 1987: a study of 93 patients. Am J Trop Med Hyg. 1989 Sep;41(3):345-51. PMID: 2802020.
- Katz AR, Ansdell VE, Effler PV, Middleton CR, Sasaki DM. Assessment of the clinical presentation and treatment of 353 cases of laboratory-confirmed leptospirosis in Hawaii, 1974-1998. Clin Infect Dis. 2001 Dec 1;33(11):1834-41. doi: 10.1086/324084. Epub 2001 Oct 23. PMID: 11692294.
- Panicker JN, Mammachan R, Jayakumar RV. Primary neuroleptospirosis. Postgrad Med J. 2001 Sep;77(911):589-90. doi: 10.1136/pmj.77.911.589. PMID: 11524519; PMCID: PMC1757912.
- Im JG, Yeon KM, Han MC, Kim CW, Webb WR, Lee JS, Han YC, Chang WH, Chi JG. Leptospirosis of the lung: radiographic findings in 58 patients. AJR Am J Roentgenol. 1989 May;152(5):955-9. doi: 10.2214/ajr.152.5.955. PMID: 2705352.
- Cagliero J, Villanueva SYAM, Matsui M. Leptospirosis Pathophysiology: Into the Storm of Cytokines. Front Cell Infect Microbiol. 2018 Jun 20;8:204. doi: 10.3389/fcimb.2018.00204. PMID: 29974037; PMCID: PMC6019470.
- Tubiana S, Mikulski M, Becam J, Lacassin F, Lefèvre P, Gourinat AC, Goarant C, D’Ortenzio E. Risk factors and predictors of severe leptospirosis in New Caledonia. PLoS Negl Trop Dis. 2013;7(1):e1991. doi: 10.1371/journal.pntd.0001991. Epub 2013 Jan 10. PMID: 23326614; PMCID: PMC3542117.
- Marotto PC, Nascimento CM, Eluf-Neto J, Marotto MS, Andrade L, Sztajnbok J, Seguro AC. Acute lung injury in leptospirosis: clinical and laboratory features, outcome, and factors associated with mortality. Clin Infect Dis. 1999 Dec;29(6):1561-3. doi: 10.1086/313501. PMID: 10585813.
- Smith S, Kennedy BJ, Dermedgoglou A, Poulgrain SS, Paavola MP, Minto TL, Luc M, Liu YH, Hanson J. A simple score to predict severe leptospirosis. PLoS Negl Trop Dis. 2019 Feb 13;13(2):e0007205. doi: 10.1371/journal.pntd.0007205. PMID: 30759081; PMCID: PMC6391019.
- Pappachan MJ, Mathew S, Aravindan KP, Khader A, Bharghavan PV, Kareem MM, Tuteja U, Shukla J, Batra HV. Risk factors for mortality in patients with leptospirosis during an epidemic in northern Kerala. Natl Med J India. 2004 Sep-Oct;17(5):240-2. PMID: 15638302.
- Dupont H, Dupont-Perdrizet D, Perie JL, Zehner-Hansen S, Jarrige B, Daijardin JB. Leptospirosis: prognostic factors associated with mortality. Clin Infect Dis. 1997 Sep;25(3):720-4. doi: 10.1086/513767. PMID: 9314467.
- Daher E, Zanetta DM, Cavalcante MB, Abdulkader RC. Risk factors for death and changing patterns in leptospirosis acute renal failure. Am J Trop Med Hyg. 1999 Oct;61(4):630-4. doi: 10.4269/ajtmh.1999.61.630. PMID: 10548299.
- Lane AB, Dore MM. Leptospirosis: A clinical review of evidence based diagnosis, treatment and prevention. World J Clin Infect Dis 2016; 6(4): 61-66 [DOI: 10.5495/wjcid.v6.i4.61]
- Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, Levett PN, Gilman RH, Willig MR, Gotuzzo E, Vinetz JM; Peru-United States Leptospirosis Consortium. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003 Dec;3(12):757-71. doi: 10.1016/s1473-3099(03)00830-2. PMID: 14652202.
- Palmer MF, Zochowski WJ. Survival of leptospires in commercial blood culture systems revisited. J Clin Pathol. 2000 Sep;53(9):713-4. doi: 10.1136/jcp.53.9.713. PMID: 11041063; PMCID: PMC1731247.
- World Health Organization. (2003). Human leptospirosis : guidance for diagnosis, surveillance and control. World Health Organization. https://apps.who.int/iris/handle/10665/42667
- Cole JR Jr, Sulzer CR, Pursell AR. Improved microtechnique for the leptospiral microscopic agglutination test. Appl Microbiol. 1973 Jun;25(6):976-80. doi: 10.1128/am.25.6.976-980.1973. PMID: 4736794; PMCID: PMC380950.
- Cumberland P, Everard CO, Levett PN. Assessment of the efficacy of an IgM-elisa and microscopic agglutination test (MAT) in the diagnosis of acute leptospirosis. Am J Trop Med Hyg. 1999 Nov;61(5):731-4. doi: 10.4269/ajtmh.1999.61.731. PMID: 10586903.
- Winslow WE, Merry DJ, Pirc ML, Devine PL. Evaluation of a commercial enzyme-linked immunosorbent assay for detection of immunoglobulin M antibody in diagnosis of human leptospiral infection. J Clin Microbiol. 1997 Aug;35(8):1938-42. doi: 10.1128/jcm.35.8.1938-1942.1997. PMID: 9230359; PMCID: PMC229880.
- Levett PN, Branch SL, Whittington CU, Edwards CN, Paxton H. Two methods for rapid serological diagnosis of acute leptospirosis. Clin Diagn Lab Immunol. 2001 Mar;8(2):349-51. doi: 10.1128/CDLI.8.2.349-351.2001. PMID: 11238220; PMCID: PMC96061.
- Smits HL, Ananyina YV, Chereshsky A, Dancel L, Lai-A-Fat RF, Chee HD, Levett PN, Masuzawa T, Yanagihara Y, Muthusethupathi MA, Sanders EJ, Sasaki DM, Domen H, Yersin C, Aye T, Bragg SL, Gussenhoven GC, Goris MG, Terpstra WJ, Hartskeerl RA. International multicenter evaluation of the clinical utility of a dipstick assay for detection of Leptospira-specific immunoglobulin M antibodies in human serum specimens. J Clin Microbiol. 1999 Sep;37(9):2904-9. doi: 10.1128/JCM.37.9.2904-2909.1999. PMID: 10449473; PMCID: PMC85409.
- Philip N, Affendy NB, Masri SN, Yuhana MY, Than LTL, Sekawi Z, et al. (2020) Combined PCR and MAT improves the early diagnosis of the biphasic illness leptospirosis. PLoS ONE 15(9): e0239069. https://doi.org/10.1371/journal.pone.0239069
- Podgoršek, D., Ružić-Sabljić, E., Logar, M. et al. Evaluation of real-time PCR targeting the lipL32 gene for diagnosis of Leptospira infection. BMC Microbiol 20, 59 (2020). https://doi.org/10.1186/s12866-020-01744-4
- Edwards CN, Nicholson GD, Hassell TA, Everard CO, Callender J. Penicillin therapy in icteric leptospirosis. Am J Trop Med Hyg. 1988 Oct;39(4):388-90. doi: 10.4269/ajtmh.1988.39.388. PMID: 3189700.
- Charan J, Saxena D, Mulla S, Yadav P. Antibiotics for the treatment of leptospirosis: systematic review and meta-analysis of controlled trials. Int J Prev Med. 2013;4(5):501-510.
- Panaphut T, Domrongkitchaiporn S, Vibhagool A, Thinkamrop B, Susaengrat W. Ceftriaxone compared with sodium penicillin g for treatment of severe leptospirosis. Clin Infect Dis. 2003 Jun 15;36(12):1507-13. doi: 10.1086/375226. Epub 2003 Jun 6. PMID: 12802748.
- Suputtamongkol Y, Niwattayakul K, Suttinont C, Losuwanaluk K, Limpaiboon R, Chierakul W, Wuthiekanun V, Triengrim S, Chenchittikul M, White NJ. An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis. Clin Infect Dis. 2004 Nov 15;39(10):1417-24. doi: 10.1086/425001. Epub 2004 Oct 26. PMID: 15546074.
- Phimda K, Hoontrakul S, Suttinont C, Chareonwat S, Losuwanaluk K, Chueasuwanchai S, Chierakul W, Suwancharoen D, Silpasakorn S, Saisongkorh W, Peacock SJ, Day NP, Suputtamongkol Y. Doxycycline versus azithromycin for treatment of leptospirosis and scrub typhus. Antimicrob Agents Chemother. 2007 Sep;51(9):3259-63. doi: 10.1128/AAC.00508-07. Epub 2007 Jul 16. PMID: 17638700; PMCID: PMC2043199.
- Rodrigo C, Lakshitha de Silva N, Goonaratne R, Samarasekara K, Wijesinghe I, Parththipan B, Rajapakse S. High dose corticosteroids in severe leptospirosis: a systematic review. Trans R Soc Trop Med Hyg. 2014 Dec;108(12):743-50. doi: 10.1093/trstmh/tru148. Epub 2014 Sep 28. PMID: 25266477.
- Shenoy VV, Nagar VS, Chowdhury AA, Bhalgat PS, Juvale NI. Pulmonary leptospirosis: an excellent response to bolus methylprednisolone. Postgrad Med J. 2006 Sep;82(971):602-6. doi: 10.1136/pgmj.2005.044255. PMID: 16954459; PMCID: PMC2585719.
